Advanced Synthesis of 2-Aminopyrimidine-5-Carboxylate for Commercial API Production
Advanced Synthesis of 2-Aminopyrimidine-5-Carboxylate for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical heterocyclic intermediates, particularly those serving as key building blocks for kinase inhibitors like Copanlisib. A significant breakthrough in this domain is documented in patent CN110655491B, which outlines a novel preparation method for 2-aminopyrimidine-5-formic ether derivatives. This technology addresses long-standing challenges in yield optimization and operational safety, offering a compelling alternative to traditional methodologies. By leveraging a sequential substitution and addition reaction strategy starting from 2-methyl acrylate, the process achieves high conversion rates while maintaining a benign environmental profile. For R&D directors and procurement specialists, understanding the mechanistic advantages of this patent is crucial for evaluating potential supply chain partnerships. The structural integrity of the target molecule, as depicted below, is preserved through a controlled cyclization process that minimizes byproduct formation.
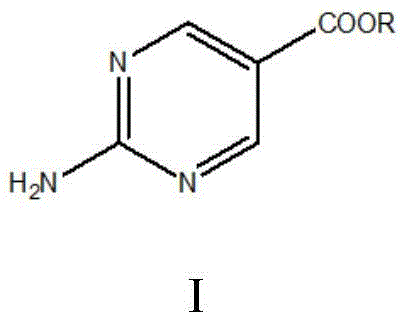
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-aminopyrimidine-5-carboxylates has relied heavily on routes involving 3,3-dimethoxypropionic acid methyl ester as a primary starting material. As illustrated in prior art schemes, these conventional pathways often necessitate the use of strong bases such as sodium hydride in polar aprotic solvents like DMF. The reliance on sodium hydride introduces severe safety hazards, including the risk of hydrogen gas evolution and potential explosion upon exposure to moisture or improper handling. Furthermore, these legacy methods typically suffer from moderate yields, often hovering around 50%, which drastically impacts the overall cost of goods sold. The post-treatment procedures are equally burdensome, generating substantial volumes of saline wastewater that require expensive disposal protocols. From a supply chain perspective, the dependency on specialized, moisture-sensitive reagents creates bottlenecks that can delay production timelines and compromise batch consistency.
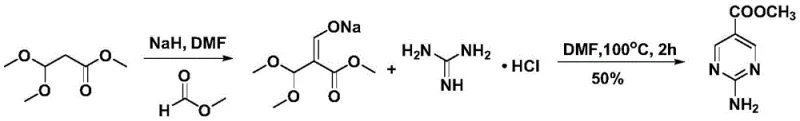
The Novel Approach
In stark contrast, the methodology disclosed in CN110655491B revolutionizes the production landscape by utilizing 2-methyl acrylate, a commodity chemical that is both inexpensive and readily available on a global scale. This innovative route bypasses the need for hazardous strong bases, instead employing a controlled halogenation strategy that proceeds under much milder conditions. The process is designed to be operationally simple, reducing the technical barrier for entry and allowing for smoother technology transfer between facilities. By eliminating the complex protection and deprotection steps associated with dimethoxy intermediates, the overall process flow is drastically simplified. This simplification not only enhances the safety profile for plant operators but also significantly reduces the consumption of organic solvents. For procurement managers, this translates to a more resilient supply chain where raw material availability is less of a constraint, ensuring continuous production capabilities even during market fluctuations.
Mechanistic Insights into Halogenation-Driven Cyclization
The core innovation of this patent lies in its sophisticated two-step reaction sequence that transforms a simple acrylate into a complex pyrimidine ring system. The first stage involves a radical substitution reaction where 2-methacrylate reacts with a halogenating agent, facilitated by a catalyst such as benzoyl peroxide. This step selectively introduces halogen atoms at the allylic position, creating a reactive intermediate primed for further functionalization. Subsequently, an addition reaction occurs under the influence of a Lewis acid or protonic acid catalyst, leading to the formation of a 2,3,3-trihalo-2-halomethylpropionate species. This specific halogenation pattern is critical, as it activates the carbon backbone for the subsequent nucleophilic attack by the guanidine salt. The precision of this halogen introduction ensures that the reactive sites are perfectly aligned for cyclization, minimizing the formation of regio-isomers that often plague less controlled synthetic routes.
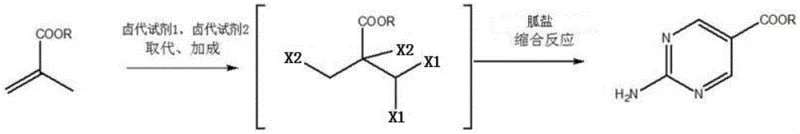
Following the formation of the tetrahalo intermediate, the process moves to a condensation phase where the pyrimidine ring is constructed. This cyclization is driven by the reaction between the halogenated propionate and a guanidine salt in the presence of a mild base. The mechanism proceeds through a nucleophilic substitution where the nitrogen atoms of the guanidine displace the halogen leaving groups, closing the ring to form the stable pyrimidine core. A key advantage of this mechanistic pathway is the inherent impurity control; the high selectivity of the halogenation step means that fewer side products are generated during the cyclization. This results in a crude product with exceptionally high purity, often exceeding 99% as confirmed by liquid phase analysis. For quality control teams, this means reduced burden on downstream purification processes, allowing for more efficient resource allocation and faster release times for the final API intermediate.
How to Synthesize 2-Aminopyrimidine-5-Carboxylate Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control and catalyst loading during the halogenation phase. The patent details a 'one-pot' strategy that allows for the sequential addition of reagents without the need to isolate the unstable trihalo intermediate, thereby reducing material handling risks. Operators must ensure that the substitution reaction catalyst is effectively neutralized before proceeding to the addition step to prevent competing reactions that could lower yield. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are critical for reproducing the high yields reported in the patent examples. Adhering to these protocols ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved with consistent quality and safety.
- Perform radical substitution and addition reactions on 2-methacrylate using halogenating agents to form 2,3,3-trihalo-2-halomethylpropionate.
- Condense the resulting trihalo intermediate with a guanidine salt in the presence of a base and solvent to cyclize the pyrimidine ring.
- Isolate the final high-purity ester product through solvent recovery and filtration, ensuring minimal waste generation.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the adoption of this patented technology offers transformative benefits that extend beyond mere chemical efficiency. The shift from expensive, specialized starting materials to commodity acrylates fundamentally alters the cost structure of the manufacturing process. By removing the dependency on hazardous reagents like sodium hydride, facilities can reduce their insurance premiums and safety compliance costs, leading to substantial cost savings in API manufacturing. Furthermore, the simplified workflow reduces the total processing time, allowing for higher throughput within existing infrastructure. This efficiency gain is crucial for meeting the demanding delivery schedules of global pharmaceutical clients who require just-in-time delivery of critical intermediates. The robustness of the process also ensures supply continuity, mitigating the risks associated with raw material shortages that often affect more fragile synthetic routes.
- Cost Reduction in Manufacturing: The elimination of expensive reagents and the use of cheap, bulk-available raw materials like 2-methyl acrylate drastically lowers the direct material costs. Additionally, the high yield and purity reduce the need for extensive chromatographic purification, which is often the most expensive step in fine chemical production. The reduction in solvent consumption and waste generation further contributes to lower operational expenditures, making the final product highly competitive in the global market.
- Enhanced Supply Chain Reliability: Sourcing 2-methyl acrylate is significantly more reliable than sourcing specialized dimethoxy esters, as it is produced by major chemical companies worldwide. This abundance ensures that production is not halted due to raw material scarcity. The operational safety improvements also mean that unplanned shutdowns due to safety incidents are less likely, providing a more predictable and stable supply of high-purity API intermediates to downstream customers.
- Scalability and Environmental Compliance: The process generates minimal wastewater and avoids the use of heavy metals or toxic cyanides, aligning perfectly with modern environmental regulations. This compliance simplifies the permitting process for new production lines and reduces the cost of waste treatment. The mild reaction conditions are easily transferable from pilot scale to multi-ton production, facilitating the commercial scale-up of complex pharmaceutical intermediates without the need for specialized high-pressure or cryogenic equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing clarity for stakeholders evaluating this route. Understanding these details is essential for making informed decisions about process adoption and supplier qualification. The responses highlight the practical advantages of the method in a real-world industrial context, focusing on safety, quality, and scalability.
Q: What are the safety advantages of this new synthesis route compared to conventional methods?
A: Unlike conventional methods requiring hazardous sodium hydride, this patent utilizes mild halogenation conditions, significantly reducing explosion risks and operational hazards.
Q: How does this process impact the purity profile of the final API intermediate?
A: The specific catalytic control and one-pot design minimize side reactions, resulting in liquid phase purity exceeding 99% and a simplified impurity spectrum.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the use of cheap, readily available raw materials and the elimination of complex post-treatment steps make it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminopyrimidine-5-Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving medications. Our CDMO expertise allows us to adapt and optimize complex pathways like the one described in CN110655491B to meet the rigorous demands of the pharmaceutical industry. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-aminopyrimidine-5-carboxylate meets the highest quality standards required for API synthesis. We are committed to being a partner that not only supplies chemicals but also provides technical solutions that enhance your overall production efficiency.
We invite you to discuss how our capabilities can support your specific project requirements and help you achieve your cost and timeline goals. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. We encourage you to contact us to request specific COA data and route feasibility assessments, ensuring that you have all the necessary information to move your project forward with confidence. Let us help you secure a reliable supply of high-quality intermediates for your next breakthrough therapy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
