Advanced Heck Reaction Technology for Commercial Scale Trifluoromethyl Acetophenone Production
Advanced Heck Reaction Technology for Commercial Scale Trifluoromethyl Acetophenone Production
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high efficiency with environmental sustainability. Patent CN101161621A introduces a groundbreaking methodology for the preparation of trifluoromethyl acetophenone compounds, a critical structural motif found in numerous bioactive molecules. This technology leverages a modified Heck reaction protocol that replaces traditional homogeneous bases with a macroporous weakly basic styrene-based anion exchange resin. By integrating palladium catalysis with solid-phase base assistance, this approach addresses long-standing challenges in waste management and catalyst recovery. The innovation represents a significant leap forward for manufacturers aiming to optimize their production of fluorinated intermediates while adhering to stricter green chemistry standards. This report analyzes the technical merits and commercial implications of this novel synthetic pathway for global supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl acetophenone derivatives has relied on several conventional pathways, each carrying distinct operational burdens and safety risks. Traditional methods often involve Grignard reactions using trifluoromethyl cyanobenzene and methyl iodide, which require stringent anhydrous conditions and generate substantial magnesium salt waste. Alternative routes utilizing trifluoromethylbenzoic acid derivatives necessitate chlorination steps followed by reactions with cadmium reagents, introducing heavy metal toxicity concerns and complex wastewater treatment requirements. Furthermore, methods employing diazomethane pose severe explosion hazards and toxicity issues, making them unsuitable for large-scale industrial adoption. Even standard Heck reactions typically depend on soluble organic amines or inorganic bases, which complicate product isolation and generate significant saline waste streams. These legacy processes often struggle to meet modern cost-efficiency and environmental compliance targets simultaneously.
The Novel Approach
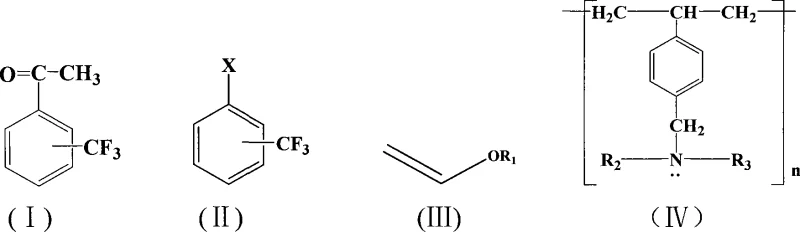
The patented methodology offers a transformative solution by utilizing a heterogeneous catalytic system that streamlines the reaction workflow and minimizes environmental impact. By employing a macroporous weakly basic styrene-based anion exchange resin as a solid base, the process eliminates the need for soluble organic amines or inorganic bases entirely. This substitution not only simplifies the work-up procedure but also enables the potential recycling of the resin support, thereby reducing raw material consumption over multiple batches. The reaction proceeds smoothly in aprotic polar solvents such as dimethyl sulfoxide or dimethyl formamide under moderate thermal conditions. This novel approach effectively mitigates the pollution problems associated with traditional chemical reaction processes while maintaining high reactivity. It stands as a prime example of green synthesis technology designed for scalable industrial application.
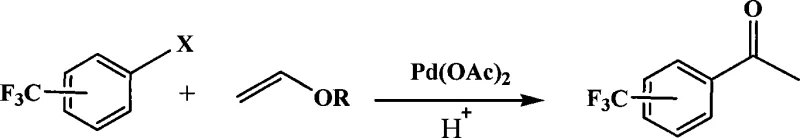
Mechanistic Insights into Resin-Assisted Palladium Catalysis
The core of this synthetic innovation lies in the synergistic interaction between the palladium catalyst and the solid support base within the reaction matrix. The palladium acetate catalyst facilitates the oxidative addition of the trifluoromethyl halobenzene, initiating the catalytic cycle essential for carbon-carbon bond formation. Unlike traditional systems where soluble bases neutralize the hydrogen halide byproduct in the bulk solution, the macroporous anion exchange resin acts as a localized proton scavenger. This heterogeneous interaction ensures that the basic sites are available at the interface where the catalytic turnover occurs, enhancing the overall reaction kinetics without introducing soluble impurities. The absence of organic phosphine ligands further simplifies the coordination sphere around the palladium center, reducing the risk of ligand-induced side reactions or catalyst deactivation. This mechanistic clarity allows for precise control over the reaction parameters, ensuring consistent performance across different batches of production.
Impurity control is significantly enhanced through the use of this solid-phase base system, which inherently limits the formation of soluble salt byproducts that often co-elute with the desired product. In conventional homogeneous base systems, the neutralization of acid byproducts generates stoichiometric amounts of salts that require extensive washing and purification steps to remove. By contrast, the resin-bound base traps the acidic species within its polymer matrix, allowing the organic product to remain in the solution phase with higher purity. This separation mechanism drastically reduces the burden on downstream purification processes such as column chromatography or crystallization. Consequently, the final trifluoromethyl acetophenone compounds exhibit purity levels exceeding 98%, meeting the rigorous standards required for pharmaceutical intermediate applications. The structural integrity of the resin, as depicted in the chemical diagrams, ensures stability under the reaction conditions of 50°C to 150°C.
How to Synthesize Trifluoromethyl Acetophenone Efficiently
Implementing this synthesis route requires careful attention to the stoichiometric ratios and reaction conditions outlined in the patent data to ensure optimal yield and purity. The process begins with the precise weighing of trifluoromethyl halobenzene and vinyl ether compounds, which are then dissolved in a selected aprotic polar solvent. The addition of the palladium catalyst and the specific type of styrene-based resin must be conducted under controlled stirring to ensure uniform dispersion throughout the reaction mixture. Heating is applied to reach the target temperature range, maintaining thermal stability to prevent degradation of the sensitive fluorinated intermediates. Detailed standardized synthesis steps see the guide below for exact operational parameters and safety precautions.
- Prepare the reaction mixture by combining trifluoromethyl halobenzene and vinyl ether compounds in an aprotic polar solvent such as DMSO or DMF.
- Add palladium acetate catalyst and macroporous weakly basic styrene-based anion exchange resin (e.g., D301 type) to the mixture.
- Heat the reaction to 50-150°C for 1-50 hours, then purify via acidification, extraction, and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resin-assisted Heck reaction offers tangible benefits in terms of operational expenditure and logistical reliability. The elimination of expensive organic phosphine ligands and small molecule organic amines directly translates to a reduction in raw material procurement costs. Furthermore, the ability to recycle the macroporous resin support introduces a circular economy element to the manufacturing process, decreasing the frequency of consumable purchases. The simplified work-up procedure, which avoids complex salt removal steps, reduces the consumption of solvents and energy during the purification phase. These factors collectively contribute to a more cost-effective production model that enhances the overall margin potential for high-volume manufacturing campaigns without compromising on product quality.
- Cost Reduction in Manufacturing: The removal of organic phosphine ligands and soluble bases eliminates significant cost centers associated with high-purity reagent procurement and waste disposal. By utilizing a recyclable solid base, the process reduces the recurring expense of base materials, leading to substantial cost savings over the lifecycle of the production campaign. The simplified purification workflow also lowers utility costs related to solvent recovery and wastewater treatment, further optimizing the cost structure. This economic efficiency makes the technology highly attractive for large-scale commercial production where margin optimization is critical.
- Enhanced Supply Chain Reliability: The reliance on commercially available macroporous resins and standard palladium salts ensures a stable supply of critical reagents, minimizing the risk of procurement bottlenecks. Unlike specialized ligands that may have long lead times or single-source dependencies, the materials required for this process are widely accessible in the global chemical market. This availability enhances supply chain resilience, allowing manufacturers to maintain consistent production schedules even during periods of market volatility. The robustness of the reaction conditions also reduces the risk of batch failures, ensuring reliable delivery of intermediates to downstream customers.
- Scalability and Environmental Compliance: The green chemistry attributes of this method, specifically the reduction of hazardous waste and the avoidance of toxic reagents like diazomethane, facilitate easier regulatory compliance in diverse jurisdictions. The process is inherently scalable, as the heterogeneous nature of the catalyst system allows for straightforward adaptation from laboratory to pilot and full commercial scale. The reduced environmental footprint aligns with corporate sustainability goals, making it a preferred choice for companies aiming to minimize their ecological impact. This scalability ensures that supply can be ramped up to meet increasing market demand without significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this technology into existing manufacturing lines. The answers reflect the objective performance metrics and operational advantages verified through the reported embodiments.
Q: What are the advantages of using anion exchange resin over traditional organic amines?
A: Using macroporous weakly basic styrene-based anion exchange resin eliminates the need for small molecule organic amines or inorganic bases, simplifying purification, reducing waste water treatment burden, and allowing for resin recycling.
Q: Does this method require expensive phosphine ligands?
A: No, the patented method operates effectively without organic phosphine ligands, which significantly reduces raw material costs and removes the need for complex ligand removal steps during downstream processing.
Q: What is the expected purity of the final trifluoromethyl acetophenone product?
A: The process described in the patent consistently yields products with purity greater than 98%, meeting stringent specifications required for high-quality pharmaceutical intermediate applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Acetophenone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing, leveraging advanced technologies like the resin-assisted Heck reaction to deliver superior pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of trifluoromethyl acetophenone meets the exacting standards of the global pharmaceutical industry. Our commitment to quality and consistency makes us a trusted partner for companies seeking reliable long-term supply solutions.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this efficient synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Please contact us to request specific COA data and route feasibility assessments for your project. We are dedicated to supporting your R&D and commercial goals with transparent communication and expert technical guidance.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
