Scalable Production of Insecticidal Nitroguanidine Derivatives via Salt-Mediated Aqueous Chemistry
Scalable Production of Insecticidal Nitroguanidine Derivatives via Salt-Mediated Aqueous Chemistry
The global demand for high-efficiency neonicotinoid insecticides continues to drive innovation in the synthesis of their key intermediates, specifically nitroguanidine derivatives. Patent CN101379035B introduces a transformative methodology that addresses long-standing stability and purity challenges in this chemical class. By leveraging a unique aqueous system saturated with inorganic salts, this technology enables the direct conversion of inexpensive nitroisourea precursors into high-value agrochemical intermediates with exceptional selectivity. For R&D directors and procurement strategists, this represents a significant opportunity to optimize manufacturing costs while adhering to stricter environmental regulations. The process eliminates the need for volatile organic solvents and avoids the generation of hazardous sulfur-containing waste streams associated with legacy synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of guanidine derivatives with insecticidal activity has relied heavily on exchange reactions between isothiourea derivatives and amines, as disclosed in earlier patents such as JP-A-7-179448. This conventional pathway suffers from critical drawbacks that impact both operational safety and economic efficiency. The primary issue is the liberation of thiol compounds as by-products, which possess extremely offensive odors and require specialized scrubbing systems to manage, thereby increasing capital expenditure. Furthermore, alternative routes utilizing isourea compounds (General Formula A) as intermediates, while avoiding sulfur, necessitate the use of expensive starting materials that drive up the overall cost of goods sold. These traditional methods often struggle with low stability of intermediates and the formation of difficult-to-remove impurities, complicating the purification process and reducing overall plant throughput.
The Novel Approach
The methodology described in CN101379035B fundamentally shifts the paradigm by utilizing a nitroisourea derivative (General Formula 1) and an amine derivative (General Formula 2) in a specialized aqueous environment. 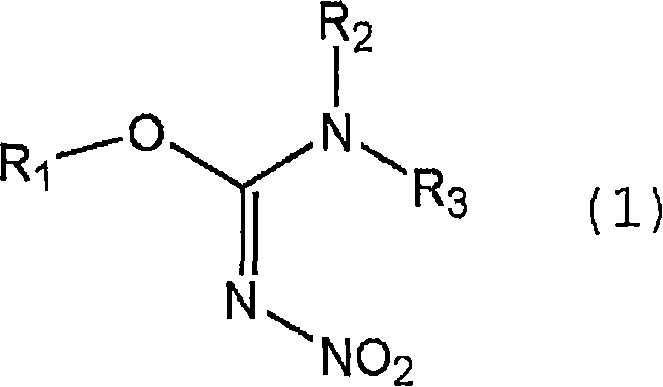
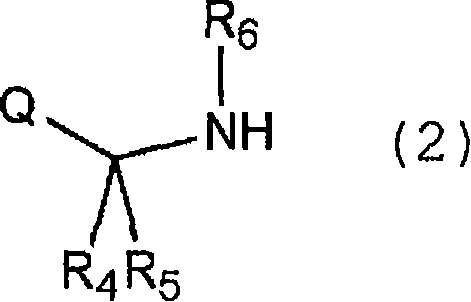
 This novel approach operates in water containing an inorganic salt at a concentration of 50% or more of its saturation solubility. This specific condition is not merely a solvent choice but a critical process parameter that stabilizes the reactive nitroisourea species against hydrolytic decomposition. By conducting the reaction in the presence of a base within this high-ionic-strength medium, the process achieves high selectivity, effectively suppressing the formation of unwanted bis-guanidine by-products (General Formula 5) that typically plague aqueous syntheses. This results in a cleaner reaction profile and significantly reduces the burden on downstream purification units.
This novel approach operates in water containing an inorganic salt at a concentration of 50% or more of its saturation solubility. This specific condition is not merely a solvent choice but a critical process parameter that stabilizes the reactive nitroisourea species against hydrolytic decomposition. By conducting the reaction in the presence of a base within this high-ionic-strength medium, the process achieves high selectivity, effectively suppressing the formation of unwanted bis-guanidine by-products (General Formula 5) that typically plague aqueous syntheses. This results in a cleaner reaction profile and significantly reduces the burden on downstream purification units.
Mechanistic Insights into Salt-Mediated Stabilization and Nucleophilic Substitution
The core chemical innovation lies in the manipulation of solubility and reaction kinetics through the salting-out effect. In standard aqueous conditions, nitroisourea derivatives are prone to rapid decomposition, leading to poor yields. However, by saturating the water with inorganic salts such as sodium chloride or potassium carbonate, the solubility of the organic nitroisourea reactant is drastically reduced, creating a solid-liquid two-phase system. This heterogeneity protects the sensitive nitroisourea from bulk water attack while allowing the reaction to proceed at the interface or within the micro-environment facilitated by the base. The inorganic salt also functions as an internal refrigerant; the endothermic dissolution or the high heat capacity of the brine allows the reaction to be maintained at low temperatures, typically between -10°C and 10°C, which is crucial for kinetic control. This precise thermal management prevents exothermic runaways and minimizes side reactions, ensuring that the nucleophilic attack by the amine occurs selectively at the desired carbon center.
Impurity control is another mechanistic advantage of this system. In the absence of the specific salt concentration and base optimization, the reaction tends to produce symmetrical nitroguanidine by-products (General Formula 4 and 5), which are structurally similar to the target molecule and notoriously difficult to separate via crystallization. The patented method suppresses the formation of these impurities to levels below 1 mol%, as demonstrated in comparative examples where standard water usage led to 5 mol% impurity generation. The presence of the base neutralizes acidic by-products and facilitates the deprotonation of the amine nucleophile, enhancing its reactivity without promoting the degradation of the nitroisourea electrophile. This delicate balance allows for the production of high-purity crystals directly from the reaction mixture, often eliminating the need for chromatographic purification steps that are impractical at a multi-ton scale.
How to Synthesize Nitroguanidine Derivatives Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing compounds like 1-methyl-2-nitro-3-[(3-tetrahydrofuryl)methyl]guanidine. The process begins with the preparation of a brine solution, where an inorganic salt is dissolved in water to reach at least 50% of its saturation point at the target reaction temperature. An amine derivative is then introduced to this medium, followed by the addition of a base such as sodium hydroxide or potassium carbonate. The nitroisourea starting material is added portion-wise or as a slurry to maintain the heterogeneous nature of the system. The reaction mixture is stirred at low temperatures, typically starting around -10°C and gradually warming to 10°C or 20°C over several hours to ensure complete conversion. Upon completion, the pH is adjusted to acidic conditions to precipitate the product or facilitate extraction. The detailed standardized synthesis steps are provided below.
- Prepare an aqueous reaction medium containing an inorganic salt (e.g., NaCl) at 50% to 100% of its saturation solubility at the reaction temperature.
- Mix the amine derivative and base in the salt solution, then add the nitroisourea derivative while maintaining low temperatures (-10°C to 10°C).
- Stir the mixture to complete the reaction, adjust pH to acidic conditions, and isolate the product via crystallization or extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this salt-mediated aqueous synthesis offers tangible strategic benefits beyond simple yield improvements. The shift from organic solvents to water-based systems drastically reduces the cost of raw materials and eliminates the complex logistics associated with solvent recovery and disposal. Since water and common inorganic salts are commoditized chemicals with stable pricing and abundant global availability, the supply chain becomes far more resilient to market fluctuations compared to processes reliant on specialized organic reagents. Furthermore, the elimination of malodorous thiol by-products removes the need for expensive odor-control infrastructure and hazardous waste treatment, directly lowering the operational expenditure (OPEX) of the manufacturing facility. This process intensification allows for higher batch turnover rates and more efficient use of reactor volume.
- Cost Reduction in Manufacturing: The utilization of inexpensive nitroisourea intermediates instead of costly isourea or isothiourea precursors significantly lowers the direct material cost. Additionally, the ability to crystallize high-purity product directly from the reaction mixture reduces the number of unit operations required, such as distillation or column chromatography. This simplification of the downstream processing train leads to substantial savings in energy consumption and labor costs. The avoidance of sulfur-based waste also mitigates environmental compliance costs, which are increasingly becoming a major financial burden for chemical manufacturers operating under strict regulatory frameworks.
- Enhanced Supply Chain Reliability: By relying on water and common salts like sodium chloride as the primary reaction medium, the process decouples production from the volatility of the organic solvent market. This ensures consistent production scheduling and reduces the risk of shutdowns due to solvent shortages. The robustness of the reaction conditions, which tolerate a range of temperatures and mixing rates without significant yield loss, makes the process highly transferable between different manufacturing sites. This flexibility is crucial for multinational corporations seeking to diversify their supplier base and mitigate geopolitical risks associated with single-source dependencies for critical agrochemical intermediates.
- Scalability and Environmental Compliance: The aqueous nature of the reaction makes it inherently safer and easier to scale from pilot plant to commercial production. Water has a high heat capacity, which aids in temperature control during exothermic reactions, reducing the risk of thermal runaway incidents. From an environmental perspective, the process aligns with Green Chemistry principles by minimizing the use of volatile organic compounds (VOCs) and preventing the release of toxic sulfur emissions. This eco-friendly profile facilitates easier permitting for new production lines and enhances the corporate sustainability metrics of the final agrochemical product, which is increasingly valued by end-users in the agricultural sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitroguanidine synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a reliable basis for feasibility assessments. Understanding these nuances is essential for engineering teams evaluating the retrofitting of existing facilities or the design of new production lines. The data confirms that the method is not only theoretically sound but has been validated through multiple comparative examples demonstrating superior performance over prior art.
Q: Why is inorganic salt added to the reaction water?
A: The inorganic salt serves a dual purpose: it lowers the solubility of the unstable nitroisourea intermediate to prevent hydrolysis and acts as a refrigerant to maintain low reaction temperatures, significantly improving yield and selectivity.
Q: What are the advantages over traditional isothiourea exchange methods?
A: Traditional methods generate malodorous thiol by-products and require expensive isourea intermediates. This novel aqueous method uses cheaper nitroisourea starting materials, eliminates sulfur waste, and simplifies purification.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the process utilizes water as the primary solvent and common inorganic salts, making it environmentally friendly, safe, and highly scalable for multi-ton production without complex organic solvent recovery systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitroguanidine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes for the production of high-performance agrochemical intermediates. Our technical team has extensively analyzed the salt-mediated aqueous synthesis described in CN101379035B and possesses the expertise to implement this technology at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to full-scale manufacturing is seamless and efficient. Our facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of nitroguanidine derivative meets the exacting standards required for modern insecticide formulations.
We invite potential partners to engage with our technical procurement team to discuss how this innovative process can be tailored to your specific supply chain needs. By leveraging our manufacturing capabilities, you can secure a stable supply of high-purity intermediates while achieving significant cost efficiencies. Please contact us to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments for your target molecules. We are committed to being your strategic partner in delivering sustainable and economically viable chemical solutions for the global agrochemical market.
