Revolutionizing Atorvastatin Side Chain Synthesis: Safe, Ambient-Temperature Manufacturing for Global Supply Chains
The pharmaceutical industry's relentless pursuit of efficient statin synthesis has long been hindered by the complexities of constructing the chiral side chain required for Atorvastatin calcium. Patent CN108033899B introduces a transformative preparation method for (R)-6-cyano-5-hydroxy-3-carbonyl hexanoate tert-butyl ester, a critical intermediate that dictates the efficacy and purity of the final drug substance. Unlike conventional pathways that rely on hazardous alkali metals or energy-intensive cryogenic conditions, this novel approach leverages 1,8-diazabicycloundec-7-ene (DBU) as a non-nucleophilic base in 2-methyltetrahydrofuran (2-MeTHF). This strategic shift not only mitigates significant safety risks associated with pyrophoric reagents but also aligns with modern green chemistry principles by enabling reagent recovery and reducing the overall E-factor. For R&D directors and supply chain leaders, this patent represents a viable pathway to de-risk the manufacturing of high-purity pharmaceutical intermediates while optimizing the cost structure through simplified thermal management and solvent recycling protocols.
The Limitations of Conventional Methods vs. The Novel Approach
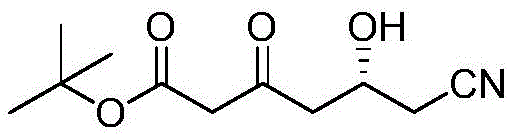
The Limitations of Conventional Methods
Historically, the synthesis of this key statin side chain has been plagued by severe operational hazards and thermodynamic inefficiencies. Traditional routes, such as those disclosed in prior art references, frequently depend on the use of strong, dangerous bases like sodium hydride (NaH), lithium diisopropylamide (LDA), or even elemental lithium and sodium metals. These reagents are inherently pyrophoric, posing catastrophic fire risks upon exposure to moisture or air, which necessitates expensive inert atmosphere engineering and specialized safety training for plant personnel. Furthermore, these reactions typically require cryogenic conditions, often demanding temperatures as low as -78°C maintained by liquid nitrogen cooling baths. The energy consumption required to sustain such low temperatures on a commercial scale is exorbitant, and the recovery of liquid nitrogen is technically difficult, leading to substantial operational expenditures. Additionally, the use of stoichiometric metal bases generates large quantities of inorganic salt waste, complicating downstream purification and creating significant environmental disposal burdens that conflict with increasingly stringent global regulatory standards.
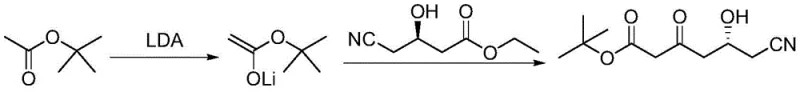
The Novel Approach
In stark contrast, the methodology outlined in CN108033899B circumvents these bottlenecks by employing a mild, organocatalytic strategy that operates comfortably at ambient temperatures. By utilizing DBU, a strong but non-nucleophilic amidine base, the process facilitates the necessary enolization of tert-butyl acetate without the need for cryogenic cooling. The reaction is conducted in 2-methyltetrahydrofuran, a solvent derived from renewable resources that offers excellent stability and facilitates easy workup. The procedure involves the pre-formation of an enolate solution at reflux followed by cooling to a manageable 20-30°C range, where the condensation with the chiral ester proceeds smoothly. This elimination of extreme thermal conditions drastically reduces the energy load on the manufacturing facility and removes the requirement for specialized low-temperature reactors. Moreover, the aqueous workup allows for the separation of the organic product while retaining the DBU in the aqueous phase, from which it can be recovered and reused, thereby transforming a consumable cost into a recyclable asset and significantly enhancing the economic viability of the process.
Mechanistic Insights into DBU-Mediated Claisen Condensation
The core chemical innovation lies in the precise control of the Claisen condensation mechanism using DBU in a biphasic-friendly solvent system. In this pathway, tert-butyl acetate is first treated with DBU in 2-MeTHF under reflux to generate the corresponding enolate species. Unlike lithium enolates which are highly reactive and unstable at higher temperatures, the DBU-generated enolate system possesses sufficient stability to be cooled to room temperature without premature decomposition or side reactions. When this enolate solution is mixed with (R)-4-cyano-3-hydroxy ethyl butyrate, the nucleophilic attack occurs selectively at the ester carbonyl, forming the beta-keto ester backbone essential for the statin structure. The use of 2-MeTHF is particularly advantageous as it stabilizes the transition state and prevents the racemization of the chiral center at the 5-position, ensuring high optical purity which is critical for the biological activity of the final Atorvastatin molecule. The reaction kinetics are managed through controlled dripping rates, preventing local exotherms that could degrade the sensitive cyano and hydroxy functionalities.
Impurity control is inherently built into this mechanistic design through the avoidance of harsh reagents that typically generate complex byproduct profiles. In metal-mediated reactions, over-reaction or metal-halogen exchange can lead to difficult-to-remove impurities that require extensive chromatography. Here, the mildness of DBU ensures that side reactions such as self-condensation of the starting ester or elimination of the hydroxy group are minimized. The subsequent workup involves the simple addition of water, which quenches the reaction and induces phase separation. The organic phase contains the crude product, while the aqueous phase retains the DBU salt. This clean separation profile means that the crude product requires less intensive purification, reducing the consumption of silica gel or recrystallization solvents. The ability to recover the solvent and the base from the aqueous layer further demonstrates the robustness of the mechanism, as it indicates that the reagents do not degrade into unrecoverable waste streams, maintaining a closed-loop material flow that is highly desirable for sustainable manufacturing.
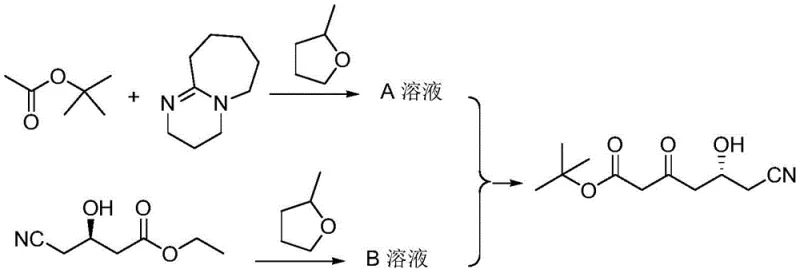
How to Synthesize (R)-6-cyano-5-hydroxy-3-carbonyl hexanoate tert-butyl ester Efficiently
Implementing this synthesis route requires precise adherence to the thermal profiles and mixing protocols defined in the patent to ensure maximum yield and purity. The process begins with the activation of tert-butyl acetate, followed by the controlled addition of the chiral substrate. The key to success lies in maintaining the reaction temperature within the narrow 20-30°C window during the addition phase to balance reaction rate with selectivity. Detailed standard operating procedures regarding pumping rates, stirring speeds, and quenching protocols are essential for technology transfer. For a comprehensive breakdown of the specific equipment setup and parameter optimization, please refer to the standardized synthesis steps provided in the technical guide below.
- Prepare Solution A by reacting tert-butyl acetate with 1,8-diazabicycloundec-7-ene in 2-methyltetrahydrofuran under reflux, then cooling to 25-30°C.
- Dissolve (R)-4-cyano-3-hydroxy ethyl butyrate in 2-methyltetrahydrofuran to obtain Solution B.
- Simultaneously drip Solution A and Solution B into 2-methyltetrahydrofuran at 20-30°C, stir for 1-5 hours, then quench with water for phase separation and solvent recovery.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented process offers profound strategic advantages beyond mere technical feasibility. The primary value driver is the drastic simplification of the manufacturing infrastructure. By eliminating the need for cryogenic cooling systems and specialized handling for pyrophoric metals, the capital expenditure (CAPEX) required for production facilities is significantly reduced. This allows for production in a wider range of multipurpose plants without the need for costly retrofitting. Furthermore, the operational expenditure (OPEX) is lowered through reduced energy consumption, as maintaining ambient temperature is far less costly than sustaining -78°C conditions over long reaction times. The safety profile improvement also translates to lower insurance premiums and reduced regulatory compliance burdens, making the supply chain more resilient against safety-related shutdowns.
- Cost Reduction in Manufacturing: The economic benefits are derived from the elimination of expensive, single-use reagents and the implementation of a circular solvent system. Traditional methods consume stoichiometric amounts of lithium or sodium bases which end up as waste salts. In this new process, the DBU base is recovered from the aqueous phase and can be recycled, effectively turning a variable cost into a fixed, amortized asset. Additionally, the use of 2-MeTHF allows for efficient solvent recovery, reducing the volume of fresh solvent required per kilogram of product. The removal of low-temperature operations also cuts utility costs substantially, as there is no need for liquid nitrogen or specialized chillers. These factors combine to create a leaner cost structure that provides a competitive edge in pricing negotiations for high-volume pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized reagents and the logistical challenges of transporting hazardous materials. Lithium and sodium metals are subject to strict transport regulations and can face supply shortages. DBU and 2-MeTHF are commodity chemicals with robust global supply chains, ensuring that production is not halted due to raw material scarcity. The simplified process also reduces the risk of batch failure due to temperature excursions, which are common in cryogenic reactions. A more robust process means higher first-pass yield and more predictable lead times, allowing procurement teams to maintain lower safety stock levels while still meeting production schedules. This reliability is crucial for just-in-time manufacturing models employed by major pharmaceutical companies.
- Scalability and Environmental Compliance: Scaling chemical processes often magnifies safety and environmental issues, but this technology scales linearly without exponential risk increase. The absence of pyrophoric reagents means that large-scale reactors do not require the same level of hazard mitigation as those used for lithiation. The green chemistry profile, characterized by solvent recycling and reduced salt waste, aligns perfectly with the sustainability goals of modern pharmaceutical companies. This compliance reduces the administrative burden of waste disposal permits and environmental reporting. The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to ramp up from pilot scale to multi-ton production with minimal engineering changes, ensuring that supply can meet market demand fluctuations efficiently.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific technical disclosures and beneficial effects outlined in the patent documentation. Understanding these details is crucial for evaluating the feasibility of adopting this technology within your existing manufacturing framework. The answers provided reflect the objective data and logical deductions based on the patented method's parameters and outcomes.
Q: How does this new method improve safety compared to traditional lithiation routes?
A: Traditional methods often utilize pyrophoric reagents like lithium metal, sodium hydride, or require cryogenic temperatures (-78°C) using liquid nitrogen. This patented process operates at ambient temperatures (20-30°C) using 1,8-diazabicycloundec-7-ene (DBU), eliminating fire hazards and the need for specialized low-temperature infrastructure.
Q: What are the solvent recovery capabilities of this process?
A: The process utilizes 2-methyltetrahydrofuran (2-MeTHF), a green solvent that allows for efficient phase separation upon water addition. Both the organic solvent and the DBU base can be recovered from the aqueous phase through concentration, significantly reducing material costs and environmental waste.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the elimination of cryogenic cooling and dangerous reagents makes this route highly scalable. The exothermic control is managed via dripping rates at ambient temperature, reducing the engineering complexity and energy consumption typically associated with scaling up low-temperature condensation reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-6-cyano-5-hydroxy-3-carbonyl hexanoate tert-butyl ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires a partner with deep technical expertise and robust manufacturing capabilities. Our CDMO team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this DBU-mediated process are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of this critical Atorvastatin intermediate meets the highest global pharmacopoeia standards. Our commitment to quality ensures that the chiral integrity and impurity profile of the material are consistent, safeguarding the efficacy of your final drug product.
We invite you to collaborate with us to optimize your supply chain for statin intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this safer, more efficient route. We encourage you to contact us to request specific COA data and route feasibility assessments tailored to your project's unique requirements. By leveraging our expertise, you can secure a reliable, cost-effective, and sustainable supply of high-value pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
