Scalable Synthesis of Biphenyl-Based Terphenyl Macrocycles for Industrial Applications
The landscape of advanced functional materials is constantly evolving, driven by the need for more efficient molecular recognition and separation technologies. A significant breakthrough in this domain is documented in patent CN112794794A, which details a novel preparation method for biphenyl aromatic hydrocarbon-based terphenyl macrocyclic compounds. This technology addresses long-standing challenges in macrocycle synthesis, specifically targeting the issues of low yield and complex reaction pathways that have historically hindered the widespread adoption of such sophisticated structures. By leveraging a strategic combination of Suzuki coupling and Lewis acid-catalyzed cyclization, this invention offers a robust pathway to produce high-purity 2,2',4,4',6,6'-hexamethoxy terphenyl arene macrocycles and their tetramethoxy counterparts. For R&D directors and procurement specialists in the specialty chemical sector, this represents a pivotal shift towards more reliable and cost-effective manufacturing of complex organic frameworks.
The implications of this patent extend far beyond the laboratory bench, offering tangible benefits for supply chain stability and cost management in the production of high-value intermediates. The ability to synthesize these macrocycles with enhanced efficiency means that downstream applications in adsorption, separation, and potentially even biological sensing can be realized with greater economic feasibility. As a leading manufacturer, we recognize that the transition from academic discovery to industrial reality requires not just chemical ingenuity but also process reliability. This patent provides the foundational chemistry needed to establish a reliable advanced materials supplier network, ensuring that critical components for next-generation separation technologies are available without the bottlenecks associated with traditional synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of macrocyclic compounds has been plagued by inherent inefficiencies that drive up costs and limit scalability. Traditional methods often rely on cumbersome multi-step sequences where each stage introduces potential yield losses and purification challenges. The formation of the macrocyclic ring itself is frequently the rate-limiting step, suffering from entropic penalties that result in poor conversion rates and significant formation of linear oligomers as byproducts. Furthermore, many existing protocols require harsh reaction conditions or expensive catalysts that are difficult to remove, leading to contamination issues that are unacceptable for high-purity applications. These factors collectively create a barrier to entry for many organizations seeking to utilize macrocyclic hosts in commercial products, as the cost reduction in advanced materials manufacturing becomes nearly impossible to achieve with legacy techniques.
The Novel Approach
In stark contrast, the methodology outlined in CN112794794A introduces a streamlined, one-pot synthetic strategy that fundamentally alters the economic equation of macrocycle production. By first establishing a robust monomer synthesis via Suzuki coupling and then proceeding directly to cyclization using paraformaldehyde and a Lewis acid catalyst, the process eliminates the need for intermediate isolation and purification steps that typically drain resources. This approach not only simplifies the operational workflow but also significantly enhances the overall yield by minimizing material handling losses. The use of readily available starting materials such as dibromobenzene and methoxyphenylboronic acids ensures that the supply chain remains resilient against raw material fluctuations. For procurement managers, this translates to a more predictable cost structure and a reduced risk of production delays, making the commercial scale-up of complex macrocyclic compounds a viable reality.
Mechanistic Insights into Suzuki Coupling and Lewis Acid Catalysis
The core of this technological advancement lies in the precise orchestration of two distinct chemical transformations. The initial phase involves a palladium-catalyzed Suzuki coupling reaction, which is renowned for its tolerance of functional groups and high fidelity in forming carbon-carbon bonds. In this specific application, 1,4-dibromobenzene is coupled with either 2,4,6-trimethoxyphenylboronic acid or 2,3-dimethoxyphenylboronic acid to generate the requisite terphenyl monomers. This step is critical as it establishes the structural backbone of the final macrocycle, ensuring that the methoxy substituents are positioned correctly to facilitate the subsequent ring-closing reaction. The efficiency of this coupling is paramount, as any impurities carried forward can compromise the cyclization efficiency, underscoring the need for stringent quality control during the monomer preparation phase.
Following monomer synthesis, the system undergoes a Lewis acid-catalyzed cyclization using boron trifluoride diethyl etherate. This catalyst activates the paraformaldehyde, generating an electrophilic species that attacks the electron-rich aromatic rings of the terphenyl monomer. The reaction proceeds through a series of electrophilic aromatic substitutions that ultimately close the ring, forming the stable macrocyclic structure. The presence of multiple methoxy groups plays a dual role here: they activate the ring towards electrophilic attack and provide steric guidance that favors cyclization over polymerization. 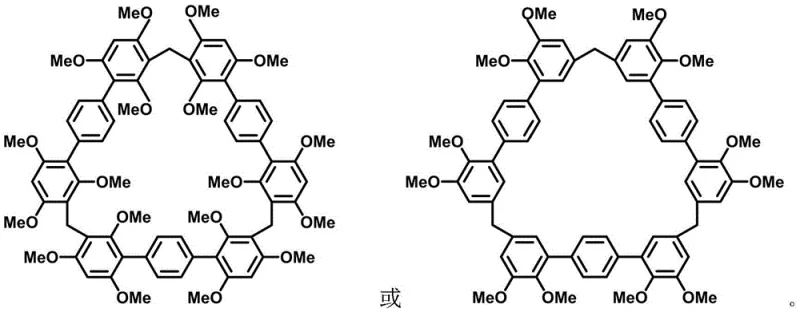 The resulting structures, as depicted in the visual data, showcase the symmetrical arrangement of the methoxy groups which is essential for their function in host-guest chemistry. This mechanistic understanding allows chemists to fine-tune reaction conditions to maximize the formation of the desired macrocycle while suppressing oligomeric side products.
The resulting structures, as depicted in the visual data, showcase the symmetrical arrangement of the methoxy groups which is essential for their function in host-guest chemistry. This mechanistic understanding allows chemists to fine-tune reaction conditions to maximize the formation of the desired macrocycle while suppressing oligomeric side products.
How to Synthesize Terphenyl Macrocyclic Compound Efficiently
Implementing this synthesis in a production environment requires careful attention to reaction parameters and purification protocols to ensure consistent quality. The process begins with the preparation of the terphenyl monomer, followed by the one-pot cyclization in a solvent system such as 1,2-dichloroethane. Detailed standard operating procedures are essential to maintain the delicate balance between reaction rate and selectivity. The ability to execute this synthesis reliably is a key differentiator for any organization aiming to lead in the supply of high-purity advanced materials.
- Prepare monomers via Suzuki coupling of dibromobenzene and methoxyphenylboronic acids.
- React monomers with paraformaldehyde in 1,2-dichloroethane using BF3·Et2O catalyst.
- Purify the resulting macrocyclic product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers profound advantages for supply chain optimization and cost management. The simplification of the synthetic pathway directly correlates to reduced operational expenditures, as fewer unit operations mean lower energy consumption and reduced labor requirements. Moreover, the high yield associated with this method ensures that raw material utilization is maximized, minimizing waste generation and the associated disposal costs. For supply chain heads, the use of commodity chemicals as starting materials reduces dependency on specialized vendors, thereby enhancing supply security and reducing lead time for high-purity macrocyclic compounds. This resilience is crucial in a volatile global market where continuity of supply is often as valuable as the product itself.
- Cost Reduction in Manufacturing: The elimination of complex multi-step purification processes significantly lowers the overall cost of goods sold. By avoiding the need for expensive transition metal removal steps often associated with other catalytic systems, the process achieves substantial cost savings without compromising on product quality. The one-pot nature of the cyclization step further reduces solvent usage and processing time, contributing to a leaner manufacturing footprint. These efficiencies allow for a more competitive pricing strategy while maintaining healthy margins, making the technology attractive for large-scale industrial adoption.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as dibromobenzene and boronic acids ensures that production is not held hostage by niche supply constraints. This accessibility means that inventory planning can be more accurate and that safety stocks can be maintained at lower levels without increasing risk. Additionally, the robustness of the reaction conditions means that production can be scaled up or down relatively quickly in response to market demand, providing a level of agility that is often lacking in fine chemical manufacturing. This flexibility is a key asset for maintaining strong relationships with downstream customers who require just-in-time delivery.
- Scalability and Environmental Compliance: The streamlined process inherently generates less waste, aligning with modern environmental, social, and governance (ESG) goals. The reduction in solvent volume and the avoidance of hazardous reagents simplify waste treatment protocols, reducing the environmental burden of the manufacturing process. Furthermore, the high efficiency of the reaction means that less energy is required per unit of product, contributing to a lower carbon footprint. These factors make the technology not only economically sound but also environmentally responsible, a combination that is increasingly demanded by global corporate buyers.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into their supply chains. The following questions address common inquiries regarding the feasibility, purity, and application of these macrocyclic compounds. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the primary advantages of this macrocyclic synthesis method?
A: The method utilizes a one-pot reaction strategy that significantly simplifies the process compared to traditional multi-step cyclizations, resulting in higher yields and easier purification.
Q: Can the methoxy groups on the macrocycle be further modified?
A: Yes, the presence of multiple methoxy side chains provides abundant reactive sites for further chemical derivatization and functionalization.
Q: Is this synthesis suitable for large-scale production?
A: The use of readily available raw materials and efficient one-pot conditions makes the process highly amenable to commercial scale-up and industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terphenyl Macrocyclic Compound Supplier
The transition from patent to product requires a partner with deep technical expertise and a proven track record in process development. NINGBO INNO PHARMCHEM stands ready to bridge this gap, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs ensuring that every batch meets the exacting standards required for advanced material applications. We understand that the successful commercialization of complex molecules like terphenyl macrocycles demands more than just chemical knowledge; it requires a holistic approach to manufacturing that prioritizes safety, efficiency, and consistency.
We invite you to explore how this technology can enhance your product portfolio and optimize your supply chain. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By collaborating with us, you gain access to a partner dedicated to driving innovation and delivering value through superior chemical manufacturing solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
