Advanced Manufacturing of 3-Chloro-2-Vinylphenyl Sulfonates for High-Performance Fungicides
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to synthesize critical intermediates that serve as the backbone for active ingredients. Patent CN107406374B discloses a groundbreaking methodology for the preparation of 3-chloro-2-vinylphenyl sulfonate derivatives, specifically targeting the synthesis of potent fungicidal agents. This innovation addresses long-standing inefficiencies in prior art, particularly the reliance on tetrachlorocyclohexanone, which suffers from poor atom economy and complex purification requirements. By shifting the synthetic strategy to a radical chlorination and elimination pathway starting from readily available phenols, this technology offers a robust alternative for reliable agrochemical intermediate supplier networks aiming to optimize their supply chains. The ability to produce high-purity precursors with improved yield profiles is essential for maintaining competitiveness in the global fungicide market.
The significance of this patent extends beyond mere chemical novelty; it represents a strategic shift towards more sustainable and cost-effective manufacturing practices. The disclosed process minimizes waste generation by retaining chlorine atoms more effectively throughout the synthesis and utilizes standard reagents that are easily sourced on a global scale. For procurement teams and R&D directors alike, understanding the nuances of this pathway is critical for evaluating potential partnerships and licensing opportunities. As we delve deeper into the technical specifics, it becomes clear that this method not only solves specific synthetic bottlenecks but also aligns with broader industry goals of reducing environmental impact while enhancing production reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-chloro-2-vinylphenol and its subsequent sulfonate derivatives has been plagued by significant technical hurdles that hinder commercial viability. The traditional route, as documented in earlier patents like EP 0511036 B1, relies on tetrachlorocyclohexanone as the primary starting material. This approach is fundamentally flawed due to its abysmal atom economy; the starting material contains four chlorine atoms, yet only one is retained in the final molecular structure, resulting in substantial chemical waste and increased disposal costs. Furthermore, the process requires the formation of a vinyl Grignard reagent and a subsequent ring-opening step in N,N-dimethylformamide (DMF) under reflux conditions, which often leads to lower overall yields and the formation of difficult-to-remove impurities. These factors combined make the conventional method economically unattractive for large-scale cost reduction in agrochemical manufacturing.
The Novel Approach
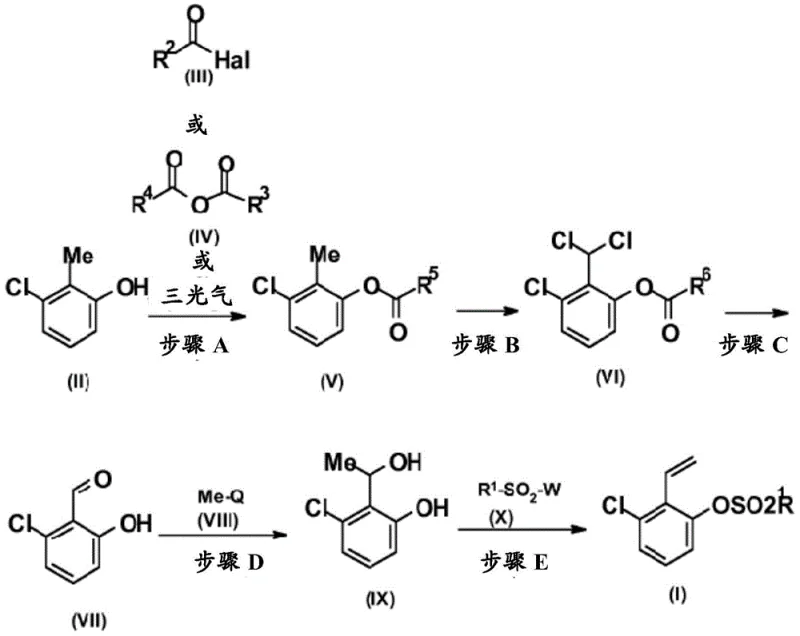
In stark contrast, the novel process outlined in CN107406374B leverages a completely different disconnection strategy that begins with 2-methyl-3-chlorophenol, a much more accessible and cost-efficient feedstock. This new pathway ingeniously constructs the necessary carbon framework through a sequence of esterification, radical chlorination, and hydrolysis, avoiding the wasteful degradation of the cyclohexane ring seen in older methods. By protecting the phenolic hydroxyl group early in the sequence, the process allows for selective functionalization of the methyl group without interference, ensuring higher regioselectivity and purity. This strategic redesign not only improves the theoretical yield but also simplifies the downstream processing, making it an ideal candidate for the commercial scale-up of complex agrochemical intermediates.
Mechanistic Insights into Radical Chlorination and Elimination
The core of this innovative synthesis lies in the precise execution of a radical chlorination step followed by a controlled elimination. In Step B of the process, the methyl group of the protected phenol intermediate is transformed into a dichloromethyl group using elemental chlorine under UV irradiation. This free-radical mechanism is highly efficient and can be driven to completion without significant over-chlorination, a common concern in benzylic chlorinations. The use of UV light as an initiator ensures a steady generation of chlorine radicals, facilitating the substitution of hydrogen atoms with chlorine at elevated temperatures ranging from 50°C to 150°C. This step is crucial as it installs the two-carbon functionality required for the eventual vinyl group, setting the stage for the subsequent hydrolysis to the aldehyde.
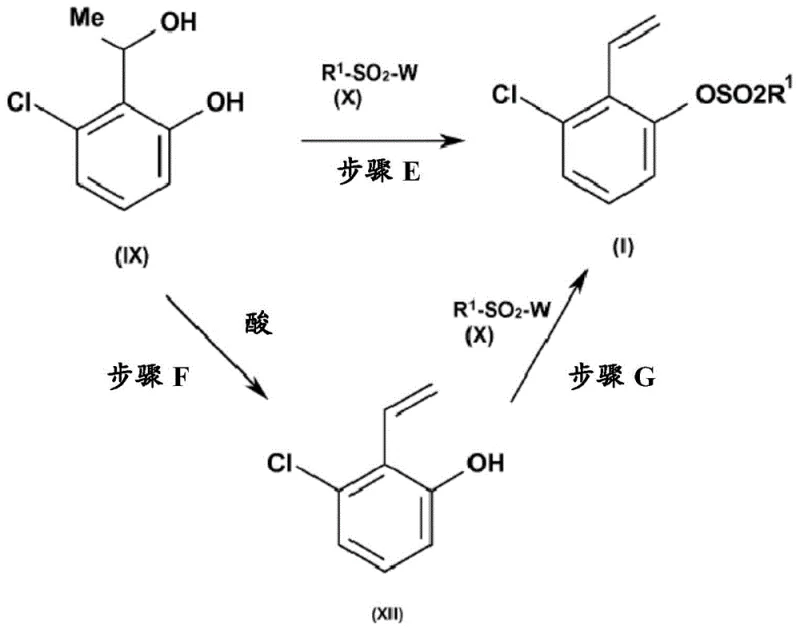
Following the formation of the aldehyde intermediate, the process employs a Grignard addition using methyl magnesium chloride or bromide to generate a secondary alcohol. The final transformation involves a sophisticated elimination strategy where the hydroxyl group is removed to form the vinyl double bond. Interestingly, the patent describes two viable pathways for this final stage: either a direct elimination to the phenol followed by sulfonylation, or a concerted process where the alcohol is activated by a sulfonyl chloride and eliminated in situ. This flexibility allows manufacturers to tailor the process based on their specific equipment capabilities and purity requirements, ensuring that the high-purity agrochemical intermediate specifications are met consistently.
How to Synthesize 3-Chloro-2-Vinylphenyl Sulfonate Efficiently
The synthesis of this valuable intermediate requires careful control of reaction parameters across multiple steps to ensure optimal yield and safety. The process begins with the protection of the phenol, followed by the critical radical chlorination which demands strict temperature and UV intensity management. Subsequent hydrolysis converts the dichloromethyl group into an aldehyde, which is then elaborated via Grignard chemistry. The final steps involve the delicate balance of elimination and sulfonylation to install the leaving group necessary for downstream coupling reactions. For a detailed breakdown of the specific reagents, stoichiometry, and workup procedures described in the patent, please refer to the standardized guide below.
- Esterify 2-methyl-3-chlorophenol with trichloroacetyl chloride or phosgene to form the protected ester intermediate.
- Perform radical chlorination using chlorine gas under UV irradiation to convert the methyl group into a dichloromethyl group.
- Hydrolyze the dichloromethyl ester under acidic or high-temperature aqueous conditions to yield 2-chloro-6-hydroxybenzaldehyde.
- React the aldehyde with methyl magnesium chloride (Grignard reagent) to form the secondary alcohol intermediate.
- Convert the alcohol to the final vinyl sulfonate via acid-catalyzed elimination followed by sulfonylation, or a one-pot variation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the pain points of modern chemical supply chains. By switching to a starting material that is derived from abundant chlorophenols, manufacturers can significantly reduce raw material volatility and cost. The elimination of the tetrachlorocyclohexanone route removes the need for specialized, waste-intensive chlorination processes at the very beginning of the synthesis, thereby streamlining the upstream supply chain. Furthermore, the use of standard reagents like chlorine gas, phosgene equivalents, and Grignard reagents ensures that sourcing remains stable and predictable, mitigating the risk of production delays caused by exotic reagent shortages.
- Cost Reduction in Manufacturing: The new route drastically improves atom economy by retaining chlorine atoms that would otherwise be discarded as waste in traditional methods. This efficiency translates directly into lower raw material consumption per kilogram of product. Additionally, the avoidance of harsh ring-opening conditions in polar aprotic solvents like DMF reduces solvent recovery costs and energy consumption. The simplified purification profile means less time and resources are spent on distillation and crystallization, leading to substantial operational expenditure savings without compromising quality.
- Enhanced Supply Chain Reliability: Because the synthesis relies on commodity chemicals and well-established unit operations such as radical chlorination and esterification, it is less susceptible to supply disruptions. The robustness of the radical chlorination step, which tolerates slight variations in conditions without forming excessive by-products, ensures consistent batch-to-bquality. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery expectations of major agrochemical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing reaction conditions that are easily managed in standard stainless steel reactors. The reduction in chemical waste, particularly the avoidance of losing three chlorine atoms per molecule as seen in the old route, aligns with stricter environmental regulations and sustainability goals. Easier waste treatment and lower effluent loads make this technology a preferred choice for facilities aiming to minimize their environmental footprint while expanding production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is vital for stakeholders evaluating the feasibility of adopting this new process for their manufacturing portfolios. The answers are derived directly from the experimental data and claims presented in the patent documentation.
Q: What are the key advantages of this new synthesis route over the traditional tetrachlorocyclohexanone method?
A: The new route utilizes 2-methyl-3-chlorophenol, a more accessible starting material, and avoids the poor atom economy of the tetrachlorocyclohexanone route where three out of four chlorine atoms are lost. It also eliminates the need for harsh ring-opening steps in DMF, leading to higher overall yields and simpler purification.
Q: How is the vinyl group introduced in this process?
A: The vinyl group is generated through a dehydration elimination of a 1-hydroxyethyl intermediate. This intermediate is first formed by adding a methyl Grignard reagent to an aldehyde, and subsequently eliminated under acidic conditions or during the sulfonylation step to form the double bond.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process relies on standard industrial unit operations such as radical chlorination with UV light, standard esterification, and Grignard reactions. The conditions (temperatures between 0°C and 160°C) are manageable, and the reagents are commercially available, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloro-2-Vinylphenyl Sulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in CN107406374B for the production of next-generation fungicides. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3-chloro-2-vinylphenyl sulfonate performs reliably in your downstream coupling reactions.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your agrochemical manufacturing operations.
