Advanced Optical Resolution of Ubenimex Intermediates for Commercial Scale-up
Introduction to Advanced Chiral Resolution Technologies
The pharmaceutical landscape for antitumor agents continues to evolve, with Ubenimex standing out as a critical aminopeptidase N inhibitor derived from Streptomyces cultures. As detailed in patent CN102391145B, the synthesis of high-quality Ubenimex relies heavily on the stereochemical purity of its key intermediates. The molecule possesses three chiral centers, making the control of optical isomers during synthesis a paramount challenge for process chemists. Traditional methods often struggle with the separation of diastereomers, leading to complex purification workflows that hinder commercial viability. This report analyzes a novel resolution method that utilizes water as a green solvent to achieve exceptional stereochemical purity, addressing the critical needs of a reliable ubenimex intermediate supplier.
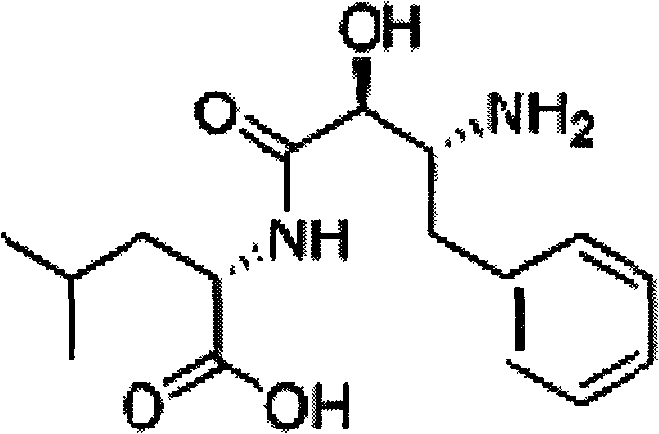
The core innovation lies in the efficient separation of the (2RS, 3SR)-3-acetamido-2-hydroxy-4-oxo-4-phenyl butyrate from its erythro-isomer counterparts. By leveraging specific solubility characteristics in an aqueous environment, this technology enables the production of intermediates with erythro-isomer content below 2 percent. Such high purity is essential for downstream processing, ensuring that the final active pharmaceutical ingredient meets stringent regulatory standards for safety and efficacy. For procurement and R&D teams, understanding this resolution pathway is key to securing a stable supply chain for this vital oncology therapeutic.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes, such as those described in J. Antibiot. 1983, typically involve the condensation of alpha-acetamido-methyl phenyl ketone with glyoxylic acid. While this reaction generates the necessary carbon skeleton, it inherently produces a mixture of four isomers: two threo forms and two erythro forms. In practice, the desired threo-isomer usually accounts for only 65 to 75 percent of the crude product, while the unwanted erythro-isomer constitutes a significant 22 to 32 percent impurity burden. Separating these diastereomers is notoriously difficult due to their similar physicochemical properties, often requiring multiple recrystallization steps with hazardous organic solvents or expensive chromatographic techniques.
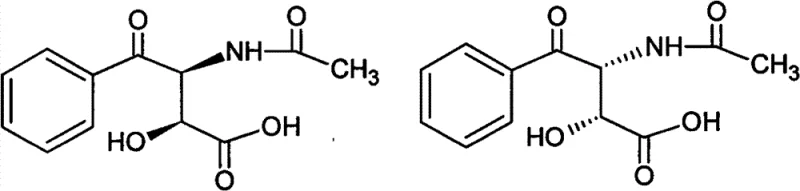
Furthermore, conventional purification strategies frequently rely on solvents like tetrahydrofuran, acetone, or ethyl acetate, which have been proven ineffective for this specific separation task. The inability to remove the erythro-impurity efficiently leads to yield losses in subsequent steps and complicates the final drug substance purification. From a manufacturing perspective, the use of volatile organic compounds increases operational costs related to solvent recovery and environmental compliance. These bottlenecks create significant friction in the supply chain, making cost reduction in pharmaceutical intermediates manufacturing a priority for industry stakeholders seeking more robust production methodologies.
The Novel Approach
The patented method introduces a paradigm shift by utilizing water as the exclusive recrystallization solvent. Contrary to initial expectations that organic media would be required for such a lipophilic intermediate, water uniquely facilitates the selective crystallization of the threo-isomer. The process involves dissolving the crude mixture in water at elevated temperatures, specifically between 50 and 100 DEG C, followed by a controlled natural cooling phase. This simple yet profound change in solvent system exploits the subtle solubility differences between the diastereomers, allowing the desired threo-form to precipitate rapidly while the erythro-impurity remains dissolved in the mother liquor.
Operational data indicates that maintaining a mass ratio of substrate to water between 1:8 and 1:10 yields optimal results. Unlike rapid cooling, which causes co-crystallization of impurities, natural cooling to ambient temperatures (20-30 DEG C) ensures high selectivity. The result is a dramatic improvement in purity, with the erythro-isomer content dropping to negligible levels, often below 0.1 percent in optimized embodiments. This approach not only simplifies the unit operations by eliminating solvent exchange steps but also aligns with green chemistry principles, offering a sustainable alternative for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Diastereomeric Crystallization
The success of this resolution strategy hinges on the distinct crystal lattice energies and solvation shells of the threo and erythro isomers in an aqueous environment. The target molecule, (2RS, 3SR)-3-acetamido-2-hydroxy-4-oxo-4-phenyl butyrate, possesses a specific spatial arrangement that favors intermolecular hydrogen bonding conducive to precipitation in water. In contrast, the erythro-isomers, characterized by different relative stereochemistry at the chiral centers, exhibit higher solubility in the aqueous phase under the specified cooling conditions. This differential solubility is the driving force behind the purification, effectively acting as a thermodynamic filter that excludes the unwanted stereoisomers from the solid phase.
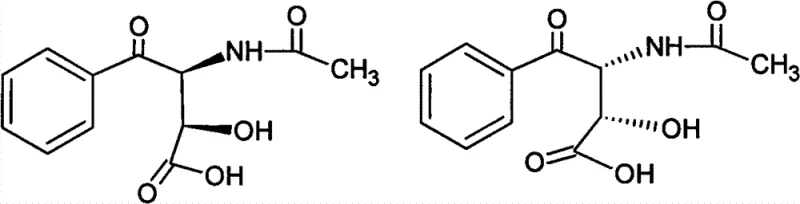
Understanding the kinetics of crystallization is equally critical for process control. The patent data reveals that the crystallization speed of the threo-isomer is significantly faster than that of the erythro-form during natural cooling. If the solution is cooled too rapidly, the system does not have sufficient time to discriminate between the isomers, leading to the entrapment of impurities within the crystal lattice. Therefore, the protocol mandates a slow, natural cooling trajectory to 10-40 DEG C. This kinetic control ensures that the growing crystals are composed almost exclusively of the desired threo-diastereomer, thereby achieving the high-purity specifications required for high-purity OLED material or pharmaceutical applications where stereochemical integrity is non-negotiable.
How to Synthesize 3-Acetamido-2-hydroxy-4-oxo-4-phenyl-butyrate Efficiently
Implementing this resolution technique requires precise control over dissolution and cooling parameters to maximize yield and purity. The process begins with the crude reaction mixture obtained from the condensation of alpha-acetamido-methyl phenyl ketone and glyoxylic acid. Operators must carefully manage the water-to-substrate ratio, as deviations can impact the saturation point and subsequent crystal growth. The following guide outlines the standardized operational procedure derived from the patent examples, ensuring reproducibility across different batch sizes.
- Dissolve the crude mixture containing erythro and threo isomers in water at a mass ratio of 1: 6 to 1:15, preferably 1:8 to 1:10.
- Heat the mixture to 50-100 DEG C with stirring until completely dissolved, then perform hot filtration to remove insoluble impurities.
- Allow the filtrate to cool naturally to 10-40 DEG C without rapid cooling to ensure selective crystallization of the threo-isomer while retaining the erythro-isomer in the mother liquor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this water-based recrystallization process offers tangible strategic benefits beyond mere technical feasibility. The elimination of expensive and hazardous organic solvents directly translates to a reduction in raw material expenditure and waste disposal costs. Furthermore, the simplicity of the operation—dissolve, heat, cool, filter—reduces the complexity of the manufacturing equipment required, lowering capital expenditure barriers for scale-up. This streamlined workflow enhances the overall agility of the supply chain, allowing for faster response times to market demands for ubenimex intermediates.
- Cost Reduction in Manufacturing: The substitution of organic solvents with water drastically lowers the cost of goods sold (COGS). Organic solvents require rigorous recovery systems and pose fire hazards, necessitating expensive infrastructure. By using water, the process eliminates these overheads and reduces the environmental burden associated with volatile organic compound (VOC) emissions. Additionally, the high selectivity of the crystallization minimizes product loss during purification, improving the overall mass balance and yield of the valuable chiral intermediate.
- Enhanced Supply Chain Reliability: Water is a universally available and inexpensive resource, insulating the production process from the price volatility and supply disruptions often seen with specialty organic solvents. The robustness of the method, which tolerates standard industrial cooling rates without requiring cryogenic conditions, ensures consistent production schedules. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers, reducing lead time for high-purity pharmaceutical intermediates and mitigating the risk of stockouts.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from kilogram to multi-ton production without fundamental changes to the chemistry. The use of water as the primary medium simplifies effluent treatment, as the mother liquor contains primarily biodegradable organic impurities rather than toxic halogenated or aromatic solvents. This alignment with environmental regulations facilitates easier permitting and long-term operational sustainability, making it an ideal candidate for green manufacturing initiatives in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this optical resolution technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on process capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of adopting this method for their specific production requirements.
Q: Why is water preferred over organic solvents for this recrystallization?
A: Experiments demonstrated that common organic solvents like tetrahydrofuran, acetone, and ethanol failed to effectively separate the erythro-isomer. Water uniquely exploits the solubility differences, allowing the threo-isomer to crystallize selectively while keeping the erythro-impurity in solution.
Q: What is the achievable purity of the threo-isomer using this method?
A: By optimizing the water ratio and cooling conditions, the content of the unwanted erythro-isomer can be reduced to less than 2 percent, with the desired threo-isomer content reaching up to 99.938 percent.
Q: How does this method impact industrial scalability?
A: The process eliminates the need for hazardous organic solvents and complex chromatographic separations. Using water simplifies waste treatment, reduces raw material costs, and allows for straightforward natural cooling crystallization, making it highly suitable for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ubenimex Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of stereochemical purity in the development of antitumor therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering ubenimex intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in chiral resolution and crystallization engineering allows us to optimize processes like the water-based method described, guaranteeing consistent quality for our global partners.
We invite pharmaceutical companies and contract research organizations to collaborate with us on optimizing their supply chains for complex oncology intermediates. By leveraging our technical proficiency, you can achieve significant process improvements and cost efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your project timelines and quality goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
