Optimizing Prolindac Production: Advanced Intermediate Synthesis for Commercial Scale-up
The pharmaceutical landscape for oncology treatments continues to evolve with the development of targeted drug delivery systems, among which the aminomalonate platinum complex known as Prolindac (AP5346) represents a significant advancement in reducing sensory neurotoxicity associated with traditional oxaliplatin therapies. Patent CN101724018B introduces a critical innovation in the supply chain of this potent anticancer agent by detailing a novel preparation method for 2-(2-methacrylamide triglycyl) aminomalonate ester, a key intermediate that streamlines the overall manufacturing process. This technical breakthrough addresses long-standing inefficiencies in the synthesis of polymer-drug conjugates, offering a pathway that is not only chemically robust but also commercially viable for large-scale production. By re-engineering the synthetic route to focus on a pre-functionalized monomer, the technology enables manufacturers to bypass cumbersome multi-step polymer modifications, thereby enhancing the reliability of the reliable pharmaceutical intermediates supplier network. The implications of this patent extend beyond mere chemical curiosity, providing a tangible framework for cost reduction in pharmaceutical intermediates manufacturing while maintaining the stringent quality standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Prolindac and its associated polymeric carriers has been plagued by convoluted reaction sequences that introduce significant operational friction and economic inefficiency into the supply chain. Conventional literature and prior art, such as those disclosed in US7166733 and WO2005079396, typically describe a three-step reaction pathway starting from hydroxypropyl methacrylate (HPMA) to achieve the final platinum complex. This traditional methodology requires the initial polymerization of HPMA followed by sequential conjugation steps to attach the peptide linker and the aminomalonate moiety, a process that inherently suffers from cumulative yield losses at each stage. The complexity of modifying a pre-formed polymer often leads to heterogeneous product distributions, making purification difficult and resulting in lower overall throughput. Furthermore, the handling of high molecular weight intermediates in multiple reaction vessels increases the risk of batch-to-batch variability, which is a critical concern for commercial scale-up of complex pharmaceutical intermediates. These structural inefficiencies translate directly into higher production costs and extended lead times, creating bottlenecks that hinder the ability to meet global demand for this vital oncology medication.
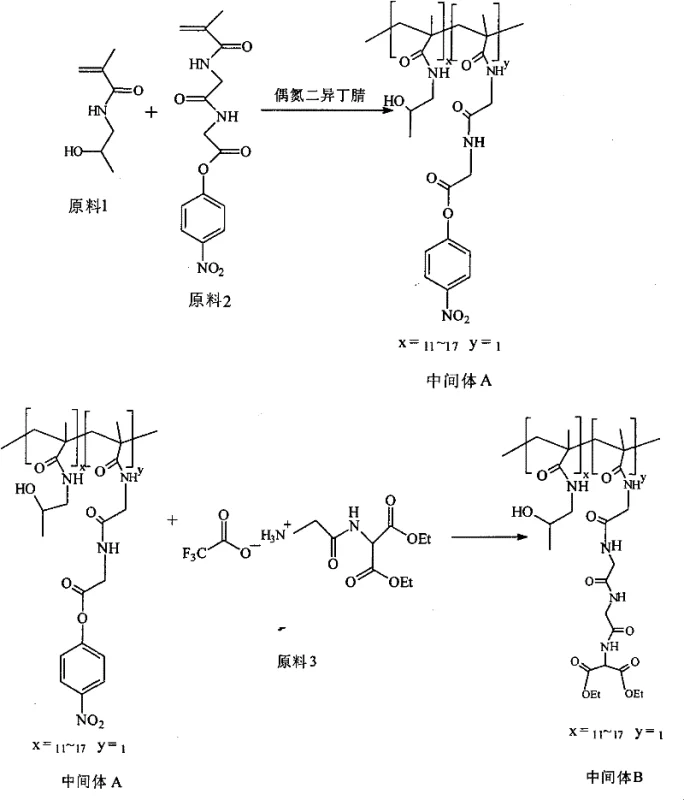
The Novel Approach
In stark contrast to the legacy methodologies, the technology outlined in CN101724018B proposes a paradigm shift by synthesizing the functional monomer, 2-(2-methacrylamide triglycyl) aminomalonate ester, prior to polymerization. This strategic inversion of the synthesis order allows for the rigorous purification of small molecule intermediates before they are committed to the polymer backbone, ensuring a much higher degree of structural uniformity in the final product. The new route effectively condenses the critical functionalization steps, allowing the target aminomalonate platinum complex to be prepared via a simplified two-step reaction sequence from the key monomer and HPMA. By utilizing specific protecting group strategies involving Fmoc or Boc chemistry combined with efficient coupling agents like DCC and HOBT, the process achieves superior control over the peptide bond formation. This refinement not only enhances the chemical purity of the intermediate but also drastically simplifies the downstream processing requirements. For procurement teams, this translates to a more predictable supply model where reducing lead time for high-purity pharmaceutical intermediates becomes a achievable reality through streamlined operations and reduced waste generation.
Mechanistic Insights into Peptide Coupling and Methacrylation
The core of this technological advancement lies in the precise execution of peptide coupling reactions followed by a targeted methacrylation step, which together construct the sophisticated architecture of the intermediate. The synthesis begins with the reaction of an aminomalonate ester, where the ester group R can vary from C1 to C5 alkyl chains, with a protected triglycine derivative such as Fmoc-glycylglycylglycine. This coupling is mediated by dicyclohexylcarbodiimide (DCC) and 1-hydroxybenzotriazole (HOBT), a combination chosen for its ability to activate the carboxylic acid while suppressing racemization, a critical factor in maintaining the biological activity of the peptide linker. The resulting protected intermediate is then subjected to deprotection conditions, utilizing either anhydrous piperidine for Fmoc removal or trifluoroacetic acid for Boc cleavage, to reveal the free amine necessary for the final functionalization. This deprotection step is meticulously controlled to prevent side reactions that could compromise the integrity of the malonate ester, ensuring that the reactive site remains available for the subsequent attachment of the methacryloyl group.
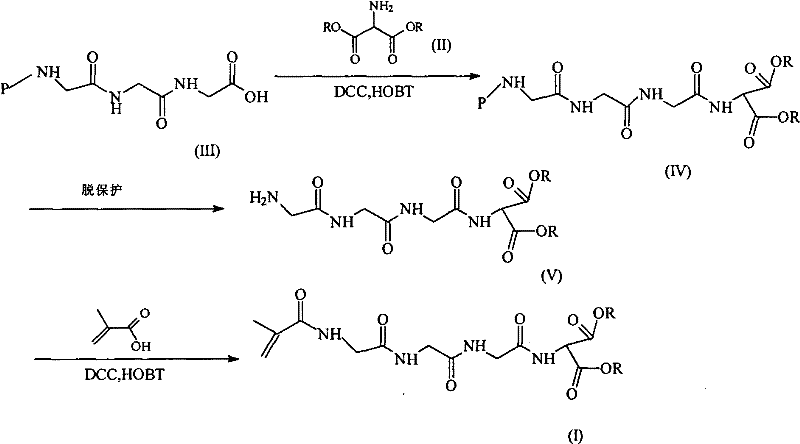
Following deprotection, the free amine undergoes a condensation reaction with 2-methacrylic acid, again employing DCC and HOBT to drive the formation of the amide bond that links the polymerizable methacrylate group to the peptide chain. This final structure, 2-(2-methacrylamide triglycyl) aminomalonate ester, is designed to be copolymerized with HPMA, creating a water-soluble polymer backbone that carries the platinum drug payload. The mechanistic elegance of this route is evident in its modularity; by isolating the synthesis of the functional monomer, manufacturers can implement rigorous quality control checks on the small molecule before polymerization, thereby minimizing the risk of incorporating defective units into the high-value polymer. This level of control is essential for managing the impurity profile, as the removal of unreacted starting materials or side products is far more efficient at the monomer stage than in the final polymeric drug substance. Consequently, this approach supports the production of high-purity pharmaceutical intermediates that meet the exacting standards of regulatory bodies worldwide.
How to Synthesize 2-(2-Methacrylamide Triglycyl) Aminomalonate Ester Efficiently
The practical implementation of this synthesis route involves a series of well-defined operational steps that balance reaction efficiency with ease of isolation. The process initiates with the mixing of aminomalonate ester and the protected peptide in a solvent system such as acetonitrile, followed by the controlled addition of coupling agents at low temperatures to manage exothermic activity. After the coupling reaction reaches completion, typically over a period of 10 to 30 hours at room temperature, the byproduct dicyclohexylurea is removed via filtration, and the product is precipitated using diethyl ether to achieve high purity solids. Subsequent deprotection and methacrylation steps follow similar protocols of reaction, workup, and purification, ensuring that each intermediate is isolated in a stable form suitable for storage or immediate use. Detailed standardized synthesis steps see the guide below.
- React protected triglycine with aminomalonate ester using DCC and HOBT coupling agents to form the protected intermediate.
- Perform deprotection using piperidine or trifluoroacetic acid to remove Fmoc or Boc groups, yielding the free amine.
- Condense the free amine with methacrylic acid using DCC/HOBT to finalize the 2-(2-methacrylamide triglycyl) aminomalonate ester structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers profound advantages that resonate deeply with the strategic goals of procurement managers and supply chain directors. The primary benefit stems from the significant reduction in synthetic complexity, which directly correlates to lower operational expenditures and a more resilient supply chain. By eliminating the need for multiple polymer modification steps, the process reduces the consumption of expensive reagents and solvents, leading to substantial cost savings in raw material procurement. Furthermore, the improved yield profile associated with the two-step approach means that less starting material is required to produce the same amount of final active intermediate, enhancing the overall economic efficiency of the manufacturing campaign. This efficiency is crucial for maintaining competitive pricing in the global market for oncology drugs, where cost pressures are increasingly intense.
- Cost Reduction in Manufacturing: The streamlined synthesis eliminates several unit operations that are traditionally resource-intensive, such as repeated polymer purification and characterization. By shifting the complexity to the small molecule stage, the process leverages standard chemical engineering practices that are inherently cheaper and faster than specialized polymer modifications. The removal of transition metal catalysts or complex purification columns in the later stages further contributes to a leaner cost structure, allowing manufacturers to allocate resources more effectively across their portfolio. This logical deduction of cost benefits ensures that the financial advantages are realized without compromising the quality or safety of the final therapeutic product.
- Enhanced Supply Chain Reliability: The use of readily available starting materials such as aminomalonate esters and protected amino acids ensures that the supply chain is not vulnerable to shortages of exotic or highly specialized reagents. The robustness of the reaction conditions, which operate at ambient or moderately elevated temperatures, reduces the risk of batch failures due to equipment malfunction or thermal runaway. This reliability translates into more consistent delivery schedules for downstream drug product manufacturers, mitigating the risk of production delays that can have cascading effects on patient access. A stable supply of high-quality intermediates is the backbone of a resilient pharmaceutical supply chain, and this technology provides exactly that foundation.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and reagents that are compatible with large-scale industrial reactors. The simplified workup procedures, involving filtration and precipitation rather than complex chromatography, reduce the generation of hazardous waste and lower the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate social responsibility profile of the manufacturer. The ability to scale from kilogram to tonne quantities without significant process re-engineering ensures that the technology can grow with market demand, providing a future-proof solution for long-term production needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, drawing directly from the patent specifications and industry best practices. These insights are intended to clarify the operational benefits and technical feasibility for stakeholders evaluating this route for potential adoption or partnership. Understanding these details is crucial for making informed decisions about integrating this intermediate into existing manufacturing workflows.
Q: How does this new synthesis route improve upon conventional methods for Prolindac intermediates?
A: The novel approach reduces the synthesis from three complex steps to a more direct two-step polymerization process, significantly lowering production costs and improving overall yield stability.
Q: What are the key purity specifications for this aminomalonate ester intermediate?
A: The process utilizes rigorous purification techniques including ether precipitation and filtration, ensuring high chemical purity suitable for subsequent platinum complexation without extensive downstream cleaning.
Q: Is this intermediate scalable for commercial pharmaceutical manufacturing?
A: Yes, the reaction conditions operate at manageable temperatures and utilize standard solvents like acetonitrile and ethyl acetate, facilitating easy scale-up from laboratory to industrial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Methacrylamide Triglycyl) Aminomalonate Ester Supplier
As the demand for advanced oncology therapeutics continues to rise, the need for a partner who can deliver complex intermediates with precision and reliability has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to support the global pharmaceutical community. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which ensure that every batch of 2-(2-methacrylamide triglycyl) aminomalonate ester meets the highest standards of chemical integrity. We understand the critical nature of supply chain continuity in drug development and are equipped to handle the complexities of polymer-drug conjugate intermediates with the expertise they demand.
We invite you to engage with our technical procurement team to discuss how our capabilities can align with your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this optimized synthesis route. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate our performance against your internal benchmarks. Together, we can accelerate the development of life-saving medications and ensure that patients around the world have access to the treatments they need.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
