Advanced Two-Step Synthesis of Cyadox Intermediate for Scalable Veterinary Drug Manufacturing
Advanced Two-Step Synthesis of Cyadox Intermediate for Scalable Veterinary Drug Manufacturing
The global demand for efficient and safe veterinary pharmaceuticals continues to drive innovation in synthetic chemistry, particularly for growth-promoting feed additives like Cyadox. A pivotal advancement in this sector is detailed in patent CN101323599B, which outlines a revolutionary chemical synthesis method for 2-formylcyanoacetylhydrazone-quinoxaline-1,4-dioxide. This compound serves as a critical intermediate and active agent in animal husbandry, offering broad-spectrum antimicrobial properties with a significantly improved safety profile compared to older generations of quinoxaline derivatives. The patent introduces a streamlined methodology that replaces hazardous heavy metal oxidants with benign organic catalysts, marking a substantial shift towards green chemistry in the production of high-purity veterinary drug intermediates. By leveraging this technology, manufacturers can achieve superior product consistency while adhering to increasingly stringent environmental regulations.
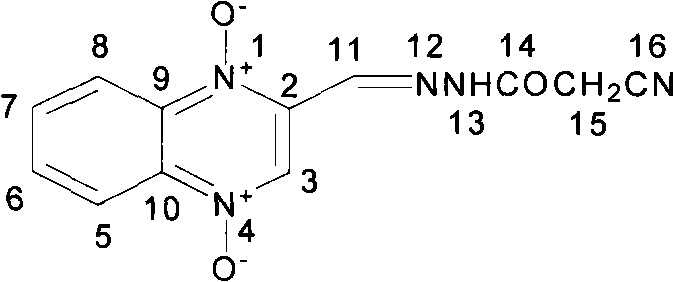
Understanding the molecular architecture is essential for quality control, as illustrated by the structural diagram which details the specific positioning of the cyanoacetylhydrazone moiety on the quinoxaline ring. This precise structural integrity is vital for ensuring the biological efficacy of the final feed additive. As a reliable veterinary drug intermediate supplier, adopting such validated synthetic routes ensures that the supply chain remains robust against regulatory shifts that often penalize toxic manufacturing processes. The transition to this method represents not just a chemical optimization, but a strategic alignment with the future of sustainable agrochemical and pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoxaline-N,N-dioxide derivatives has been plagued by inefficient multi-step protocols that rely on dangerous reagents and energy-intensive conditions. Traditional methods, such as those cited in prior art publication CN101016268A, typically involve a cumbersome three-step reaction sequence that necessitates the use of toxic oxidizing agents like tin anhydride, manganese dioxide, or chromium trioxide. These heavy metal reagents pose severe environmental disposal challenges and introduce significant safety risks to plant personnel, requiring specialized containment and waste treatment infrastructure. Furthermore, conventional processes often demand harsh reaction conditions, including prolonged heating under reflux, which increases energy consumption and the risk of thermal degradation of sensitive intermediates. The complexity of these legacy routes also leads to lower overall yields, often hovering around 44%, resulting in higher raw material costs and increased solvent waste generation per kilogram of product.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN101323599B simplifies the production landscape into a concise two-step process that operates under remarkably mild conditions. By utilizing benzofuroxan and methylglyoxal 1,1-dimethyl acetal as starting materials, the new route achieves cyclization at temperatures ranging from 5°C to 30°C, effectively eliminating the need for energy-draining reflux operations. The replacement of toxic metal oxidants with pyrrolidine, a common and manageable organic catalyst, drastically reduces the toxicity profile of the entire manufacturing workflow. This innovation not only streamlines the operational workflow by removing a full reaction step but also enhances the economic viability of the process through improved atom economy. The comparative data clearly demonstrates the superiority of this approach, showing a tangible increase in efficiency and a reduction in operational complexity.

The visual comparison highlights key metrics where the new invention outperforms existing technology, specifically noting the shift from heated reflux to room temperature reactions and the elimination of hazardous catalysts. For procurement managers, this translates directly into cost reduction in feed additive manufacturing, as the reliance on expensive and regulated heavy metal chemicals is completely removed. The simplified post-treatment process, which avoids complex pH conditioning and filtration of metal sludge, further accelerates production cycles and reduces downtime between batches. This technological leap ensures that the supply of high-purity veterinary drug intermediates remains continuous and less susceptible to disruptions caused by environmental compliance audits or raw material shortages.
Mechanistic Insights into Pyrrolidine-Catalyzed Cyclization
The core of this synthetic breakthrough lies in the efficient construction of the quinoxaline ring system via a nucleophilic attack facilitated by pyrrolidine. In the first step, benzofuroxan reacts with methylglyoxal 1,1-dimethyl acetal in a DMF solvent medium, where the amine catalyst activates the acetal for cyclization. This mechanism allows for the formation of the quinoxaline-1,4-dioxygen-2-formaldehyde dimethyl acetal intermediate with high regioselectivity, minimizing the formation of unwanted isomers that could complicate downstream purification. The mild temperature range of 5°C to 30°C is critical here, as it prevents the decomposition of the unstable N-oxide functionalities which are prone to degradation under the high heat required by traditional methods. By maintaining these gentle conditions, the process preserves the integrity of the quinoxaline backbone, ensuring that the final product meets stringent purity specifications required for animal health applications.
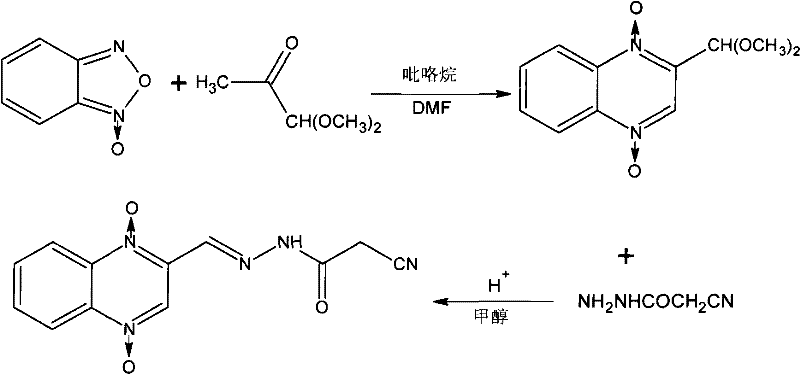
Following the initial cyclization, the second stage involves an acid-catalyzed alcoholysis and condensation reaction that installs the critical cyanoacetylhydrazone side chain. The intermediate acetal is hydrolyzed in methanol with hydrochloric acid to generate the reactive aldehyde in situ, which immediately condenses with cyanoacetohydrazide to form the final target molecule. This tandem operation in a single pot minimizes solvent exchange and handling losses, contributing to the overall high yield observed in experimental embodiments. The mechanistic pathway avoids the generation of halogenated byproducts often associated with chlorinating agents used in older routes, thereby simplifying the impurity profile. For R&D directors, this clean reaction profile means less time spent on developing complex chromatographic purification methods, allowing for a more direct path from crude reaction mass to commercial-grade API intermediate.
How to Synthesize 2-formylcyanoacetylhydrazone-quinoxaline-1,4-dioxide Efficiently
The practical implementation of this synthesis requires precise control over stoichiometry and addition rates to maximize the benefits of the catalytic system. The process begins with the dissolution of benzofuroxan in DMF, followed by the controlled addition of the acetal and the pyrrolidine catalyst, ensuring the exotherm is managed within the 5-30°C window. Detailed standard operating procedures regarding mixing speeds, addition times, and crystallization parameters are essential for reproducing the high yields reported in the patent embodiments. Operators must adhere to the specified molar ratios, particularly the excess of methylglyoxal acetal, to drive the equilibrium towards the desired intermediate without generating polymeric side products. The subsequent conversion to the final hydrazone relies on the careful addition of aqueous cyanoacetohydrazide to the acidic methanolic solution, where temperature control remains paramount to prevent hydrolysis of the nitrile group.
- React benzofuroxan with methylglyoxal 1,1-dimethyl acetal in DMF using pyrrolidine catalyst at 5-30°C to form the quinoxaline intermediate.
- Perform alcoholysis of the intermediate in methanol with hydrochloric acid, then react with cyanoacetohydrazide at 10-35°C.
- Filter, wash with water and ethanol, and dry to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the bottom line and logistical flow, the adoption of this patented synthesis route offers profound strategic benefits that extend beyond simple chemistry. The elimination of heavy metal catalysts removes a significant cost center associated with the purchase, handling, and disposal of hazardous substances, leading to substantial cost savings in raw material procurement. Furthermore, the use of commodity solvents like methanol, ethanol, and DMF, which are readily available in the global chemical market, insulates the supply chain from the volatility often seen with specialized or regulated reagents. The ability to run reactions at ambient temperatures significantly lowers utility costs related to heating and cooling, enhancing the overall energy efficiency of the manufacturing plant. These factors combine to create a more resilient supply chain capable of meeting large-volume demands without the bottlenecks imposed by complex safety protocols or waste treatment limitations.
- Cost Reduction in Manufacturing: The removal of expensive and toxic oxidants like chromium trioxide and tin anhydride eliminates the need for costly waste remediation and specialized storage facilities. By switching to pyrrolidine and common acids, the variable cost per kilogram of product is significantly decreased, improving margin potential for high-volume production runs. Additionally, the reduction from three steps to two steps inherently lowers labor costs and equipment occupancy time, allowing for higher throughput within existing infrastructure.
- Enhanced Supply Chain Reliability: Sourcing raw materials becomes more predictable as the process relies on bulk commodities rather than niche fine chemicals that may face supply disruptions. The stability of the reaction conditions reduces the risk of batch failures due to thermal runaway or reagent instability, ensuring consistent delivery schedules to downstream pharmaceutical customers. This reliability is crucial for maintaining long-term contracts with major animal health companies that require uninterrupted supply of critical feed additives.
- Scalability and Environmental Compliance: The mild reaction conditions facilitate easier scale-up from pilot plant to commercial tonnage, as heat transfer limitations are less severe compared to exothermic reflux processes. The reduced toxicity of reagents simplifies regulatory compliance and environmental permitting, accelerating the timeline for bringing new production capacity online. This alignment with green chemistry principles future-proofs the manufacturing asset against tightening global environmental regulations regarding heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these nuances helps stakeholders make informed decisions about integrating this process into their existing manufacturing portfolios. The focus remains on practical feasibility, safety improvements, and economic viability.
Q: How does this new synthesis method improve safety compared to traditional routes?
A: The novel process eliminates the use of highly toxic heavy metal oxidants such as tin anhydride and chromium trioxide required in conventional 3-step methods, significantly reducing environmental hazards and operator risk.
Q: What are the yield improvements observed in this patented technology?
A: Comparative data indicates a yield increase from approximately 44% in prior art methods to 58% or higher in this optimized 2-step process, demonstrating superior atom economy.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (5-35°C) and do not require energy-intensive reflux or complex pH adjustments, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-formylcyanoacetylhydrazone-quinoxaline-1,4-dioxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global veterinary pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. By leveraging the efficiencies of the CN101323599B process, we can offer our partners a product that is not only chemically superior but also economically optimized for large-scale feed additive manufacturing.
We invite you to collaborate with us to explore how this innovative synthesis route can enhance your product portfolio and reduce your overall manufacturing footprint. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for sustainable and cost-effective chemical sourcing.
