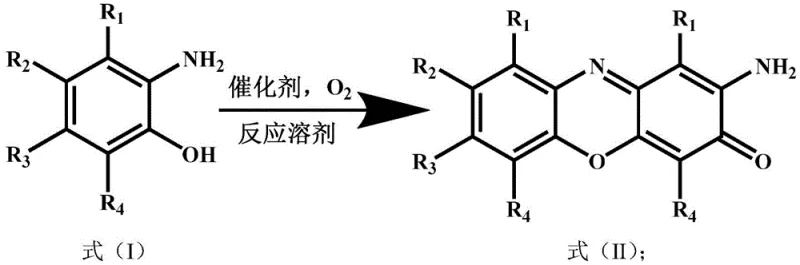
Scalable Green Synthesis of 2-Amino-3H-Phenoxazin-3-One for Commercial API Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally sustainable pathways for the production of critical heterocyclic intermediates. A significant technological breakthrough in this domain is documented in Chinese Patent CN113441135B, which details a novel preparation method for 2-amino-3H-phenoxazin-3-one and its derivatives. This specific compound, often identified by CAS number 1916-59-2, serves as a pivotal building block in the synthesis of bioactive molecules, including potential antibiotics and anticancer agents. The patent introduces a paradigm shift by utilizing a cerium-manganese composite oxide catalyst to facilitate the oxidative cyclization of o-aminophenol precursors. Unlike legacy processes that rely on stoichiometric toxic oxidants, this innovation leverages molecular oxygen or air as the terminal oxidant, aligning perfectly with modern green chemistry principles. For R&D directors and procurement strategists, understanding the implications of this technology is crucial for optimizing supply chains and reducing the environmental footprint of API manufacturing. The method promises not only high chemical efficiency but also a simplified downstream processing workflow that translates directly into commercial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenoxazinone scaffolds has been plagued by significant safety and environmental challenges associated with traditional oxidizing agents. Prior art frequently describes the use of heavy metal-based oxidants such as potassium dichromate, lead oxide, or potassium ferricyanide, which generate substantial quantities of hazardous waste streams requiring complex and costly disposal protocols. Furthermore, alternative methods employing hydrogen peroxide, while seemingly greener, often necessitate the use of large excesses to drive conversion, creating severe safety hazards related to thermal runaway and potential explosion risks during large-scale amplification. These conventional routes also frequently suffer from poor atom economy and require harsh reaction conditions that can degrade sensitive functional groups on the substrate. The reliance on homogeneous metal catalysts in some newer methods introduces another bottleneck, as removing trace metal residues to meet stringent pharmaceutical purity specifications often demands additional purification steps involving expensive scavengers. These cumulative inefficiencies result in inflated production costs and extended lead times, making the reliable supply of high-purity phenoxazinone intermediates a persistent challenge for global supply chain managers.
The Novel Approach
The methodology disclosed in the patent data presents a transformative solution by employing a heterogeneous cerium-manganese composite oxide catalyst that operates efficiently under mild conditions. This innovative approach utilizes air or oxygen as the primary oxidant, effectively eliminating the need for hazardous stoichiometric oxidants and significantly reducing the generation of toxic byproducts. The reaction proceeds smoothly in common organic solvents such as 1,4-dioxane or toluene at moderate temperatures, typically around 80°C, ensuring a safe operational profile suitable for industrial scale-up. The heterogeneous nature of the catalyst allows for easy separation via simple filtration, thereby streamlining the workup process and minimizing solvent consumption. By replacing dangerous peroxide-based systems with a controlled aerobic oxidation process, this method mitigates the risk of explosive decomposition, offering a much safer environment for plant operators. The robustness of this catalytic system enables consistent high-yield production, providing a stable foundation for long-term commercial manufacturing agreements.
Mechanistic Insights into Ce-Mn Catalyzed Oxidative Cyclization
The core of this technological advancement lies in the unique redox properties of the cerium-manganese composite oxide, which facilitates the efficient transfer of oxygen atoms to the substrate. The catalytic cycle likely involves the activation of molecular oxygen on the surface of the mixed metal oxide, generating reactive oxygen species that selectively oxidize the ortho-aminophenol moiety. This surface-mediated mechanism ensures high chemoselectivity, preventing over-oxidation or degradation of the sensitive amine and phenol functional groups present in the starting material. The synergy between cerium and manganese creates active sites that lower the activation energy for the cyclization step, allowing the reaction to proceed rapidly at 80°C without the need for extreme pressure or temperature. This mechanistic efficiency is evidenced by the exceptional conversion rates observed, where the target phenoxazinone ring is formed with minimal formation of side products. For process chemists, this level of control is vital as it simplifies the impurity profile, reducing the burden on analytical quality control and ensuring that the final material meets the rigorous specifications required for pharmaceutical applications.
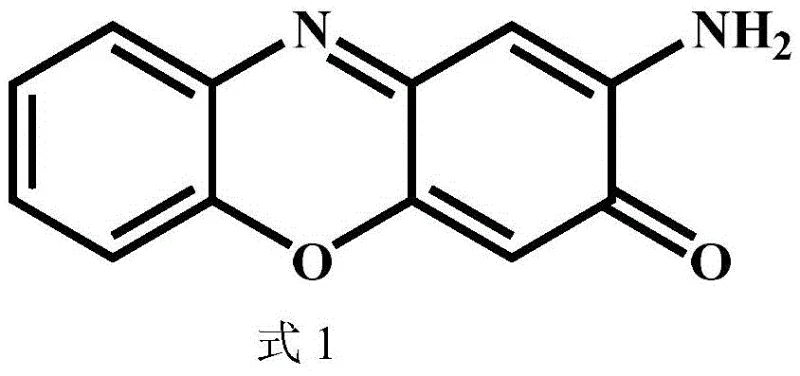
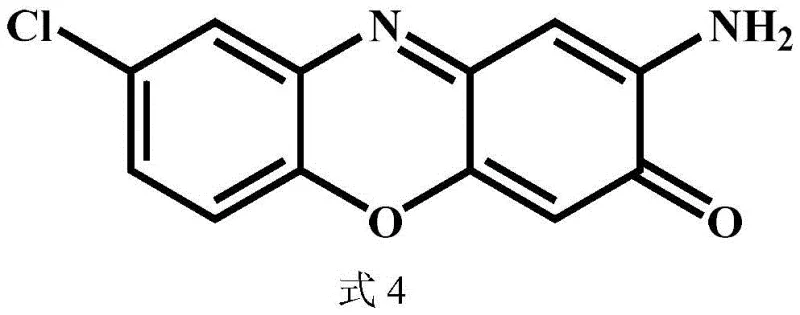
Beyond the primary transformation, the catalyst system demonstrates remarkable tolerance towards various substituents on the aromatic ring, which is critical for generating diverse libraries of analogs. The electronic and steric properties of substituents such as methyl, fluoro, or chloro groups do not significantly inhibit the catalytic activity, allowing for the synthesis of a wide range of derivatives with consistent performance. This versatility is essential for medicinal chemistry programs where structure-activity relationship (SAR) studies require rapid access to substituted intermediates. The high selectivity of the reaction ensures that even with electron-withdrawing or donating groups, the oxidative coupling occurs specifically at the desired positions to form the phenoxazine core. Impurity control is further enhanced by the stability of the catalyst, which does not leach significant amounts of metal into the solution, thereby avoiding contamination issues that could complicate downstream purification. This clean reaction profile supports the production of high-purity intermediates, directly addressing the concerns of R&D directors regarding the quality and consistency of raw materials used in drug substance manufacturing.
How to Synthesize 2-Amino-3H-Phenoxazin-3-One Efficiently
Implementing this synthesis route in a production environment involves a straightforward sequence of unit operations that leverage standard chemical engineering equipment. The process begins with the preparation of the cerium-manganese catalyst via a simple precipitation method using inexpensive salt precursors, followed by calcination to activate the material. Once the catalyst is ready, the reaction is conducted by charging the o-aminophenol substrate and solvent into a pressure vessel, adding the catalyst, and pressurizing with air or oxygen. The mixture is then heated to the optimal temperature range of 80°C and maintained for approximately 8 hours to ensure complete conversion. Following the reaction, the solid catalyst is removed by filtration, and the product is isolated through solvent distillation and recrystallization. This streamlined workflow minimizes the number of processing steps and reduces the overall cycle time compared to traditional methods.
- Prepare the cerium-manganese composite oxide catalyst via simple precipitation of cerium and manganese salts followed by calcination at 100-400°C.
- Charge the reactor with o-aminophenol substrate, catalyst, and 1,4-dioxane solvent, then pressurize with air or oxygen to 0.1-2MPa.
- Maintain reaction temperature at 80°C for 8 hours, followed by filtration, distillation, and recrystallization to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this cerium-manganese catalytic technology offers profound benefits for procurement managers and supply chain heads focused on cost optimization and risk mitigation. The elimination of expensive and hazardous oxidants like potassium dichromate or hydrogen peroxide drastically reduces the raw material costs associated with the synthesis. Furthermore, the removal of heavy metal scavenging steps significantly lowers the consumption of auxiliary chemicals and reduces waste disposal fees, contributing to a leaner and more cost-effective manufacturing process. The use of air or oxygen as an oxidant not only cuts costs but also secures the supply chain against fluctuations in the price of specialized chemical reagents. The simplicity of the catalyst preparation and recovery means that production can be scaled up rapidly without requiring complex new infrastructure, ensuring supply continuity even during periods of high demand. These factors combine to create a resilient supply model that protects margins and enhances competitiveness in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The transition to a heterogeneous cerium-manganese catalyst eliminates the need for costly homogeneous metal catalysts and the subsequent expensive purification steps required to remove metal residues. By utilizing air as a free and abundant oxidant instead of purchasing stoichiometric amounts of specialized oxidizing agents, the direct material costs are substantially decreased. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, leading to higher overall yields and reduced loss of valuable starting materials. This efficiency translates into significant economic savings per kilogram of produced intermediate, allowing for more competitive pricing strategies in B2B negotiations.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as o-aminophenol and common solvents like 1,4-dioxane ensures that raw material sourcing is stable and less susceptible to geopolitical disruptions. The robustness of the catalytic system allows for consistent batch-to-batch reproducibility, which is critical for maintaining long-term supply contracts with pharmaceutical clients. The safety improvements inherent in using air rather than explosive peroxides reduce the risk of plant shutdowns due to safety incidents, thereby guaranteeing uninterrupted delivery schedules. This reliability is a key value proposition for supply chain heads who prioritize vendor stability and on-time performance metrics.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard pressure reactors and filtration equipment that are readily available in most multipurpose chemical plants. The green nature of the synthesis, characterized by the absence of toxic heavy metal waste and the use of benign oxidants, simplifies regulatory compliance and environmental permitting. This alignment with increasingly strict environmental regulations future-proofs the manufacturing asset against potential legislative changes. The ease of waste treatment and the reduced environmental footprint enhance the corporate sustainability profile, which is becoming an increasingly important criterion for selection by major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on safety, purity, and operational parameters. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing production portfolios.
Q: What are the safety advantages of this Ce-Mn catalytic method over traditional peroxide oxidation?
A: Unlike traditional methods requiring excessive hydrogen peroxide which pose explosion risks during scale-up, this patent utilizes air or oxygen at low pressure (0.1-2MPa) with a stable solid catalyst, drastically improving operational safety.
Q: Does this process require expensive transition metal scavengers for purification?
A: No, the cerium-manganese composite oxide is a heterogeneous catalyst that can be removed via simple filtration, eliminating the need for costly and complex heavy metal scavenging steps often required with homogeneous copper or palladium catalysts.
Q: What is the substrate scope for this phenoxazinone synthesis method?
A: The method demonstrates broad applicability across various o-aminophenol derivatives, including methyl, fluoro, and chloro-substituted substrates, maintaining high yields above 80% for most tested derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-3H-Phenoxazin-3-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and sustainable synthetic routes for high-value pharmaceutical intermediates like 2-amino-3H-phenoxazin-3-one. Our technical team has thoroughly analyzed the potential of the cerium-manganese catalytic system described in CN113441135B and is fully prepared to implement this technology at commercial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to plant floor is seamless and efficient. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the low metal content and high chemical purity required for API synthesis. We are committed to delivering a reliable supply of this critical intermediate, leveraging our expertise in green chemistry to provide our partners with a competitive edge in their drug development programs.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with our technical procurement team, you can gain access to specific COA data and route feasibility assessments that demonstrate the tangible benefits of this advanced manufacturing process. Let us help you optimize your supply chain with a solution that balances cost, quality, and sustainability. Contact us today to discuss how we can support your project needs with our state-of-the-art production capabilities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
