Advanced Synthesis of Macromolecule Difunctional Alpha-Hydroxy-Ketone Photoinitiators for Industrial UV Curing
Advanced Synthesis of Macromolecule Difunctional Alpha-Hydroxy-Ketone Photoinitiators for Industrial UV Curing
The global demand for high-performance ultraviolet (UV) curing systems has driven intense research into photoinitiators that balance reactivity with stability. Patent CN102020726B introduces a groundbreaking macromolecule difunctional group alpha-hydroxy-ketone photoinitiator that addresses critical limitations found in conventional small-molecule alternatives. This technology leverages a sophisticated multi-step organic synthesis pathway to construct a high-molecular-weight architecture that exhibits superior resin compatibility and drastically reduced volatility. By integrating bulky cyclohexyl groups and aromatic linkers, the resulting photoinitiator maintains robust photo-initiation activity while mitigating the migration issues that plague traditional products like DAROCUR 1173. For R&D directors and procurement specialists in the coatings and ink industries, this patent represents a viable route to next-generation additives that comply with increasingly stringent environmental and safety regulations regarding volatile organic compounds (VOCs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional small-molecule alpha-hydroxy ketone photoinitiators, while effective in initiating polymerization, suffer from inherent physicochemical drawbacks that limit their application in high-value sectors such as food packaging and medical device coatings. The primary issue is their relatively low molecular weight, which leads to significant volatility during the curing process and potential migration of unreacted initiator fragments into the cured matrix over time. This migration can cause undesirable odor, yellowing, and contamination, rendering the final product unsuitable for sensitive applications. Furthermore, the absorption spectra of these small molecules are often fixed, limiting their efficiency when used with specific light sources or in thick film applications where deeper penetration is required. The reliance on these legacy structures forces manufacturers to accept a trade-off between curing speed and final product purity, a compromise that is becoming unacceptable in modern high-performance material manufacturing.
The Novel Approach
The methodology disclosed in CN102020726B circumvents these issues by engineering a macromolecular structure that inherently suppresses volatility without sacrificing photochemical efficiency. By synthesizing a difunctional molecule where two active alpha-hydroxy ketone groups are tethered by a rigid aromatic and cyclohexyl backbone, the effective molecular weight is increased substantially. This structural modification results in a significant red shift in the absorption wavelength due to extended conjugation, allowing for higher light absorption efficiency and better matching with industrial UV lamps. The novel approach ensures that the photoinitiator remains anchored within the polymer network, effectively eliminating migration and odor problems. This strategic molecular design offers a clear pathway for producing high-purity UV curing additives that meet the rigorous demands of advanced electronic materials and specialty coatings.
Mechanistic Insights into the Multi-Step Synthesis Pathway
The synthesis of this advanced photoinitiator is a testament to precise organic construction, relying on a sequence of well-controlled reactions to build complexity from simple precursors. The process initiates with the activation of hexahydrobenzoic acid, converting it into a reactive acyl chloride species using chlorinating agents such as phosphorus trichloride or thionyl chloride. This activated intermediate then undergoes a Friedel-Crafts acylation with fluorobenzene in the presence of a Lewis acid catalyst, typically aluminum chloride, to install the cyclohexyl ketone moiety onto the aromatic ring. The presence of the fluorine atom is critical, as it serves as a leaving group for the subsequent nucleophilic aromatic substitution. This step-wise construction allows for the precise placement of functional groups, ensuring that the final molecule possesses the exact symmetry and electronic properties required for optimal photoinitiation performance.
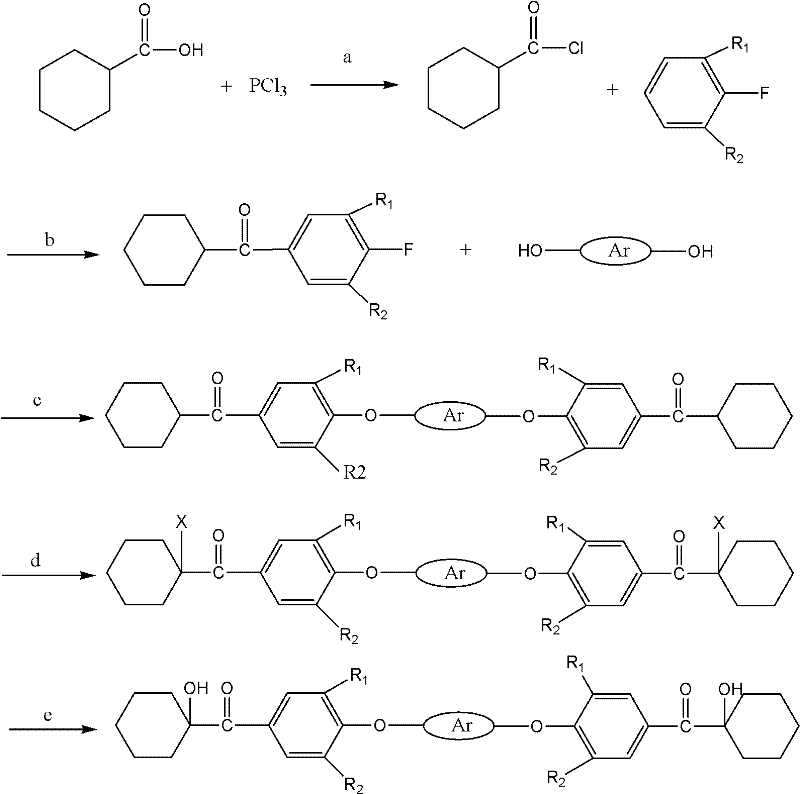
Following the formation of the fluorophenyl cyclohexyl ketone intermediate, the process advances to a nucleophilic substitution reaction with various diphenols, such as resorcinol or bisphenol A. This step links two ketone units together, forming the central 'macromolecular' core of the initiator. The reaction is catalyzed by bases like potassium carbonate in polar aprotic solvents, facilitating the displacement of the fluorine atoms. Subsequently, the alpha-position of the ketone groups is halogenated, introducing a reactive handle that is finally converted into the active hydroxyl group via hydrolysis. This final transformation yields the target alpha-hydroxy ketone functionality. The entire pathway is designed to maximize yield at each stage, with the patent reporting conversion rates exceeding 90% for key steps, demonstrating a robust and efficient chemical process suitable for industrial replication.
How to Synthesize Macromolecule Difunctional Alpha-Hydroxy-Ketone Photoinitiator Efficiently
Executing this synthesis requires careful control of reaction parameters, particularly temperature and stoichiometry, to minimize side reactions and ensure high purity of the final product. The initial acylation and substitution steps must be conducted under anhydrous conditions to prevent hydrolysis of the acid chloride or interference with the Lewis acid catalyst. Detailed operational guidelines regarding solvent selection, reaction times, and workup procedures are essential for reproducing the high yields described in the patent literature. For process chemists looking to implement this technology, understanding the nuances of the halogenation and hydrolysis steps is vital, as these determine the final activity of the photoinitiator.
- Preparation of cyclohexanecarbonyl chloride from hexahydrobenzoic acid using chlorination reagents like PCl3.
- Friedel-Crafts acylation with fluorobenzene under Lewis acid catalysis to form fluorophenyl cyclohexyl ketone.
- Nucleophilic substitution with diphenols to create the diketone intermediate backbone.
- Alpha-site halogenation followed by hydrolysis to yield the final alpha-hydroxy ketone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this synthesis route offers compelling economic and logistical benefits derived from the use of commodity feedstocks and scalable unit operations. The raw materials required, including hexahydrobenzoic acid, fluorobenzene, and common diphenols, are widely available in the global chemical market, reducing the risk of supply bottlenecks associated with exotic or proprietary intermediates. This accessibility translates directly into a more stable cost structure and enhanced negotiating power for purchasing teams. Furthermore, the synthetic pathway avoids the use of expensive transition metal catalysts or complex purification techniques like preparative HPLC, relying instead on standard distillation and recrystallization methods that are cost-effective at scale.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing high-yield reactions that minimize raw material waste and reduce the need for extensive downstream purification. By avoiding the use of precious metal catalysts and employing standard organic solvents, the overall production cost per kilogram is drastically simplified compared to complex organometallic routes. The high conversion rates reported in the patent embodiments suggest that material throughput can be maximized, leading to substantial cost savings in large-scale manufacturing environments without compromising on product quality.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals ensures a resilient supply chain that is less susceptible to the volatility of niche ingredient markets. Since the synthesis does not depend on single-source proprietary reagents, procurement managers can diversify their supplier base for raw materials, thereby mitigating the risk of production stoppages. The robustness of the chemical steps also implies consistent batch-to-batch quality, which is critical for maintaining long-term contracts with downstream formulators in the paint and ink industries who require reliable material specifications.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, utilizing reaction types that are easily transferred from laboratory glassware to industrial reactors. The process generates manageable waste streams that can be treated using standard effluent handling protocols, aligning with modern environmental compliance standards. The elimination of volatile small-molecule byproducts in the final application further enhances the environmental profile of the customer's end-product, supporting sustainability goals and regulatory compliance in regions with strict VOC limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photoinitiator technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this new material into their existing product lines.
Q: How does this macromolecule photoinitiator improve upon traditional small molecule initiators?
A: Unlike small molecule initiators like Irgacure 184 which suffer from high volatility and migration, this macromolecule design significantly reduces volatility and improves resin compatibility while maintaining high initiation efficiency through conjugation effects.
Q: What are the key raw materials required for this synthesis?
A: The process utilizes readily available commodity chemicals including hexahydrobenzoic acid, fluorobenzene, various diphenols (such as resorcinol or bisphenol A), and standard chlorinating agents.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the synthesis relies on standard organic unit operations such as acylation, substitution, and hydrolysis, which are well-established in fine chemical manufacturing and allow for straightforward scale-up from kilogram to tonnage levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Hydroxy-Ketone Photoinitiator Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced patent technologies like CN102020726B into commercial reality for the global coatings and polymer industries. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of photoinitiator meets the high-performance standards required for UV curing applications. We understand the critical nature of supply continuity and are committed to delivering high-purity alpha-hydroxy-ketone photoinitiators that drive innovation in your formulations.
We invite you to collaborate with our technical team to explore how this macromolecule photoinitiator can enhance your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and sourcing strategies, ensuring a seamless transition to this superior next-generation technology.
