Advanced Three-Step Synthesis of Novel Benzenesulfonamido Quinazoline Intermediates for Scalable Pharmaceutical Manufacturing
Advanced Three-Step Synthesis of Novel Benzenesulfonamido Quinazoline Intermediates for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks novel heterocyclic scaffolds that offer potent biological activity alongside manufacturability. Patent CN108727282B introduces a groundbreaking class of benzenesulfonamido-containing quinazoline compounds, specifically designated as Formula (7), which demonstrate significant inhibitory effects on inflammatory markers such as IL-6. This chemical architecture merges the pharmacological versatility of the quinazoline core with the metabolic stability often conferred by sulfonamide moieties. The structural novelty lies in the specific substitution patterns allowed at R1, R2, and R3 positions, enabling fine-tuning of physicochemical properties for drug development. As a reliable pharmaceutical intermediate supplier, understanding the precise molecular geometry is crucial for downstream formulation. The following structure represents the core scaffold that has shown promising anti-inflammatory potential in preclinical models.
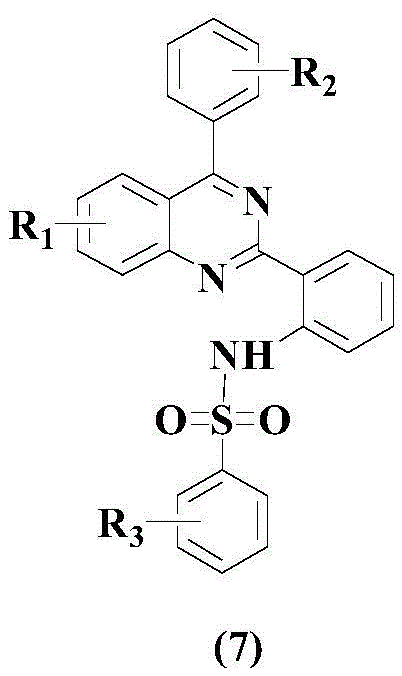
Historically, the construction of quinazoline rings has relied heavily on harsh cyclization conditions or stoichiometric amounts of toxic oxidants. Conventional methodologies often employ transition metals like copper or palladium in conjunction with strong oxidizing agents such as DDQ, MnO2, or NaClO to drive the aromatization process. These traditional approaches frequently suffer from poor atom economy, generate substantial hazardous waste, and often result in complex impurity profiles that are difficult to purge during purification. Furthermore, the use of strong oxidizers can lead to over-oxidation of sensitive functional groups, limiting the scope of substrates that can be tolerated. For procurement managers, these inefficiencies translate directly into higher raw material costs and increased environmental compliance burdens. The industry demand has shifted towards catalytic systems that operate under milder conditions while maintaining high selectivity and yield.
In stark contrast, the novel approach detailed in the patent utilizes a streamlined three-step sequence that circumvents the need for aggressive oxidative cyclization. The strategy employs a stepwise assembly beginning with a nucleophilic substitution, followed by a palladium-catalyzed cross-coupling, and concluding with a mild sulfonylation. This modular design allows for the introduction of diverse aryl groups at the C2 and C4 positions of the quinazoline ring with exceptional precision. By decoupling the ring formation from the functionalization steps, chemists gain greater control over the regioselectivity of the reaction. The elimination of stoichiometric oxidants not only enhances the safety profile of the manufacturing process but also simplifies the workup procedures. This methodological shift represents a significant advancement in cost reduction in anti-inflammatory compound manufacturing, offering a cleaner and more efficient pathway to high-value intermediates.
Mechanistic Insights into Palladium-Catalyzed Cross-Coupling and Sulfonylation
The heart of this synthetic innovation lies in the second step, where a palladium-catalyzed cross-coupling reaction constructs the biaryl linkage essential for biological activity. The mechanism involves the oxidative addition of the palladium catalyst to the aryl halide bond, followed by transmetallation with the boronic acid species. Crucially, the selection of the organic ligand plays a pivotal role in stabilizing the active palladium species and facilitating the reductive elimination step. Screening data indicates that Ligand L1, a bidentate nitrogen-based ligand, outperforms other candidates by providing the optimal electronic and steric environment for the catalytic cycle. The presence of an acidic additive, specifically p-toluenesulfonic acid monohydrate, further accelerates the reaction kinetics by activating the boronic acid and preventing the formation of inactive palladium black. This delicate balance of catalyst, ligand, and additive ensures high conversion rates even at moderate temperatures ranging from 70°C to 90°C.
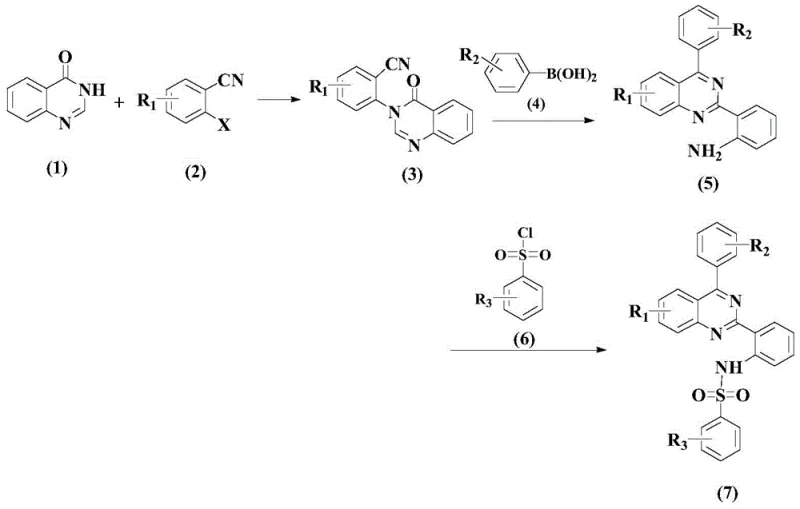
Impurity control is rigorously managed through the choice of solvents and reaction parameters in each stage. In the initial substitution step, the use of dimethyl sulfoxide (DMSO) as a polar aprotic solvent facilitates the dissolution of inorganic bases like potassium carbonate, ensuring homogeneous reaction conditions that minimize side reactions. The subsequent coupling step utilizes toluene, which allows for easy removal of water formed during the reaction via azeotropic distillation, thereby driving the equilibrium towards the desired product. Finally, the sulfonylation is conducted in pyridine, which acts as both solvent and base, scavenging the hydrochloric acid byproduct and preventing acid-catalyzed degradation of the sensitive quinazoline core. This strategic solvent selection across the three steps ensures that the final crude product possesses a high purity profile, reducing the burden on chromatographic purification. Such attention to mechanistic detail is vital for R&D directors aiming to replicate this chemistry for high-purity quinazoline derivatives.
How to Synthesize Benzenesulfonamido Quinazoline Efficiently
The operational protocol for this synthesis is designed to be robust and reproducible, making it ideal for technology transfer from laboratory to pilot plant. The process begins with the activation of the quinazolinone nitrogen, followed by the precise installation of the aryl group using the optimized palladium system described previously. The final sulfonylation step is particularly noteworthy for its mild conditions, proceeding effectively at temperatures as low as 20°C to 50°C, which preserves the integrity of thermally labile substituents. Detailed standardized operating procedures regarding reagent addition rates, temperature ramping, and quenching protocols are essential to maintain batch-to-batch consistency. For process chemists, adhering to the specified molar ratios, particularly the excess of boronic acid and the precise loading of the palladium catalyst, is critical to maximizing yield. The detailed standardized synthesis steps see the guide below for specific operational parameters.
- Perform nucleophilic substitution of quinazolinone with cyanobenzene derivatives in DMSO using potassium carbonate at 80-120°C to form the nitrile intermediate.
- Execute palladium-catalyzed cross-coupling with boronic acids using Pd(OAc)2 and Ligand L1 in toluene with acidic additives at 70-90°C.
- Complete the synthesis by reacting the amino-quinazoline intermediate with benzenesulfonyl chloride in pyridine at mild temperatures to yield the final sulfonamide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that address key pain points in the global supply chain for complex heterocyclic intermediates. The reliance on commercially available starting materials, such as substituted cyanobenzenes and boronic acids, mitigates the risk of raw material shortages that often plague specialized pharmaceutical projects. Furthermore, the avoidance of exotic reagents or cryogenic conditions means that the process can be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical facilities. This compatibility with existing infrastructure significantly reduces the capital expenditure required for technology adoption. For supply chain heads, the robustness of the chemistry translates to reducing lead time for high-purity benzenesulfonamides, ensuring that clinical trial materials and commercial stocks can be delivered reliably without unexpected delays caused by process failures.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric oxidants and the use of catalytic amounts of palladium significantly lower the direct material costs associated with production. Additionally, the high yields achieved in each step, particularly the near-quantitative conversion in the sulfonylation step, minimize the loss of valuable intermediates. The simplified purification requirements, driven by the clean reaction profiles, reduce the consumption of silica gel and organic solvents during chromatography. These factors combine to create a substantially more economical process compared to traditional oxidative cyclization methods, delivering significant cost savings without compromising on quality standards.
- Enhanced Supply Chain Reliability: The synthetic pathway utilizes commodity chemicals that are sourced from a broad global supplier base, reducing dependency on single-source vendors for critical reagents. The moderate reaction temperatures and pressures eliminate the need for specialized high-pressure equipment or extensive cooling systems, which enhances operational uptime. This operational simplicity ensures that manufacturing schedules can be maintained consistently, even during periods of high demand. Consequently, partners can rely on a stable supply of commercial scale-up of complex heterocyclic intermediates, safeguarding their downstream drug development timelines against raw material volatility.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of aqueous salt solutions and recoverable organic solvents, aligning with modern green chemistry principles. The absence of heavy metal oxidants simplifies wastewater treatment protocols and reduces the environmental footprint of the manufacturing site. Scalability is further supported by the exothermic nature of the reactions being manageable under standard cooling conditions, allowing for safe operation at multi-kilogram scales. This environmental and operational efficiency makes the route highly attractive for long-term commercial production, ensuring compliance with increasingly stringent global regulatory standards regarding chemical manufacturing emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on critical process parameters. Understanding these nuances is essential for teams evaluating the feasibility of adopting this technology for their specific pipeline candidates. The answers reflect the robustness of the method across various substrate scopes and reaction conditions.
Q: What are the critical reaction conditions for the palladium-catalyzed step?
A: The process requires precise control using Pd(OAc)2 as the catalyst, Ligand L1 for stability, and p-toluenesulfonic acid monohydrate as an additive in toluene solvent to ensure high yields and minimize side reactions.
Q: How does this synthesis route improve impurity profiles compared to conventional methods?
A: By avoiding strong oxidizers like DDQ or MnO2 and utilizing mild sulfonylation conditions, the route significantly reduces oxidative by-products and simplifies downstream purification processes.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of readily available raw materials, standard organic solvents like toluene and DMSO, and moderate temperature ranges makes the pathway highly adaptable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzenesulfonamido Quinazoline Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate this innovative patent chemistry into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from preclinical studies to market launch. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee the quality of every batch. Our commitment to excellence means that we do not just supply chemicals; we provide validated processes that meet the demanding requirements of the global pharmaceutical industry. Partnering with us ensures access to a supply chain that is both resilient and responsive to your evolving needs.
We invite you to engage with our technical procurement team to discuss how this synthesis route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits tailored to your volume needs. We encourage potential partners to contact us for specific COA data and route feasibility assessments to verify the compatibility of this chemistry with your development goals. Let us collaborate to accelerate the delivery of next-generation anti-inflammatory therapies to patients worldwide through superior chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
