Scalable One-Pot Synthesis of N-methyl-D-prolinol for High-Purity API Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for critical chiral intermediates, particularly those serving as the backbone for novel anti-tumor agents. Patent CN114874118A discloses a groundbreaking one-pot synthesis method for N-methyl-D-prolinol, a pivotal precursor in the manufacturing of Pyrotinib intermediates. This technical disclosure represents a significant leap forward in process chemistry, addressing long-standing inefficiencies associated with traditional multi-step protections. The structural integrity of the downstream product, 1-methylpyrrolidin-2-yl acrylic acid hydrochloride, relies heavily on the stereochemical purity of this alcohol intermediate. 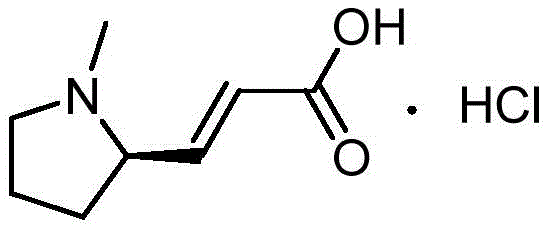 For procurement and supply chain leaders, understanding the nuances of this synthesis is crucial for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality at scale.
For procurement and supply chain leaders, understanding the nuances of this synthesis is crucial for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-methyl-D-prolinol has been plagued by cumbersome protection group strategies that inflate costs and complicate waste management. Prior art, such as the methods disclosed in WO2017186140A1, relies heavily on the use of tert-butyloxycarbonyl (Boc) protecting groups. 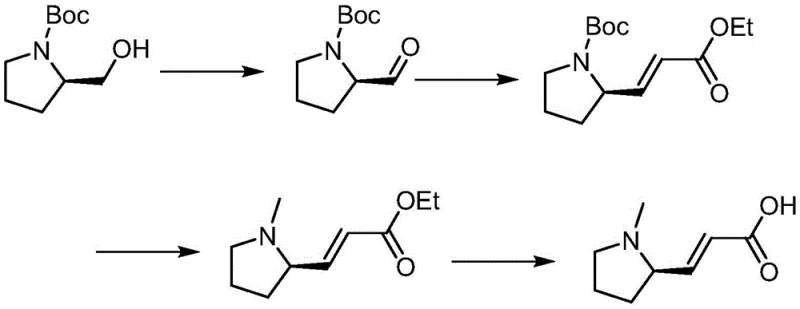 These conventional pathways necessitate distinct steps for Boc installation and subsequent removal, each adding operational time and requiring expensive reagents. Furthermore, these legacy methods often depend on column chromatography for purification, a technique that is notoriously difficult to scale and economically unviable for commercial manufacturing. The reliance on such purification methods introduces significant bottlenecks in production throughput and generates substantial solvent waste, contradicting modern green chemistry principles and increasing the overall cost of goods sold for the final API.
These conventional pathways necessitate distinct steps for Boc installation and subsequent removal, each adding operational time and requiring expensive reagents. Furthermore, these legacy methods often depend on column chromatography for purification, a technique that is notoriously difficult to scale and economically unviable for commercial manufacturing. The reliance on such purification methods introduces significant bottlenecks in production throughput and generates substantial solvent waste, contradicting modern green chemistry principles and increasing the overall cost of goods sold for the final API.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data eliminates the need for expensive protecting groups entirely, streamlining the workflow into a cohesive one-pot operation. By utilizing D-proline as the starting material, the process seamlessly integrates methylation, esterification, and reduction without isolating unstable intermediates. This strategic consolidation of reaction steps not only simplifies the operational protocol but also drastically reduces the consumption of auxiliary materials and solvents. The avoidance of Boc chemistry means that manufacturers can bypass the procurement of costly protecting reagents and the associated disposal costs. This methodological shift transforms the production landscape, offering a pathway that is inherently more suited for large-scale industrial application while maintaining rigorous control over impurity profiles and stereochemical integrity.
Mechanistic Insights into One-Pot Methylation and Reduction
The core of this technological advancement lies in the precise control of reaction conditions during the methylation and subsequent reduction phases. The process initiates with the methylation of D-proline using formaldehyde in the presence of a palladium on carbon catalyst under hydrogen pressure. This step is critical for establishing the N-methyl motif without racemization. Following this, the reaction mixture undergoes in-situ esterification where thionyl chloride is introduced at controlled temperatures not exceeding 15°C. 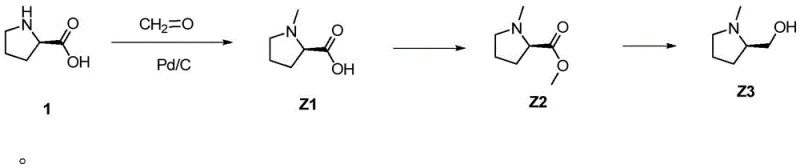 The careful regulation of temperature during the addition of thionyl chloride is paramount to preventing side reactions and ensuring the formation of the methyl ester intermediate Z2. The subsequent reduction using sodium borohydride is performed in a methanol-water system, where temperature control between 35-45°C ensures complete conversion while minimizing the formation of over-reduced byproducts. This mechanistic precision allows for the direct generation of the target alcohol with high fidelity.
The careful regulation of temperature during the addition of thionyl chloride is paramount to preventing side reactions and ensuring the formation of the methyl ester intermediate Z2. The subsequent reduction using sodium borohydride is performed in a methanol-water system, where temperature control between 35-45°C ensures complete conversion while minimizing the formation of over-reduced byproducts. This mechanistic precision allows for the direct generation of the target alcohol with high fidelity.
Impurity control is another cornerstone of this synthesis, achieved through the strategic manipulation of pH and phase separation techniques. After the reduction phase, the reaction is quenched with dilute hydrochloric acid, followed by careful neutralization to a pH of 9-10 using sodium hydroxide. This specific pH adjustment is vital for ensuring that the product remains in the free base form, facilitating efficient extraction into organic solvents like ethyl acetate. The protocol includes a salting-out step using sodium chloride to saturate the aqueous layer, which significantly enhances the partition coefficient of the product into the organic phase. This rigorous workup procedure ensures that inorganic salts and water-soluble impurities are effectively removed, resulting in a crude product that requires minimal further purification to meet stringent pharmaceutical specifications.
How to Synthesize N-methyl-D-prolinol Efficiently
Implementing this synthesis requires strict adherence to the sequential addition of reagents and temperature profiles outlined in the patent documentation. The process begins with the preparation of the N-methyl-D-proline intermediate, which serves as the foundation for the subsequent one-pot transformation. Operators must ensure that the methylation reaction proceeds to completion before introducing the esterification agents to avoid carryover of unreacted starting materials. The detailed standardized synthesis steps involve precise molar ratios, such as maintaining a D-proline to formaldehyde ratio of 1:1.1-1.2, to drive the equilibrium forward. For a comprehensive breakdown of the operational parameters and safety protocols required for execution, please refer to the technical guide below.
- Methylation of D-proline using formaldehyde and Pd/C catalyst under hydrogen pressure to form N-methyl-D-proline.
- In-situ esterification using thionyl chloride in methanol followed by neutralization to generate the methyl ester intermediate.
- Reduction of the ester using sodium borohydride in a controlled temperature environment to yield the final alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages that directly impact the bottom line and supply chain resilience for global pharmaceutical manufacturers. The elimination of the Boc protection group is not merely a chemical optimization but a significant economic driver that reduces raw material expenditure. By removing the need for column chromatography, the process enhances throughput capacity and reduces the reliance on specialized purification equipment, thereby lowering capital expenditure requirements. This streamlined approach translates into a more robust supply chain capable of meeting high-volume demands without the typical delays associated with complex multi-step purifications. For a reliable pharmaceutical intermediates supplier, adopting this methodology ensures consistent availability and competitive pricing structures.
- Cost Reduction in Manufacturing: The removal of expensive Boc reagents and the associated deprotection steps leads to substantial cost savings in raw material procurement. Additionally, the simplification of the workup process reduces solvent consumption and waste disposal costs, which are often hidden but significant expenses in fine chemical manufacturing. The ability to perform multiple transformations in a single vessel minimizes labor hours and equipment occupancy time, further driving down the cost per kilogram. These efficiencies allow for more aggressive pricing strategies while maintaining healthy margins, making the final API more cost-effective for downstream drug developers.
- Enhanced Supply Chain Reliability: The use of commodity chemicals such as thionyl chloride and sodium borohydride ensures that raw material sourcing is not subject to the volatility of specialized reagent markets. This accessibility mitigates the risk of supply disruptions and allows for better inventory management and planning. The robustness of the one-pot method also means that production schedules are less prone to delays caused by intermediate isolation failures or purification bottlenecks. Consequently, this leads to more predictable lead times and a stronger ability to fulfill large-scale orders consistently, reinforcing trust between the manufacturer and the procurement team.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, avoiding unit operations like chromatography that are difficult to translate from lab to plant. This scalability ensures that production can be ramped up quickly to meet market demand without compromising quality. Furthermore, the reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations, reducing the regulatory burden on the manufacturing site. This environmental compliance not only avoids potential fines but also enhances the corporate sustainability profile, which is a growing priority for multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of N-methyl-D-prolinol. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and quality standards associated with this synthesis. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their supply chain. The answers provided reflect the consensus on best practices for handling and sourcing this critical chemical building block.
Q: Why is the one-pot method superior to Boc-protected routes for N-methyl-D-prolinol?
A: Conventional routes require expensive Boc protection and deprotection steps along with column chromatography. The one-pot method eliminates these costly reagents and purification bottlenecks, significantly reducing production costs and waste.
Q: What is the expected purity and yield of this synthesis method?
A: According to patent data, the one-pot process achieves a total yield of approximately 75% to 79% with a final product purity exceeding 99.5%, ensuring suitability for sensitive pharmaceutical applications.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the elimination of column chromatography and the use of common reagents like thionyl chloride and sodium borohydride make this route highly scalable and operationally simple for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-methyl-D-prolinol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-methyl-D-prolinol meets the exacting standards required for API synthesis. Our commitment to technical excellence ensures that your production lines remain uninterrupted by quality deviations or supply shortages.
We invite you to collaborate with us to optimize your supply chain and reduce overall manufacturing costs through our advanced synthetic capabilities. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how our manufacturing prowess can support your long-term strategic goals. Let us be your partner in delivering high-purity pharmaceutical intermediates with unmatched reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
