Advanced Synthesis of Sacubitril Valsartan Sodium Impurity for Global Quality Control and Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of patient safety has elevated the importance of impurity profiling to a critical regulatory mandate, particularly for complex combination drugs like Sacubitril Valsartan Sodium. As detailed in the groundbreaking patent CN110878039A, the ability to synthesize specific degradation products and process-related impurities with high fidelity is no longer just a research exercise but a cornerstone of quality assurance for global supply chains. This patent introduces a transformative preparation method for Impurity Compound II, a key structural analog that must be rigorously monitored during the manufacturing of the active pharmaceutical ingredient (API). By leveraging a novel PyBOP-mediated cyclization strategy, this technology addresses the longstanding challenges of low yield, poor stereoselectivity, and hazardous reaction conditions that have plagued previous synthetic routes. For R&D Directors and Quality Control heads, mastering this synthesis pathway ensures that the final drug substance meets the stringent purity specifications required by major health authorities, thereby safeguarding both patient outcomes and brand reputation in the competitive cardiovascular therapeutic market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sacubitril Valsartan Sodium impurities has been fraught with significant technical and safety hurdles that render traditional methods unsuitable for modern, large-scale industrial production. Prior art techniques, such as those disclosed in WO2009/90251, often relied on catalytic hydrogenation processes that introduced severe safety risks due to the potential for hydrogen leakage and explosion in a manufacturing environment. Furthermore, alternative synthetic pathways described in documents like WO2008/83967 necessitated the use of n-butyllithium, a highly pyrophoric reagent that requires cryogenic conditions down to -78°C to maintain stability. These extreme conditions not only escalate operational costs through specialized equipment requirements but also introduce substantial variability in reaction control, often resulting in racemic mixtures that are notoriously difficult to separate. The inability to distinguish chirality in these older methods means that manufacturers are left with crude products requiring extensive and wasteful purification steps, ultimately compromising the overall efficiency and environmental sustainability of the supply chain.
The Novel Approach
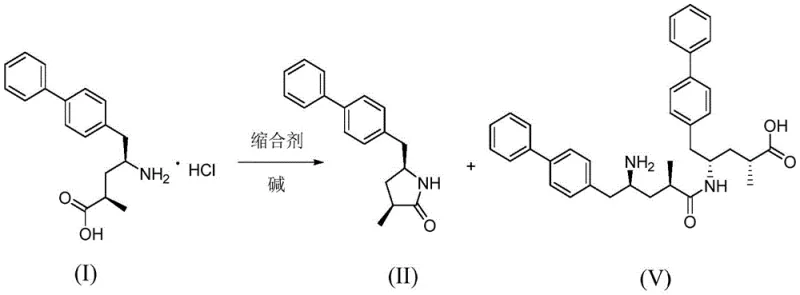
In stark contrast to these hazardous and inefficient legacy processes, the novel approach outlined in the patent utilizes a sophisticated condensation strategy that fundamentally redefines the safety and efficacy profile of impurity synthesis. By employing benzotriazole-1-yl-oxytripyrrolidinophosphonium hexafluorophosphate (PyBOP) as the primary condensing agent, the reaction proceeds through a much milder and more controllable mechanism that avoids the need for dangerous gases or cryogenic cooling. This method allows for the direct cyclization of the precursor Compound I into the target lactam structure with remarkable precision, effectively bypassing the formation of unwanted isomers that complicate downstream processing. The operational simplicity of this route means that it can be readily adapted for commercial scale-up without the need for exotic infrastructure, providing a reliable pharmaceutical intermediates supplier with a robust platform for consistent production. This shift from harsh chemical environments to温和 (mild) conditions represents a paradigm shift in how critical reference standards are manufactured, ensuring that supply continuity is maintained even under rigorous regulatory scrutiny.
Mechanistic Insights into PyBOP-Catalyzed Cyclization
The core innovation of this synthesis lies in the intricate mechanistic pathway facilitated by the PyBOP condensing agent, which orchestrates the molecular rearrangement with exceptional selectivity. Under alkaline conditions, the carboxylate anion of the starting material attacks the phosphorus center of the PyBOP reagent, generating a highly reactive acyloxyphosphonium intermediate. This activated species is subsequently attacked by benzotriazole oxyanions to form a transient acyl halide-like structure, which is then primed for an intramolecular nucleophilic attack by the pendant amino group. This sequence of events drives the formation of the stable five-membered lactam ring characteristic of Impurity Compound II, effectively outcompeting intermolecular reactions that would otherwise lead to polymeric byproducts. The elegance of this mechanism is that it leverages the specific electronic properties of the phosphonium salt to lower the activation energy for cyclization, allowing the reaction to proceed rapidly even at reduced temperatures where competing side reactions are kinetically suppressed.
Furthermore, the control of impurity profiles is meticulously managed through the suppression of Byproduct V, a dimeric species that arises from intermolecular amide condensation. The patent data reveals that without precise temperature regulation and base selection, the reaction equilibrium shifts unfavorably towards this dimer, rendering the target product unobtainable in high purity. By maintaining the reaction temperature strictly between -10°C and 0°C, the kinetic energy of the system is tuned to favor the intramolecular closure over intermolecular coupling. Additionally, the choice of base, specifically N,N-diisopropylethylamine (DIPEA), plays a crucial role in scavenging protons without inducing elimination side reactions, thereby preserving the integrity of the chiral centers. This deep understanding of the reaction dynamics allows chemists to fine-tune the process parameters to achieve purity levels exceeding 97%, a benchmark that is essential for accurate analytical method validation and regulatory compliance in the pharmaceutical sector.
How to Synthesize Sacubitril Valsartan Sodium Impurity Efficiently
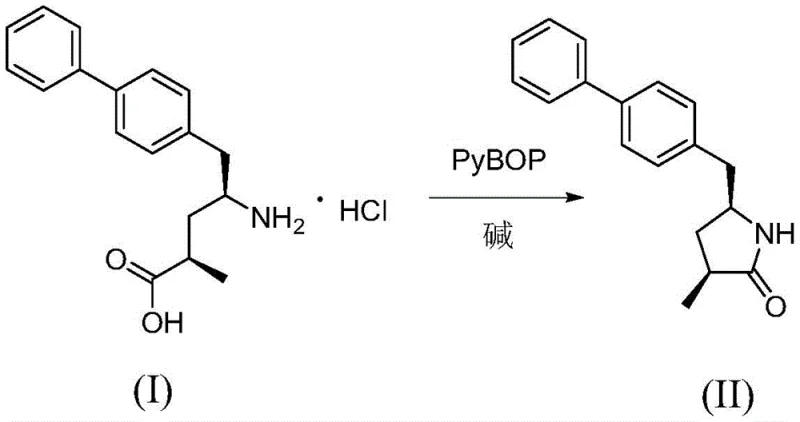
To implement this high-yield synthesis in a laboratory or pilot plant setting, operators must adhere to a strict protocol that emphasizes temperature control and reagent stoichiometry to maximize the formation of the target lactam. The process begins with the dissolution of the amino acid precursor in an aprotic solvent, followed by the careful addition of the PyBOP activator to generate the reactive intermediate. Detailed standardized synthesis steps are provided below to ensure reproducibility and safety across different production batches.
- Dissolve Compound I in an aprotic organic solvent such as acetonitrile and add the PyBOP condensing agent.
- Slowly dropwise add a base like DIPEA while maintaining the reaction temperature strictly between -10°C and 0°C.
- Filter the reaction mixture, perform aqueous workup with saturated ammonium chloride, and purify the solid via pulping and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers profound strategic advantages that extend far beyond simple chemical transformation, directly impacting the bottom line through risk mitigation and operational efficiency. By eliminating the need for hazardous hydrogenation equipment or cryogenic reactors capable of reaching -78°C, manufacturing facilities can significantly reduce their capital expenditure and maintenance overheads associated with specialized safety infrastructure. The mild reaction conditions also translate to lower energy consumption, as the process does not require intensive cooling or heating cycles, aligning with global sustainability goals and reducing the carbon footprint of the manufacturing operation. Moreover, the high selectivity of the PyBOP-mediated reaction minimizes the generation of waste streams and simplifies the purification workflow, leading to substantial cost savings in solvent usage and waste disposal fees. This streamlined process ensures a more reliable supply of high-purity reference materials, reducing the risk of production delays caused by failed batches or complex purification bottlenecks.
- Cost Reduction in Manufacturing: The transition to this novel method eliminates the dependency on expensive and dangerous reagents like n-butyllithium, which require specialized handling and storage protocols that drive up operational costs. By utilizing commercially available and stable reagents like PyBOP and DIPEA, the overall cost of goods sold is optimized through simplified procurement logistics and reduced safety compliance burdens. The high yield and purity achieved in a single step also reduce the need for repetitive recrystallization or chromatographic purification, further driving down the cost reduction in pharmaceutical intermediates manufacturing and enhancing overall margin potential for the final drug product.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent output quality, which is critical for maintaining uninterrupted supply chains in the highly regulated pharmaceutical industry. Unlike prior methods that were prone to safety shutdowns or variable yields due to sensitive reaction conditions, this process offers a stable and predictable production profile that supports long-term planning. The use of common solvents like acetonitrile and standard bases ensures that raw material availability is not a bottleneck, thereby reducing lead time for high-purity pharmaceutical intermediates and securing the continuity of supply for downstream API manufacturers who rely on timely delivery of quality control standards.
- Scalability and Environmental Compliance: The inherent safety of operating at near-ambient temperatures makes this process exceptionally suitable for commercial scale-up of complex pharmaceutical intermediates without requiring massive engineering modifications. The reduction in hazardous waste generation and the avoidance of toxic heavy metals or explosive gases align with increasingly stringent environmental regulations, facilitating smoother regulatory approvals and audits. This environmental compatibility not only protects the company from potential liabilities but also enhances its corporate social responsibility profile, making it a preferred partner for global pharmaceutical companies seeking sustainable and compliant supply chain solutions.
Frequently Asked Questions (FAQ)
The following questions address critical technical and operational concerns regarding the implementation of this synthesis method, providing clarity for stakeholders evaluating its integration into their quality control workflows. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, ensuring that all answers are grounded in verified scientific evidence.
Q: Why is PyBOP preferred over other condensing agents for this impurity synthesis?
A: PyBOP uniquely promotes the formation of the target lactam ring while minimizing the formation of intermolecular amide byproducts, which is a common failure point with agents like CDI or EDCI/HOBT in this specific steric environment.
Q: What are the safety advantages of this method compared to prior art?
A: Unlike prior methods requiring hazardous hydrogenation or cryogenic n-butyllithium at -78°C, this process operates at mild temperatures (-10°C to 0°C) without explosive gases or pyrophoric reagents.
Q: How does temperature control affect the purity of Impurity Compound II?
A: Maintaining the temperature between -10°C and 0°C is critical; higher temperatures significantly increase the formation of Byproduct V, drastically reducing the purity and yield of the target impurity reference standard.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sacubitril Valsartan Sodium Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of modern drug synthesis demands a partner with unparalleled technical expertise and a commitment to excellence in every batch produced. Our team of seasoned chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We understand that achieving stringent purity specifications is non-negotiable for regulatory success, which is why our rigorous QC labs employ state-of-the-art analytical instrumentation to verify every parameter of the final product. By choosing us as your partner, you gain access to a supply chain that is not only robust and reliable but also deeply integrated with the latest advancements in process chemistry, guaranteeing that your quality control standards are met with precision and consistency.
We invite you to collaborate with us to optimize your supply chain and secure a competitive advantage in the global market. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to reach out today to request specific COA data and comprehensive route feasibility assessments that will demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals. Let us help you navigate the complexities of pharmaceutical intermediate sourcing with confidence and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
