Advanced Synthesis of 2-Aryl Allyl Alcohol via Ligand-Free Heck Reaction for Commercial Scale-up
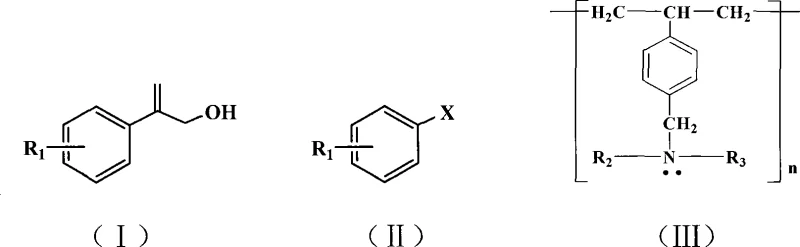
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for synthesizing critical intermediates. A significant breakthrough in this domain is documented in patent CN101177381B, which details a novel synthetic method for 2-aryl allyl alcohol compounds. This technology represents a paradigm shift from traditional homogeneous catalysis systems to a more robust heterogeneous approach. By utilizing a macroporous weakly basic styrene-based anion exchange resin as a solid base in conjunction with palladium acetate, this method successfully executes the Heck reaction between aryl halides and allyl alcohol compounds without the necessity of organic phosphine ligands. This innovation addresses long-standing challenges in process chemistry, particularly regarding catalyst recovery and waste minimization. For R&D directors and procurement managers alike, understanding the implications of this patent is crucial for optimizing supply chains and reducing the overall cost of goods sold for complex drug intermediates. The ability to operate under relatively mild conditions while achieving high conversion rates makes this technology a cornerstone for modern green chemistry initiatives in large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-aryl allyl alcohol derivatives via Heck coupling has relied heavily on homogeneous catalytic systems that require organic phosphine ligands and organic amine bases. While effective on a small laboratory scale, these conventional methods present severe drawbacks when translated to industrial production. The primary issue lies in the difficulty of removing residual phosphine ligands and palladium species from the final product, which is critical for pharmaceutical applications where heavy metal limits are strictly regulated. Furthermore, organic amines used as bases are often volatile, toxic, and challenging to recover, leading to significant environmental pollution and increased waste disposal costs. The separation processes required to purify the product from these homogeneous catalysts often involve complex chromatography or multiple extraction steps, which drastically reduce the overall throughput and increase the operational expenditure. Additionally, phosphine ligands are prone to oxidation, which can deactivate the catalyst and lead to inconsistent batch-to-batch reproducibility, posing a significant risk to supply chain reliability for high-value active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the methodology outlined in CN101177381B introduces a streamlined process that replaces soluble organic bases with a solid macroporous anion exchange resin. This substitution fundamentally alters the reaction dynamics, allowing for a heterogeneous system where the base is easily separated from the reaction mixture by simple filtration. The absence of organic phosphine ligands not only reduces the raw material cost but also eliminates a major source of product contamination, thereby simplifying the downstream purification process. This novel approach facilitates a cleaner reaction profile, as the solid resin acts as a scavenger for acid byproducts generated during the catalytic cycle, driving the equilibrium towards the desired product without introducing soluble impurities. The operational simplicity of this method means that reaction workups are less labor-intensive, requiring fewer solvent exchanges and reducing the total volume of hazardous waste generated. For manufacturing teams, this translates to a more robust process that is easier to control and scale, ensuring consistent quality and reducing the risk of production delays associated with complex purification bottlenecks.
Mechanistic Insights into Pd-Catalyzed Heck Coupling with Solid Base
The core of this technological advancement lies in the synergistic interaction between the palladium catalyst and the solid support base. In this ligand-free system, palladium acetate serves as the precatalyst, which is reduced in situ to the active Pd(0) species necessary for the oxidative addition step with the aryl halide. The macroporous weakly basic styrene-based anion exchange resin plays a dual role: it acts as a proton scavenger to neutralize the hydrogen halide produced during the reaction, and it provides a microenvironment that may stabilize the catalytic species. Unlike homogeneous bases that diffuse freely, the solid resin creates a localized basic environment that promotes the migratory insertion and beta-hydride elimination steps essential for forming the carbon-carbon double bond. This heterogeneous nature prevents the aggregation of palladium particles, maintaining high catalytic activity over extended periods. The structural integrity of the resin allows it to withstand the thermal stress of the reaction conditions, typically ranging from 50°C to 180°C, without degrading or leaching significant amounts of organic material into the product stream.
Furthermore, the impurity profile of the resulting 2-aryl allyl alcohol is significantly improved due to the absence of phosphine oxide byproducts, which are notoriously difficult to separate from the target molecule. The resin's porous structure allows reactants to access the active sites efficiently while trapping acidic impurities, effectively acting as an in-situ purification agent. This mechanism ensures that the final crude product has a higher purity level before any column chromatography or crystallization steps are applied. For quality control teams, this means fewer out-of-specification batches and a more predictable impurity spectrum, which is vital for regulatory filings. The ability to tune the resin properties, such as pore size and basicity, offers an additional layer of process optimization, allowing chemists to tailor the reaction conditions for specific substrates with varying electronic and steric properties. This level of control is essential for developing a versatile platform technology that can be applied to a wide range of pharmaceutical intermediates beyond the specific examples listed in the patent.
How to Synthesize 2-Aryl Allyl Alcohol Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and reaction parameters to maximize yield and efficiency. The process begins by dissolving the halogenated arene and allyl alcohol in a suitable aprotic polar solvent, such as dimethylformamide or sulfolane, which stabilizes the transition states involved in the catalytic cycle.
- Dissolve halogenated arene and allyl alcohol in an aprotic polar solvent such as dimethylformamide or sulfolane within a reaction vessel.
- Add palladium acetate catalyst and macroporous weakly basic styrene-based anion exchange resin to the mixture under stirring conditions.
- Heat the reaction mixture to between 50°C and 180°C for 1 to 30 hours, then acidify and extract to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ligand-free Heck reaction methodology offers substantial strategic advantages for procurement and supply chain management. The elimination of expensive and sensitive phosphine ligands directly reduces the bill of materials, contributing to a lower overall production cost without compromising on reaction efficiency. Moreover, the recyclability of the solid base resin means that a significant portion of the reagent cost can be amortized over multiple batches, further enhancing the economic viability of the process. For supply chain heads, the robustness of this heterogeneous system reduces the dependency on specialized handling equipment required for air-sensitive ligands, simplifying logistics and storage requirements. The simplified workup procedure also shortens the manufacturing cycle time, allowing for faster turnaround on customer orders and improved responsiveness to market demand fluctuations. These factors combined create a more resilient supply chain capable of sustaining high-volume production of complex intermediates with greater reliability.
- Cost Reduction in Manufacturing: The removal of organic phosphine ligands from the process equation results in direct raw material savings, as these reagents are often among the most expensive components in cross-coupling reactions. Additionally, the simplified purification process reduces solvent consumption and energy usage associated with extensive distillation or chromatography steps. The ability to recycle the solid resin base multiple times without significant loss of activity further drives down the variable costs per kilogram of product. This cumulative effect leads to a more competitive pricing structure for the final intermediate, providing a distinct margin advantage in a cost-sensitive market. By minimizing waste generation, the facility also benefits from reduced disposal fees and lower environmental compliance costs, adding another layer of financial efficiency to the operation.
- Enhanced Supply Chain Reliability: Utilizing commercially available and stable reagents like macroporous anion exchange resins mitigates the risk of supply disruptions often associated with specialized custom ligands. The robustness of the reaction conditions allows for greater flexibility in sourcing raw materials, as the process is less sensitive to minor variations in reagent quality compared to sensitive homogeneous systems. This stability ensures consistent production schedules and reduces the likelihood of batch failures that could delay downstream drug development timelines. For global supply chains, the ease of handling non-hazardous solid bases simplifies transportation and storage regulations, facilitating smoother international logistics. Consequently, partners can rely on a steady flow of high-quality intermediates, strengthening the overall security of the pharmaceutical supply network.
- Scalability and Environmental Compliance: The heterogeneous nature of this catalytic system is inherently more scalable than homogeneous alternatives, as heat and mass transfer issues are easier to manage in large reactors. The absence of toxic phosphine compounds aligns with increasingly stringent environmental regulations and corporate sustainability goals, reducing the regulatory burden on manufacturing sites. Waste streams are less contaminated with heavy metals and organic toxins, making treatment and disposal more straightforward and environmentally friendly. This green chemistry profile enhances the company's reputation and compliance standing, which is increasingly important for partnerships with major multinational pharmaceutical corporations. The process design supports a transition towards continuous manufacturing technologies, offering future opportunities for even greater efficiency and capacity expansion without significant capital reinvestment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for industry professionals.
Q: What are the advantages of using solid base resin over organic amines in Heck reactions?
A: Using macroporous weakly basic styrene anion exchange resin eliminates the need for difficult-to-recycle organic amines and toxic phosphine ligands, significantly simplifying post-treatment and reducing environmental waste.
Q: Can the anion exchange resin be recycled in this synthesis process?
A: Yes, the patent data confirms that the macroporous weakly basic styrene-based anion exchange resin can be recovered and recycled after the reaction is completed, enhancing process sustainability.
Q: What is the typical yield range for this ligand-free Heck reaction method?
A: Experimental examples in the patent demonstrate isolated yields ranging from 80% to 92%, indicating a highly efficient and robust synthetic route for various substituted aryl halides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryl Allyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemical market. Our technical team has extensively evaluated the ligand-free Heck reaction pathway described in CN101177381B and confirmed its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-aryl allyl alcohol meets the highest standards required for pharmaceutical applications. We are committed to leveraging this innovative chemistry to deliver superior value to our partners through enhanced quality and operational excellence.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments for your projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable 2-aryl allyl alcohol supplier dedicated to driving innovation and efficiency in your drug development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
