Optimized Masitinib Synthesis: Technical Breakthroughs for Commercial Scale-Up and Cost Efficiency
The pharmaceutical industry continuously seeks efficient pathways for producing tyrosine kinase inhibitors, particularly for oncology applications. Patent CN115850258A introduces a transformative synthesis method for masitinib, addressing critical bottlenecks in traditional manufacturing. This technical insight report analyzes the novel route, which utilizes 3-acetylpyridine as a starting substrate to construct the thiazole core, diverging from conventional methods that rely on costly brominated precursors. By optimizing reaction conditions and reagent selection, this approach offers a compelling value proposition for reliable pharmaceutical intermediate supplier networks aiming to enhance supply chain resilience. The strategic shift in synthetic design not only simplifies operational complexity but also aligns with modern green chemistry principles, ensuring that cost reduction in API manufacturing is achieved without compromising molecular integrity or therapeutic efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of masitinib has been plagued by excessive step counts and reliance on hazardous or expensive reagents, creating significant barriers for commercial scale-up of complex kinase inhibitors. Traditional Scheme 1 involves protecting group strategies with Boc anhydride and subsequent deprotection using trifluoroacetic acid, which generates substantial chemical waste and increases processing time. Furthermore, the reliance on 3-(2-bromoacetyl)pyridine introduces volatility in raw material pricing and supply continuity, as brominated building blocks often face regulatory scrutiny and logistical challenges. The use of strong acids and bases in multiple steps also necessitates specialized corrosion-resistant equipment, driving up capital expenditure for production facilities. These cumulative inefficiencies result in prolonged lead times and elevated production costs, ultimately impacting the affordability of the final therapeutic agent for patients worldwide.
The Novel Approach
In stark contrast, the novel methodology described in the patent streamlines the synthetic trajectory by initiating the sequence with readily available 3-acetylpyridine. This strategic substitution eliminates the need for pre-functionalized brominated pyridines, thereby stabilizing the supply chain and reducing raw material procurement risks. The direct cyclization with thiourea under iodine catalysis proceeds under mild conditions, avoiding the harsh environments typical of legacy processes. By bypassing protection and deprotection cycles, the overall atom economy is significantly improved, and the number of isolation steps is reduced. This refined approach not only accelerates the time-to-market for high-purity pharmaceutical intermediates but also establishes a more robust foundation for consistent quality control across large-scale batches, ensuring that commercial partners receive a product that meets rigorous international standards.
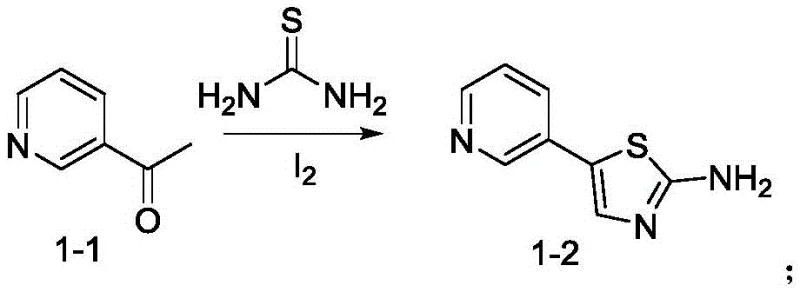
Mechanistic Insights into Iodine-Catalyzed Thiazole Formation and Cu-Mediated Coupling
The cornerstone of this innovative synthesis lies in the efficient construction of the 5-(pyridin-3-yl)thiazol-2-amine scaffold through an iodine-catalyzed cyclization. In this mechanism, 3-acetylpyridine reacts with thiourea in tetrahydrofuran, where iodine acts as a mild oxidant to facilitate the formation of the carbon-sulfur bond and subsequent ring closure. The molar ratio of iodine to substrate is carefully optimized at 0.75:1, ensuring complete conversion while minimizing halogen waste. Following this, the copper-catalyzed coupling step utilizes cuprous iodide and N,N-dimethylethylenediamine as a ligand system to link the thiazole amine with 2-bromo-4-nitrotoluene. This Ullmann-type coupling is critical for establishing the C-N bond between the heterocyclic core and the aromatic ring, proceeding efficiently at 110-115°C in a toluene-pyridine mixture. The precise control of these catalytic cycles ensures high regioselectivity and minimizes the formation of side products, which is essential for maintaining the purity profile required for downstream pharmaceutical applications.
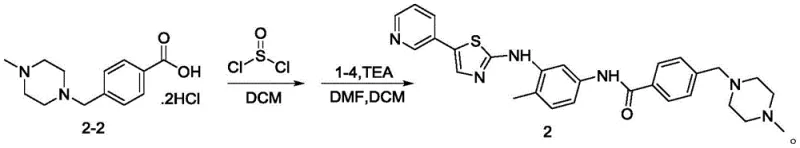
Impurity control is rigorously managed throughout the synthetic sequence, particularly during the reduction and amidation stages. The reduction of the nitro group to the amine is executed using zinc powder in ethanol with acetic acid, a method chosen for its selectivity and ease of workup compared to catalytic hydrogenation which might reduce other sensitive functionalities. The subsequent amidation step avoids the use of trimethylaluminum, a reagent known for its instability and safety hazards, by employing thionyl chloride to activate the carboxylic acid in situ. This modification prevents the formation of aluminum-containing impurities that are notoriously difficult to remove to trace levels. By selecting reagents that generate soluble or easily filterable byproducts, the process ensures that the final active pharmaceutical ingredient meets stringent residual solvent and heavy metal specifications, thereby safeguarding patient safety and regulatory compliance.
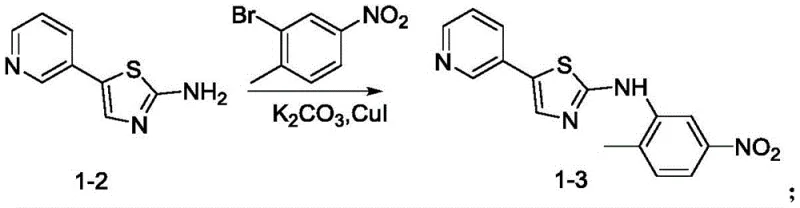
How to Synthesize Masitinib Efficiently
The implementation of this synthesis route requires precise adherence to the optimized reaction parameters detailed in the patent examples to ensure reproducibility and high yield. The process begins with the cyclization step, followed by the coupling, reduction, side-chain preparation, and final condensation, each requiring specific temperature controls and stoichiometric ratios. Operators must monitor reaction progress via TLC or HPLC to determine exact endpoints, as over-reaction can lead to degradation of the sensitive thiazole moiety. The detailed standardized synthesis steps see the guide below provide a comprehensive framework for laboratory and pilot-scale execution, ensuring that technical teams can replicate the success of the patent examples with confidence.
- Cyclization of 3-acetylpyridine with thiourea using iodine catalyst to form 5-(pyridin-3-yl)thiazol-2-amine.
- CuI-catalyzed coupling of the thiazole amine with 2-bromo-4-nitrotoluene to build the core scaffold.
- Reduction of the nitro group using zinc powder and acetic acid to generate the diamine intermediate.
- Preparation of the side chain acid via reaction of 4-chloromethylbenzoic acid with 4-methylpiperazine.
- Final amidation using thionyl chloride activation under mild alkaline conditions to yield masitinib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis protocol offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of expensive and unstable reagents directly translates to a more predictable cost structure, allowing for better long-term budget planning and contract negotiation. By utilizing commodity chemicals like 3-acetylpyridine and zinc powder, the dependency on specialized suppliers is reduced, mitigating the risk of supply disruptions caused by geopolitical or manufacturing issues. Furthermore, the simplified operational workflow reduces the burden on quality assurance teams, as fewer steps mean fewer opportunities for deviation and batch failure. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The substitution of high-cost 3-(2-bromoacetyl)pyridine with inexpensive 3-acetylpyridine fundamentally alters the economic model of production. Additionally, avoiding the use of trimethylaluminum removes the need for specialized storage and handling infrastructure, further decreasing overhead expenses. The reduction in step count also lowers labor and utility consumption per kilogram of product, contributing to substantial cost savings. These efficiencies allow manufacturers to offer more competitive pricing without sacrificing margin, creating a win-win scenario for both suppliers and buyers in the generic drug market.
- Enhanced Supply Chain Reliability: Sourcing raw materials that are widely available in the bulk chemical market ensures a stable supply chain不受 single-source bottlenecks. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in environmental parameters, leading to higher batch success rates. This consistency is vital for securing long-term supply agreements with major pharmaceutical companies who prioritize vendor reliability. By minimizing the use of hazardous materials, transportation and logistics become simpler and less regulated, speeding up the movement of goods from factory to formulation site.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of extreme pressure requirements make this process inherently safer and easier to scale from pilot plants to multi-ton reactors. The waste profile is significantly improved due to the avoidance of heavy metal catalysts and toxic reagents, facilitating easier wastewater treatment and disposal. This alignment with environmental, social, and governance (ESG) goals enhances the corporate reputation of manufacturers and ensures compliance with increasingly strict international environmental regulations. Consequently, the process supports sustainable growth and long-term viability in the evolving landscape of green pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived from the specific advantages outlined in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the practical realities of scaling this chemistry while maintaining the high standards expected in the pharmaceutical industry.
Q: How does this synthesis method reduce costs compared to conventional routes?
A: This method replaces expensive 3-(2-bromoacetyl)pyridine with low-cost 3-acetylpyridine and avoids unstable trimethylaluminum, significantly lowering raw material and handling costs.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild, operation is simple, and it avoids hazardous reagents, making it highly scalable for industrial manufacturing of kinase inhibitor intermediates.
Q: What are the purity specifications for the intermediates?
A: The process utilizes robust purification steps including recrystallization and pH adjustment, ensuring high-purity intermediates suitable for stringent pharmaceutical quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Masitinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global API market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity masitinib intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to technical excellence allows us to navigate the complexities of heterocyclic chemistry with precision, guaranteeing a supply of materials that meet the exacting demands of drug development and commercial manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this novel route. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume needs. Together, we can drive efficiency and innovation in the production of life-saving oncology therapies, ensuring a reliable supply of high-quality pharmaceutical intermediates for the future.
