Advanced Manufacturing of 2-Naphthylacetonitrile via Modified Willgerodt Rearrangement
The pharmaceutical and agrochemical industries constantly demand safer, more efficient pathways for synthesizing complex aromatic building blocks. Patent CN112041298A introduces a groundbreaking production method for aromatic nitrile compounds, specifically focusing on 2-naphthylacetonitrile, a critical intermediate for CNS drugs and other high-value therapeutics. This technology addresses long-standing challenges in industrial organic synthesis by replacing hazardous cyanation protocols with a robust Willgerodt-Kindler rearrangement followed by a novel dehydrative cyclization. By leveraging readily available ketone precursors like 2'-acetonaphthone, this process eliminates the need for toxic sodium cyanide and minimizes the formation of difficult-to-remove halogenated by-products. The result is a manufacturing route that offers superior control over impurity profiles, particularly sulfur content, while maintaining high atom economy and operational safety standards suitable for multi-ton scale production.
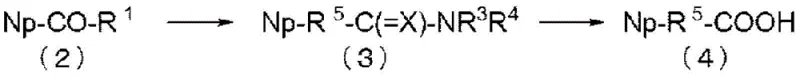
Historically, the synthesis of 2-naphthylacetonitrile relied heavily on the bromination of 2-methylnaphthalene followed by nucleophilic substitution with cyanide salts. This conventional approach, while chemically straightforward, presents severe limitations for modern GMP manufacturing. The bromination step is notoriously exothermic and prone to generating dibrominated impurities, which are structurally similar to the target and difficult to separate without extensive chromatography. Furthermore, the subsequent use of alkali metal cyanides introduces acute toxicity risks that require specialized containment infrastructure and rigorous waste treatment protocols. In contrast, the novel approach detailed in the patent utilizes a Willgerodt-Kindler reaction to convert the ketone into a thioamide, which is subsequently hydrolyzed to the carboxylic acid. This pathway avoids free radical halogenation entirely, thereby eliminating the risk of poly-halogenated side products and providing a much cleaner reaction matrix for the final transformation into the nitrile functionality.
The mechanistic elegance of this process lies in its two-stage design, which decouples the carbon skeleton construction from the functional group installation. The initial Willgerodt-Kindler rearrangement employs elemental sulfur and a secondary amine, typically morpholine, to migrate the carbonyl group and introduce the necessary nitrogen atom. A critical innovation in this patent is the management of sulfur residues; since sulfur is used in stoichiometric excess, residual sulfur species can poison downstream catalysts or contaminate the final API. The patent discloses a specific workup procedure involving contact with hydrocarbon solvents like toluene during the neutralization phase. This extraction step selectively partitions sulfur impurities away from the desired 2-naphthaleneacetic acid, achieving sulfur levels as low as 0.001 mol%. Following this purification, the carboxylic acid is converted to the nitrile via a direct dehydrative protocol using thionyl chloride and a sulfonamide reagent. This one-pot transformation bypasses the isolation of the primary amide, reducing unit operations and solvent consumption while driving the equilibrium toward the nitrile product through the formation of stable gaseous by-products.

For research and development teams aiming to implement this chemistry, the protocol offers a clear, scalable trajectory from commodity chemicals to high-value intermediates. The process begins with the condensation of 2'-acetonaphthone with sulfur and morpholine under heated conditions, typically between 115°C and 125°C, to generate the thioamide intermediate. Following hydrolysis with aqueous base and the critical toluene extraction to remove sulfur, the resulting 2-naphthaleneacetic acid is subjected to the final cyanation step. This sequence ensures that each stage is optimized for yield and purity, allowing for robust process control parameters that can be easily transferred from pilot plant to commercial manufacturing suites without significant re-engineering of the reaction infrastructure.
From a procurement and supply chain perspective, this manufacturing method offers substantial strategic advantages over legacy technologies. First, the elimination of sodium cyanide removes a Schedule 1 chemical from the supply chain, drastically simplifying regulatory compliance and reducing the costs associated with hazardous material handling and disposal. Second, the starting material, 2'-acetonaphthone, is a widely available commodity chemical with a stable global supply, reducing the risk of raw material shortages that often plague specialized halogenated precursors. Third, the solvent systems employed, primarily toluene and sulfolane, are common industrial solvents with well-established recovery and recycling streams, contributing to a lower overall environmental footprint. By streamlining the purification process through selective solvent extraction rather than energy-intensive distillation or crystallization cycles, the method significantly lowers utility consumption. These factors combine to create a cost structure that is not only competitive but also resilient against fluctuating regulatory pressures regarding toxic reagents.
Supply chain reliability is further enhanced by the robustness of the reaction conditions. The Willgerodt-Kindler step operates at atmospheric pressure and moderate temperatures, reducing the mechanical stress on reactor vessels and minimizing the risk of thermal runaway incidents. The final nitrile formation step is similarly controllable, with the patent specifying temperature ranges of 80°C to 180°C that allow for flexibility depending on the specific cooling capacity of the manufacturing facility. This thermal flexibility ensures that the process can be scaled from 100 kg batches to 100 MT annual production volumes without encountering heat transfer limitations that often bottleneck exothermic halogenation reactions. Additionally, the high purity of the intermediate carboxylic acid (>99 mol%) ensures that downstream reactions, such as the synthesis of complex bicyclic CNS drug candidates, proceed with high fidelity, reducing the burden on final API purification steps and improving overall campaign throughput.
Frequently asked questions regarding this technology often center on the specific impurity profiles and the scalability of the sulfur removal step. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this route for their specific pipeline candidates, as the ability to consistently meet strict sulfur specifications is a key differentiator for pharmaceutical grade intermediates.
- Perform a Willgerodt-Kindler reaction on 2'-acetonaphthone using sulfur and morpholine at 115-125°C to form a thioamide intermediate.
- Hydrolyze the thioamide with aqueous sodium hydroxide, then neutralize and extract with a hydrocarbon solvent like toluene to reduce sulfur content below 1 mol%.
- React the purified 2-naphthaleneacetic acid with thionyl chloride and sulfonamide in sulfolane at 95-105°C to directly yield 2-naphthylacetonitrile.
Frequently Asked Questions (FAQ)
Q: How does this process manage sulfur impurities from the Willgerodt reaction?
A: The patent describes a critical purification step where the hydrolyzed reaction product is contacted with a hydrocarbon solvent, such as toluene, during or after neutralization. This extraction effectively reduces sulfur content to between 0.001 mol% and 1 mol%, ensuring high purity for downstream pharmaceutical applications.
Q: What are the safety advantages over traditional bromination-cyanation routes?
A: Traditional methods often utilize highly toxic sodium cyanide and exothermic bromination steps which pose significant industrial hazards. This patented method avoids free cyanide salts entirely by using sulfonamide and thionyl chloride for the final nitrile formation, significantly improving the safety profile for large-scale manufacturing.
Q: What purity levels can be achieved with this manufacturing method?
A: Experimental data within the patent indicates that the final 2-naphthylacetonitrile product can achieve an HPLC purity of 98 area% or higher, with optimized conditions reaching 99.5 area%. The intermediate carboxylic acid also achieves purities exceeding 99 mol% after solvent extraction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Naphthylacetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities down to ppm levels, guaranteeing that every batch of 2-naphthylacetonitrile meets the highest industry standards. We understand the critical nature of aromatic nitrile intermediates in the synthesis of life-saving medications and are committed to delivering materials that facilitate your success.
We invite you to collaborate with our technical procurement team to explore how this advanced manufacturing process can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain insights into how switching to this safer, more efficient route can optimize your overall production budget. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your timeline, ensuring a seamless integration of high-quality intermediates into your supply chain.
