Advanced Synthesis of Nitrogenous Heterocyclic Inflammation Inhibiting Compounds for Commercial Scale
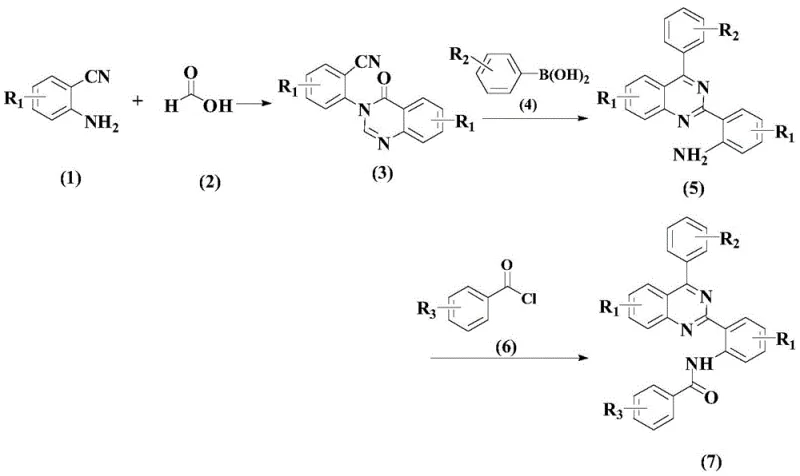
The pharmaceutical industry continuously seeks robust synthetic pathways for nitrogen-containing heterocyclic compounds due to their profound biological activities, particularly in the realm of inflammation inhibition. Patent CN108912059B discloses a groundbreaking synthetic method for a specific class of quinazoline derivatives, designated as Formula (7), which exhibit potent inhibitory effects on IL-6, a critical cytokine in inflammatory pathways. This technical insight report analyzes the novel three-step synthesis route detailed in the patent, highlighting its potential to revolutionize the production of high-purity pharmaceutical intermediates. The method leverages a palladium-catalyzed coupling strategy that circumvents the limitations of traditional oxidative cyclization, offering a cleaner, more efficient, and scalable solution for the manufacture of complex anti-inflammatory agents. By focusing on readily available raw materials and mild reaction conditions, this technology addresses key pain points in process chemistry, such as impurity control and operational safety, making it an attractive candidate for integration into existing supply chains for reliable pharmaceutical intermediates supplier networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazoline derivatives has relied heavily on methods that present significant challenges for industrial application. Traditional routes often employ strong oxidizing agents such as DDQ, MnO2, or NaClO to facilitate ring closure, which introduces severe safety hazards and complicates waste disposal protocols. These oxidative conditions can lead to over-oxidation of sensitive functional groups, resulting in complex impurity profiles that are difficult and costly to remove during purification. Furthermore, many conventional methods require harsh reaction conditions, including high temperatures or the use of corrosive reagents, which can degrade equipment and increase maintenance costs. The reliance on transition metals like copper in Ullmann-type reactions often necessitates rigorous metal scavenging steps to meet stringent regulatory limits for residual metals in active pharmaceutical ingredients. These factors collectively contribute to extended production cycles and elevated manufacturing costs, creating bottlenecks for procurement managers seeking cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the method disclosed in CN108912059B introduces a refined three-step sequence that prioritizes efficiency and selectivity. The core innovation lies in the second step, where a palladium-catalyzed coupling reaction is employed to construct the biaryl linkage under relatively mild thermal conditions ranging from 70°C to 90°C. This approach eliminates the need for hazardous strong oxidizers, thereby enhancing the environmental profile of the synthesis. The use of specific organic ligands and acidic additives ensures high conversion rates and minimizes the formation of by-products, streamlining the downstream purification process. The overall route is designed to utilize simple and readily available starting materials, which stabilizes the supply chain and reduces raw material procurement risks. By shifting from oxidative cyclization to a catalytic coupling strategy, this novel approach offers a pathway to significantly reduced operational complexity and improved batch-to-batch consistency.
Mechanistic Insights into Pd-Catalyzed Coupling and Ligand Effects
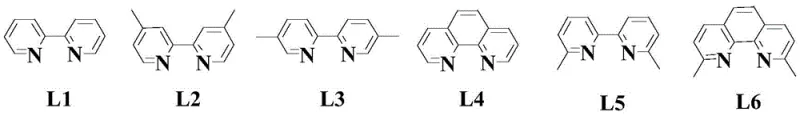
The success of the second step in this synthesis hinges on the precise orchestration of the palladium catalytic cycle. The reaction involves the coupling of a quinazolinone intermediate with an arylboronic acid, a transformation that requires careful tuning of the catalyst system to achieve high yields. The patent identifies palladium acetate as the preferred catalyst source, which generates the active Pd(0) species in situ. However, the true differentiator in this process is the selection of the organic ligand. Extensive screening revealed that Ligand L1, a bipyridine derivative, outperforms other structurally similar ligands such as L2 through L6. The superior performance of L1 can be attributed to its optimal bite angle and electronic properties, which stabilize the palladium center during the oxidative addition and transmetallation steps. This stabilization prevents catalyst deactivation and promotes the desired reductive elimination to form the carbon-carbon bond efficiently.
Furthermore, the inclusion of an acidic additive, specifically p-toluenesulfonic acid monohydrate, plays a critical role in facilitating the reaction progress. The acid likely assists in the activation of the boronic acid species or the protonation of intermediates, driving the equilibrium towards the product. Experimental data within the patent demonstrates that substituting this specific acid with others like benzoic acid or acetic acid results in no reaction or trace yields, underscoring the non-obvious nature of this formulation. The solvent choice is equally critical, with toluene proving to be the most effective medium, likely due to its ability to dissolve both organic substrates and the catalyst complex while maintaining the necessary reaction temperature without decomposition. This deep understanding of the mechanistic nuances allows for precise control over the reaction, ensuring the production of high-purity quinazoline derivatives with minimal impurity generation.
How to Synthesize Nitrogenous Heterocyclic Compounds Efficiently
The synthesis of these valuable inflammation-inhibiting compounds is achieved through a logical three-step sequence that balances reactivity with selectivity. The process begins with the cyclization of 2-aminobenzonitrile and formic acid to form the core quinazolinone scaffold, followed by the critical palladium-catalyzed arylation, and concludes with an acylation step to introduce the final amide functionality. Each step has been optimized for yield and purity, with specific attention paid to work-up procedures that avoid complex extractions. The detailed standardized synthesis steps see the guide below for operational specifics regarding molar ratios, temperature controls, and purification techniques that ensure reproducibility on a commercial scale.
- React 2-aminobenzonitrile with formic acid at 70-90°C to form the quinazolinone intermediate.
- Perform palladium-catalyzed coupling with arylboronic acid using Ligand L1 and p-toluenesulfonic acid in toluene.
- Complete the synthesis via acylation with benzoyl chloride in dichloromethane using potassium carbonate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the simplification of the manufacturing process, which directly translates to operational efficiency. By eliminating the need for hazardous oxidizers and reducing the number of purification steps required to remove metal residues, the overall production timeline is drastically simplified. This efficiency gain allows for faster turnover of batches and better utilization of manufacturing assets. Additionally, the use of readily available raw materials mitigates the risk of supply disruptions, ensuring a steady flow of intermediates necessary for continuous production schedules. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in input quality, further enhancing supply chain reliability.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous oxidizing agents significantly lowers the cost of goods sold. Furthermore, the high selectivity of the palladium-catalyzed step reduces the burden on purification resources, such as silica gel and solvents, leading to substantial cost savings in waste management and material consumption. The ability to operate at moderate temperatures also reduces energy consumption compared to high-temperature alternatives, contributing to a lower carbon footprint and reduced utility costs.
- Enhanced Supply Chain Reliability: The reliance on common chemical feedstocks like toluene, dichloromethane, and simple arylboronic acids ensures that raw material sourcing is not a bottleneck. These materials are widely available from multiple global suppliers, reducing dependency on single sources and enhancing negotiation leverage. The stability of the intermediates also allows for flexible inventory management, enabling the stocking of key precursors to buffer against market fluctuations and ensure uninterrupted supply for downstream API synthesis.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations that are easily transferred from pilot plant to commercial production. The avoidance of heavy metal waste streams and hazardous oxidizers simplifies environmental compliance and reduces the cost associated with waste treatment. This aligns with modern green chemistry principles, making the manufacturing process more sustainable and acceptable to regulatory bodies and environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a factual basis for decision-making. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of the supply source.
Q: What are the key advantages of this Pd-catalyzed route over traditional methods?
A: This method avoids harsh oxidizers like DDQ or MnO2, utilizing mild conditions (70-90°C) and readily available raw materials, which significantly simplifies post-treatment and improves overall yield consistency.
Q: How does the choice of ligand impact the reaction efficiency?
A: Experimental data indicates that Ligand L1 provides superior yields compared to analogues L2-L6. The specific electronic and steric properties of L1 facilitate the oxidative addition and reductive elimination steps in the palladium cycle more effectively.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the use of common solvents like toluene and dichloromethane, combined with standard purification techniques like flash column chromatography, makes this route highly adaptable for commercial scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazoline Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing the technical expertise to translate complex patent methodologies like CN108912059B into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of quinazoline derivatives meets the highest industry standards. Our commitment to quality and consistency makes us a trusted partner for pharmaceutical companies seeking to secure their supply of critical anti-inflammatory intermediates.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain insights into the specific economic benefits of adopting this method for your production needs. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements. Let us help you reduce lead time for high-purity quinazoline derivatives and achieve your commercial goals with confidence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
