Revolutionizing Chiral 3-Halophthalic Acid Derivative Synthesis for Commercial Scale-Up
Introduction to Regioselective Salt Formation Technology
The chemical industry constantly seeks methods to overcome the inherent limitations of regioselectivity in aromatic substitution reactions, particularly when dealing with halogenated phthalic anhydrides. Patent CN101010293A introduces a groundbreaking approach for the preparation of chiral 3-halophthalic acid derivatives of formula (I), addressing the longstanding challenge of isomer contamination that has plagued traditional synthesis routes. By utilizing a specific salt formation strategy involving chiral nucleophiles and suitable bases, this technology enables the production of high-purity intermediates essential for advanced agrochemical and pharmaceutical applications. The core innovation lies in the ability to drive the equilibrium towards the desired 3-halo isomer through precipitation, effectively separating it from the unwanted 6-halo analogues which remain soluble in the reaction medium. This represents a significant leap forward for manufacturers seeking reliable agrochemical intermediate supplier partnerships that prioritize purity and process efficiency.
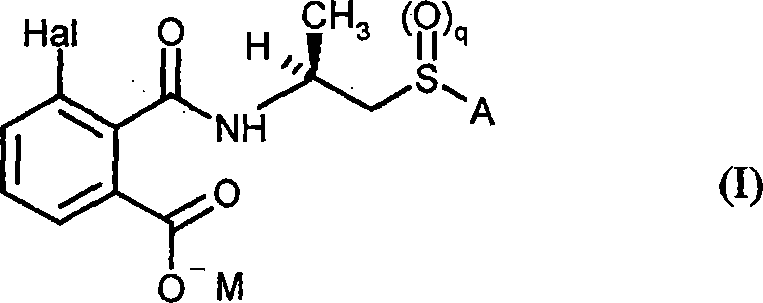
This patent details a comprehensive methodology that transforms readily available 3-halophthalic anhydrides into valuable chiral salts, which serve as versatile precursors for complex phthalamides. The process is designed to be industrially viable, utilizing common reagents such as alkali metal hydroxides and standard organic solvents to achieve exceptional results without the need for exotic catalysts. For R&D directors focused on impurity profiles, this method offers a robust solution to minimize the formation of structural isomers that are notoriously difficult to remove via standard crystallization techniques. Furthermore, the flexibility of the synthetic route allows for the introduction of various functional groups, including sulfides, sulfoxides, and sulfones, providing a broad platform for derivative synthesis. As we delve deeper into the technical specifics, it becomes clear why this technology is pivotal for cost reduction in phthalic acid derivative manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reaction of 3-halophthalic anhydrides with nucleophiles has been fraught with complications regarding regioselectivity, often yielding a mixture of 3-halo and 6-halo isomers that are chemically similar and physically difficult to separate. Literature indicates that conventional processes can generate between 8% to 20% of the undesired 6-halogenated analogue, creating a significant burden on downstream purification units. The separation of these isomers is particularly challenging due to their similar solubility profiles in common organic solvents, frequently necessitating multiple recrystallization steps or expensive chromatographic techniques that result in substantial product loss. In many cases, the cumulative loss of material during the purification of the desired isomer can reach up to 10% to 20%, rendering the overall process economically unviable for large-scale industrial application. This inefficiency not only drives up the cost of goods sold but also creates waste management issues associated with the disposal of solvent-heavy mother liquors containing the rejected isomers.
The Novel Approach
In stark contrast to traditional methods, the novel approach described in the patent leverages the differential solubility of salt forms to achieve high-purity isolation directly from the reaction mixture. By reacting the 3-halophthalic anhydride with a chiral amine in the presence of a base such as lithium or sodium hydroxide, the desired 3-halo derivative forms a salt that precipitates out of the solution, while the unwanted 6-halo isomer remains dissolved. This elegant separation mechanism eliminates the need for complex purification trains, allowing manufacturers to obtain the target intermediate in excellent quality simply through filtration. The process is adaptable to various halogen substituents including fluorine, chlorine, bromine, and iodine, and accommodates a wide range of chiral amines, making it highly versatile for custom synthesis projects. This shift from separation-based purification to reaction-driven selectivity represents a paradigm shift in how we approach the commercial scale-up of complex polymer additives and fine chemical intermediates.
Mechanistic Insights into Regioselective Ring Opening and Salt Precipitation
The mechanistic foundation of this technology rests on the nucleophilic attack of the chiral amine on the carbonyl carbon of the 3-halophthalic anhydride, followed by immediate neutralization with a hydroxide base to form a stable carboxylate salt. The presence of the bulky chiral group on the amine likely imposes steric constraints that favor attack at the less hindered carbonyl position adjacent to the halogen, although the true power of the method lies in the thermodynamic stability of the resulting salt crystal lattice. Once formed, the specific crystal packing of the desired 3-halo salt lowers its solubility in the reaction medium, driving the equilibrium forward and effectively pulling the reaction to completion while excluding the isomeric impurity. This phenomenon is critical for maintaining high optical purity and chemical integrity, as the precipitation step acts as a dynamic filter that continuously removes product from the reactive environment. Understanding this mechanism allows process chemists to fine-tune solvent systems and temperatures to maximize yield, ensuring that the process remains robust even when scaling from laboratory benchtop to multi-ton production vessels.
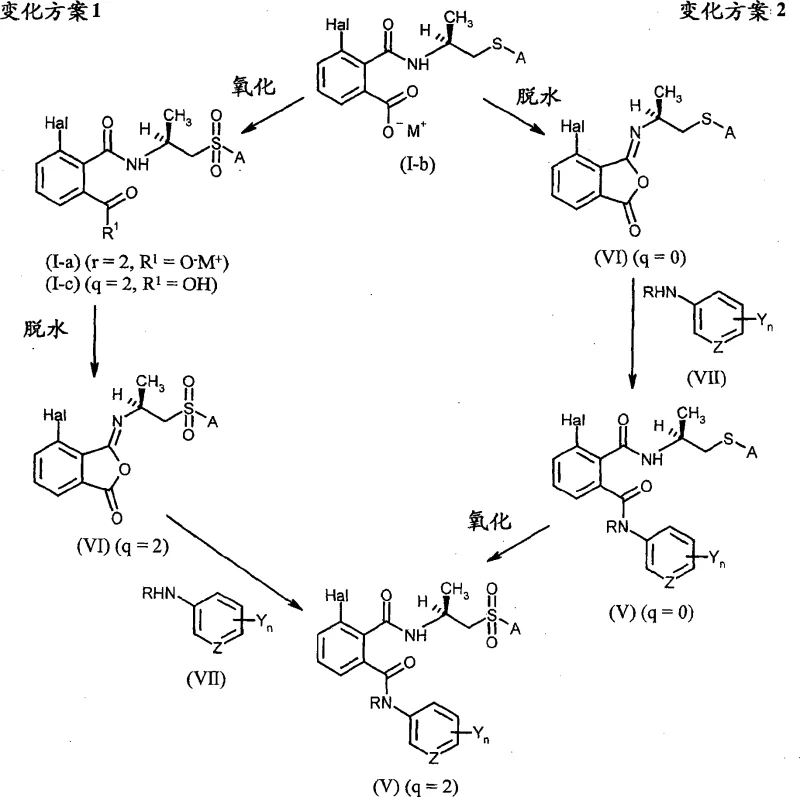
Furthermore, the patent elucidates two distinct synthetic variations for converting these salts into the final phthalamide products, offering flexibility depending on the oxidation state required for the sulfur moiety. In one pathway, the salt is first dehydrated to form an isophthalimide intermediate, which is then coupled with an arylamine, followed by oxidation if a sulfone is desired. Alternatively, the oxidation can be performed on the open-chain salt prior to cyclization, allowing for the selection of oxidants that might be incompatible with the cyclic imide structure. This modularity is invaluable for supply chain planning, as it permits the use of different raw material grades or the adjustment of reaction sequences to accommodate facility constraints. The ability to control the oxidation state (q=0, 1, or 2) with precision ensures that the final active ingredient meets stringent regulatory specifications for impurity limits. Such mechanistic clarity provides confidence to procurement teams looking for reducing lead time for high-purity phthalic acid derivatives.
How to Synthesize Chiral 3-Halophthalic Acid Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable intermediates with high efficiency and minimal waste generation. The process begins with the dissolution of the 3-halophthalic anhydride in a polar aprotic solvent such as dimethylacetamide, followed by the controlled addition of the chiral amine at low temperatures to manage exotherms. Subsequent treatment with an aqueous solution of a base like lithium hydroxide triggers the precipitation of the target salt, which is then isolated by filtration and dried to remove residual solvents. This straightforward workup procedure significantly reduces the operational complexity typically associated with isomer separations, making it an attractive option for facilities aiming to streamline their production workflows. For detailed operational parameters including specific molar ratios and temperature profiles, please refer to the standardized synthesis guide below.
- React 3-halophthalic anhydride with a chiral amine in the presence of a hydroxide base to form the specific salt intermediate.
- Isolate the desired salt via precipitation, leaving unwanted isomers in the solution phase for easy filtration.
- Convert the isolated salt into isophthalimide using a dehydrating agent, then couple with arylamines to form the final phthalamide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers profound advantages in terms of cost structure and supply reliability for global chemical manufacturers. By eliminating the need for extensive purification steps to remove isomeric impurities, the process significantly reduces solvent consumption and energy usage associated with repeated recrystallizations or distillations. This simplification of the manufacturing workflow translates directly into lower operating expenses and a reduced environmental footprint, aligning with modern sustainability goals that are increasingly important to end-users in the pharmaceutical and agrochemical sectors. Moreover, the use of commercially available starting materials such as 3-halophthalic anhydrides and simple chiral amines ensures that the supply chain remains resilient against raw material shortages. The robustness of the precipitation-based isolation method also enhances batch-to-batch consistency, a critical factor for maintaining long-term contracts with regulated industries.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the drastic reduction in yield loss typically incurred during the separation of 3-halo and 6-halo isomers in conventional processes. By leveraging the solubility differences of the salt forms, manufacturers can avoid the costly and material-intensive purification steps that often render traditional methods economically unfeasible. This efficiency gain allows for a more competitive pricing structure without compromising on the quality or purity of the final intermediate. Additionally, the ability to perform reactions at moderate temperatures and pressures reduces the energy load on production facilities, further contributing to overall cost optimization. The elimination of expensive chromatographic resins or specialized separation equipment also lowers capital expenditure requirements for new production lines.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals such as halophthalic anhydrides and alkali hydroxides ensures a stable and secure supply chain that is less susceptible to market volatility. Unlike processes that depend on scarce or proprietary catalysts, this method utilizes standard reagents that can be sourced from multiple vendors globally, mitigating the risk of single-source dependency. The simplicity of the isolation procedure, which relies on basic filtration rather than complex extraction or distillation, also means that production can be easily transferred between different manufacturing sites without significant requalification efforts. This flexibility is crucial for maintaining continuous supply in the face of logistical disruptions or regional demand spikes, ensuring that customers receive their orders on time.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with standard industrial equipment and solvents that are already approved for use in GMP environments. The reduction in solvent usage and waste generation associated with the simplified workup procedure facilitates easier compliance with increasingly stringent environmental regulations regarding volatile organic compound emissions. Furthermore, the high purity of the intermediate reduces the burden on downstream waste treatment facilities, as fewer impurities need to be managed in subsequent synthesis steps. This alignment with green chemistry principles not only improves the corporate sustainability profile but also future-proofs the manufacturing process against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
To assist our partners in evaluating the feasibility of this technology for their specific applications, we have compiled answers to some of the most common technical inquiries regarding the synthesis and handling of these derivatives. These responses are based on the detailed experimental data and process descriptions provided within the patent documentation, ensuring accuracy and relevance for industrial implementation. Understanding these nuances is essential for project managers and technical leads who are responsible for integrating new synthetic routes into existing production portfolios. We encourage stakeholders to review these insights carefully to appreciate the full scope of benefits offered by this innovative approach.
Q: How does this method improve upon conventional 3-halophthalic anhydride reactions?
A: Conventional methods produce a difficult-to-separate mixture of 3-halo and 6-halo isomers, often resulting in significant yield loss during purification. This patented process utilizes a specific salt formation step where the desired isomer precipitates while the unwanted isomer remains in solution, drastically simplifying isolation and improving overall purity.
Q: Can the oxidation step be performed at different stages of the synthesis?
A: Yes, the patent describes flexible synthetic routes where the oxidation of the sulfur moiety (from sulfide to sulfoxide or sulfone) can occur either before or after the cyclization and coupling steps, allowing manufacturers to optimize based on available reagents and equipment.
Q: What are the preferred solvents for the cyclization step?
A: The process supports a wide range of inert diluents including nitriles like butyronitrile and halogenated hydrocarbons such as chlorobenzene or dichloromethane. The use of phase transfer catalysts can further enable these reactions to proceed efficiently even at room temperature.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 3-Halophthalic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates that meet the rigorous demands of modern drug discovery and crop protection development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify the identity and quality of every batch produced. By partnering with us, you gain access to a supply chain that is not only reliable but also deeply knowledgeable about the intricacies of chiral synthesis and regioselective chemistry.
We invite you to contact our technical procurement team to discuss how this patented technology can be tailored to your specific project requirements and volume needs. Our experts are ready to provide a Customized Cost-Saving Analysis that demonstrates the potential economic benefits of switching to this more efficient synthetic route for your target molecules. Please reach out to request specific COA data and route feasibility assessments that will help you make informed decisions about your sourcing strategy. Together, we can drive innovation and efficiency in the production of next-generation agrochemical and pharmaceutical ingredients.
