Advanced Manufacturing of Vitamin B1 Intermediates via Non-Toxic Cyclization Routes
The global demand for high-purity vitamins continues to drive innovation in pharmaceutical intermediate manufacturing, particularly for essential nutrients like Vitamin B1 (Thiamine). A pivotal advancement in this sector is documented in patent CN103012282A, which details a revolutionary synthetic method for producing 2-methyl-4-amino-5-aminomethyl pyrimidine, a critical precursor in the Vitamin B1 value chain. This technology represents a paradigm shift by fundamentally restructuring the chemical pathway to exclude hazardous reagents that have historically plagued the industry. By replacing traditional toxic inputs with safer, more direct condensation reactions, this method addresses both the environmental sustainability mandates and the rigorous purity standards required by modern regulatory bodies. The strategic importance of this patent lies not just in the chemical novelty, but in its ability to decouple production capacity from the supply volatility of chlorinated anilines. For international procurement teams, understanding this transition is vital as it signals a move towards more resilient and compliant supply chains for bulk vitamin manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-methyl-4-amino-5-aminomethyl pyrimidine has relied heavily on the use of o-chloroaniline as a primary starting material, a dependency that introduces severe operational and safety liabilities. The conventional process involves reacting alpha-sodium substituted formyl-beta-formyl amine propionitrile with o-chloroaniline hydrochloride to form a complex intermediate, which subsequently undergoes ring closure. This legacy pathway is fraught with inefficiencies, primarily because o-chloroaniline is a substance of very high concern due to its severe toxicity and potential carcinogenicity, necessitating expensive containment and waste treatment protocols. Furthermore, the intermediate formed in this traditional route, alpha-(o-chloroaniline)-aminomethyl-beta-formamido group propionitrile, requires a rigorous drying step before it can proceed to cyclization. This physical handling of fine powders generates significant dust, increasing labor intensity and posing inhalation risks to plant personnel while simultaneously creating opportunities for product loss and contamination. The energy consumption associated with vacuum drying and the subsequent recovery of the chloroaniline moiety further erodes the economic viability of this older method, making it increasingly obsolete in a cost-sensitive market.
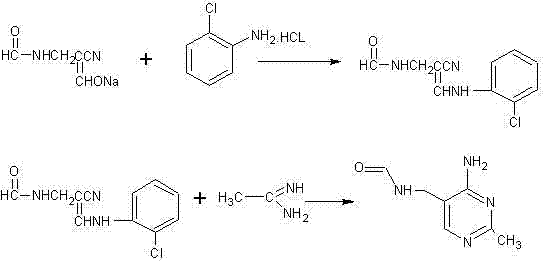
The Novel Approach
In stark contrast to the cumbersome legacy processes, the innovative methodology described in the patent data achieves the synthesis of the target pyrimidine through a direct and streamlined condensation strategy that completely omits the use of o-chloroaniline. This novel approach utilizes alpha-sodium substituted formyl-beta-formyl amine propionitrile and ethanamidine as the core building blocks, effectively shortening the synthetic sequence by eliminating the need for the toxic aniline coupling step. By bypassing the formation of the chloroaniline-containing intermediate, the process removes the necessity for the energy-intensive drying operation, thereby allowing the reaction to proceed in a more fluid, solution-phase manner. This structural simplification of the manufacturing route not only mitigates the environmental hazards associated with chlorinated waste streams but also drastically reduces the physical labor required for material handling. The result is a cleaner, more atom-economical process that aligns perfectly with the principles of green chemistry, offering a robust alternative for manufacturers seeking to upgrade their production capabilities without compromising on yield or quality.
Mechanistic Insights into Direct Pyrimidine Cyclization
The chemical elegance of this new synthesis lies in its precise control over reaction conditions to facilitate a direct heterocyclic ring closure. The mechanism initiates with the careful acidification of the sodium salt precursor, alpha-sodium substituted formyl-beta-formyl amine propionitrile, using hydrochloric acid gas in a methanol solvent system at controlled temperatures ranging from 0°C to 60°C. This step converts the unstable sodium salt into the corresponding enol form, alpha-enol base-beta-formamido group propionitrile, which is the active nucleophile required for the subsequent cyclization. Concurrently, the electrophilic partner, ethanamidine, is generated in situ by treating acetamidine hydrochloride with sodium methylate in liquid methanol, ensuring a high concentration of the free base is available for reaction. The convergence of these two reactive species occurs in a non-polar solvent such as toluene, where the thermodynamic drive for ring closure is enhanced by the continuous removal of water via azeotropic distillation. This mechanistic pathway ensures that the pyrimidine ring forms efficiently with minimal side reactions, leading to a product profile that is easier to purify compared to the complex mixtures generated by the chloroaniline route.
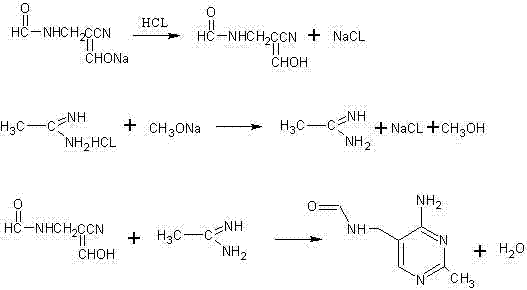
From an impurity control perspective, this mechanism offers distinct advantages by avoiding the introduction of chlorine atoms into the molecular scaffold until absolutely necessary, or in this specific improved case, avoiding them entirely in the intermediate stages. In traditional methods, residual o-chloroaniline is a persistent genotoxic impurity that is notoriously difficult to purge from the final API, often requiring multiple recrystallizations or chromatographic steps. By designing a synthesis that never introduces the chloroaniline moiety, the new process inherently prevents the formation of chlorinated byproducts and related structural analogs that typically complicate the purification landscape. The use of toluene as a reaction medium for the cyclization step further aids in impurity management, as it allows for the effective separation of water and polar byproducts, driving the equilibrium towards the desired product while leaving hydrophilic impurities in the aqueous phase or allowing them to be washed away easily. This results in a crude product with significantly higher purity, reducing the burden on downstream processing units and ensuring that the final intermediate meets the stringent specifications required for vitamin synthesis.
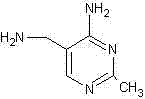
How to Synthesize 2-methyl-4-amino-5-aminomethyl pyrimidine Efficiently
Implementing this advanced synthetic route requires a disciplined approach to reaction engineering, specifically focusing on the sequential generation of reactive intermediates and their immediate consumption to maximize yield. The process begins with the conversion of the stable sodium salt into its reactive enol counterpart, followed by the parallel preparation of the free amidine base, culminating in a thermal cyclization step that leverages azeotropic water removal. Operators must maintain strict temperature controls during the acidification and free base generation phases to prevent premature decomposition or polymerization of the sensitive nitrile functionalities. The following guide outlines the standardized operational parameters derived from the patent examples to ensure reproducible results on a commercial scale.
- Acidify alpha-sodium substituted formyl-beta-formyl amine propionitrile in methanol with hydrochloric acid gas to form the enol intermediate.
- Generate free ethanamidine by reacting acetamidine hydrochloride with sodium methylate in methanol under controlled temperature.
- Perform ring-closure reaction between the enol intermediate and ethanamidine in toluene with water separation to yield the final pyrimidine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this non-chloroaniline synthetic route translates into tangible strategic benefits that extend far beyond simple chemical curiosity. The primary value proposition is the substantial reduction in operational risk associated with handling hazardous materials, which directly impacts insurance premiums, regulatory compliance costs, and community relations for manufacturing sites. By eliminating the need for o-chloroaniline, companies can avoid the complex logistics and storage requirements mandated for toxic substances, thereby simplifying their raw material inventory management. Furthermore, the removal of the intermediate drying step represents a significant optimization of the production timeline, as it eliminates a batch-processing bottleneck that traditionally limits throughput. This streamlining allows for faster turnover of reactor vessels and a more continuous flow of materials, enhancing the overall agility of the supply chain to respond to market fluctuations in Vitamin B1 demand.
- Cost Reduction in Manufacturing: The economic implications of this process improvement are profound, driven largely by the elimination of expensive waste treatment protocols required for chlorinated organic compounds. Without the need to recover or dispose of large quantities of o-chloroaniline, the variable costs associated with environmental compliance are drastically lowered, improving the overall margin profile of the intermediate. Additionally, the energy savings achieved by removing the vacuum drying stage contribute to a lower utility bill per kilogram of product, as thermal energy is one of the most significant cost drivers in fine chemical manufacturing. The simplified workflow also reduces labor costs, as fewer manual interventions are required for filtering, drying, and transferring dusty solids, allowing personnel to focus on higher-value monitoring and quality control tasks. These cumulative efficiencies create a leaner cost structure that provides a competitive edge in price-sensitive global markets.
- Enhanced Supply Chain Reliability: Relying on o-chloroaniline exposes the supply chain to volatility, as the availability of this toxic precursor can be impacted by environmental regulations and production shutdowns at upstream facilities. By shifting to a synthesis based on more benign and widely available starting materials like acetamidine and formyl propionitrile derivatives, manufacturers can diversify their supplier base and reduce the risk of raw material shortages. The robustness of the new chemical route ensures consistent production schedules, as there are fewer potential failure points related to equipment corrosion or catalyst poisoning that often plague processes involving aggressive halogenated reagents. This stability is crucial for long-term supply agreements with major pharmaceutical clients who prioritize continuity of supply above all else, ensuring that production targets are met without unexpected interruptions.
- Scalability and Environmental Compliance: Scaling chemical processes that involve toxic dusts and volatile chlorinated solvents presents significant engineering challenges, often requiring specialized containment systems that are difficult to replicate across different manufacturing sites. The new method, which operates primarily in solution with standard organic solvents like methanol and toluene, is inherently easier to scale from pilot plant to multi-tonne commercial production without requiring massive capital expenditure on new infrastructure. The reduction in hazardous waste generation aligns with increasingly strict global environmental standards, facilitating easier permitting for capacity expansion in regions with tight ecological controls. This scalability ensures that the technology remains viable as demand grows, allowing suppliers to ramp up production volumes rapidly to capture market share without being hindered by regulatory bottlenecks or waste disposal limitations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Vitamin B1 intermediate synthesis. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical rationale behind the process upgrade. Understanding these details is essential for technical teams evaluating the feasibility of adopting this greener manufacturing route.
Q: Why is the elimination of o-chloroaniline significant for Vitamin B1 production?
A: Eliminating o-chloroaniline removes a highly toxic and environmentally hazardous raw material from the supply chain, significantly reducing waste treatment costs and operator safety risks associated with traditional synthesis methods.
Q: How does the new cyclization method improve process efficiency?
A: The novel approach bypasses the isolation and drying of unstable intermediates, allowing for a more continuous flow that reduces labor intensity and minimizes product loss during physical handling steps.
Q: What solvents are utilized in this optimized synthetic pathway?
A: The process primarily utilizes methanol for the initial acidification and free base generation steps, followed by toluene for the final cyclization reaction to facilitate effective water removal and drive the equilibrium forward.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-methyl-4-amino-5-aminomethyl pyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer and more efficient synthetic routes is critical for the long-term sustainability of the pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the non-chloroaniline cyclization method can be seamlessly transferred from the laboratory to industrial reactors. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2-methyl-4-amino-5-aminomethyl pyrimidine meets the highest international standards for Vitamin B1 synthesis. Our capability to handle complex heterocyclic chemistry allows us to offer a reliable source of this key intermediate, free from the supply risks associated with traditional toxic manufacturing methods.
We invite global partners to engage with our technical procurement team to discuss how this optimized route can enhance your specific supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener process for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your Vitamin B1 production remains competitive, compliant, and uninterrupted in an evolving regulatory landscape.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
