Advanced Synthesis of Haloallyl Furfuryl Alcohol for Commercial Scale-Up
The chemical landscape for producing high-value intermediates is constantly evolving, driven by the need for efficiency and purity in sectors ranging from pharmaceuticals to electronic materials. Patent CN101108837A introduces a transformative methodology for the preparation of halogenated allyl furfuryl alcohol, a critical building block in modern organic synthesis. This technology addresses long-standing inefficiencies in the production of furan-based derivatives by integrating formylation and allylation steps into a cohesive workflow. For R&D Directors and Procurement Managers alike, understanding this process is vital for optimizing supply chains and reducing the cost of goods sold. The core innovation lies in the manipulation of reaction intermediates, specifically avoiding the thermal degradation that plagues traditional isolation techniques. By maintaining the intermediate furfural derivatives in an organic solution, the process ensures maximum material throughput and minimizes waste generation. This report analyzes the technical merits and commercial implications of this patented route, providing a roadmap for stakeholders seeking a reliable haloallyl furfuryl alcohol supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of haloallyl furfuryl alcohol has been hindered by the instability of key intermediates, particularly furfural derivatives. Conventional methods, as disclosed in prior art such as JP-A-02-124882 and JP-A-57-91982, typically require the isolation of furfural intermediates through distillation. This unit operation is energetically expensive and chemically hazardous for thermally sensitive compounds. During the distillation process, a significant portion of the furfural derivative decomposes due to heat exposure, leading to a substantial reduction in the overall recovery rate based on the starting furan material. Furthermore, the residual material left in the distillation pot often contains unreacted precursors that are difficult to recover without further processing, thereby increasing the production load and operational complexity. These inefficiencies translate directly into higher manufacturing costs and longer lead times, creating bottlenecks for companies aiming for cost reduction in pharmaceutical intermediates manufacturing. The need for multiple isolation steps also increases the risk of contamination and variability in product quality, which is unacceptable for high-purity applications.
The Novel Approach
The methodology described in CN101108837A circumvents these thermal stability issues by employing a telescoped reaction strategy. Instead of isolating the furfural derivative, the process generates it in situ within an organic solvent, typically toluene, and immediately subjects it to the subsequent allylation reaction. This approach eliminates the need for high-temperature distillation of the intermediate, thereby preserving the chemical integrity of the molecule and maximizing yield. The reaction mixture from the initial Vilsmeier-Haack formylation is hydrolyzed to obtain a solution of the furfural derivative, which is then directly mixed with zinc and a dihalogenated compound. This seamless transition between reaction stages not only simplifies the operational workflow but also significantly enhances the overall yield based on the starting furan or its derivatives. By suppressing the loss of intermediates, this novel approach offers a robust pathway for the commercial scale-up of complex fine chemicals, ensuring that manufacturers can meet demand without compromising on efficiency or product consistency.
Mechanistic Insights into Vilsmeier-Haack Formylation and Zinc-Mediated Allylation
The chemical foundation of this process rests on two well-established but strategically combined reactions: the Vilsmeier-Haack formylation and a Barbier-type allylation. Initially, a furan derivative, such as 2-methylfuran, reacts with a chlorinating agent like phosgene and a formamide derivative such as N,N-dimethylformamide (DMF) in an organic solvent. This interaction generates a reactive Vilsmeier complex, which subsequently attacks the electron-rich furan ring to form an immonium salt intermediate. The precise control of reaction temperature, typically between 30 to 80°C, is crucial to suppress the formation of by-products while ensuring complete conversion. Following this, the reaction mixture undergoes hydrolysis, often using an aqueous alkaline solution, to convert the immonium salt into the corresponding furfural derivative. This step is critical as it prepares the electrophile for the subsequent nucleophilic attack without exposing it to harsh isolation conditions.
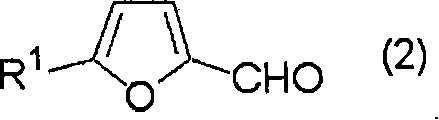
Once the furfural derivative is generated in the organic phase, the process moves to the allylation stage. The solution containing the furfural derivative is mixed with zinc powder and a dihalogenated compound, such as 2,3-dichloro-1-propene. The zinc acts as a reducing agent, facilitating the formation of an organozinc species that attacks the carbonyl group of the furfural. This Barbier-type reaction is highly advantageous as it can be performed in the presence of water or protic solvents, unlike Grignard reactions which require strictly anhydrous conditions. The use of acetic acid as an additive can further promote the reaction smoothness. The result is the formation of the target haloallyl furfuryl alcohol with high stereochemical fidelity and purity. This mechanistic understanding is essential for R&D teams aiming to replicate or optimize the process for specific high-purity agrochemical intermediates or electronic material precursors.
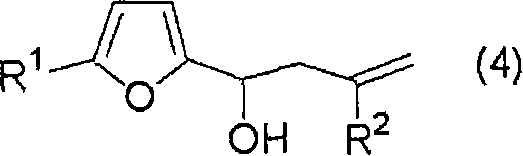
How to Synthesize Haloallyl Furfuryl Alcohol Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and phase separation techniques to ensure optimal performance. The process begins with the co-feeding of furan derivatives, chlorinating agents, and formamides into a reactor containing toluene, which helps suppress by-product formation through dilution and temperature control. After the formation of the immonium salt, hydrolysis is conducted, and the organic phase containing the furfural derivative is separated. Crucially, this organic solution is not distilled but is instead directly utilized in the next reactor or the same vessel after adjustment. Zinc powder and the dihalogenated compound are added, often with a slight excess to drive the reaction to completion. The detailed standardized synthesis steps see the guide below.
- React furan derivatives with chlorinating agents and formamide in toluene to form an immonium salt intermediate.
- Hydrolyze the reaction mixture with aqueous alkali to generate the furfural derivative solution in situ.
- Mix the furfural solution directly with zinc powder and dihalogenated compounds to yield the final haloallyl furfuryl alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the technical improvements in this patent translate directly into tangible business benefits. The elimination of the distillation step for the intermediate furfural derivative significantly reduces energy consumption and equipment wear, leading to substantial cost savings in the manufacturing process. By avoiding thermal decomposition, the overall yield is drastically improved, meaning less raw material is required to produce the same amount of final product. This efficiency gain is critical for reducing lead time for high-purity electronic materials where supply consistency is paramount. Furthermore, the use of common solvents like toluene and readily available reagents like zinc powder enhances supply chain reliability, as there is no dependence on exotic or hard-to-source catalysts. The simplified workflow also reduces the operational footprint, allowing for faster batch turnover and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of the intermediate isolation step. Distillation is an energy-intensive unit operation that requires significant capital investment in equipment and ongoing utility costs. By telescoping the formylation and allylation steps, the process eliminates this energy burden entirely. Additionally, the higher overall yield means that the cost per kilogram of the final haloallyl furfuryl alcohol is significantly lower. The reduction in waste generation also lowers disposal costs, contributing to a leaner and more economical production model. This logical deduction of cost benefits makes the technology highly attractive for large-scale production environments.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the chemical route. The reagents involved, such as 2-methylfuran, phosgene, and zinc, are commodity chemicals with stable global supply networks. This reduces the risk of production stoppages due to raw material shortages. Moreover, the process tolerance to minor variations in reaction conditions ensures consistent output quality, which is vital for maintaining long-term contracts with downstream pharmaceutical or agrochemical manufacturers. The ability to produce high-purity intermediates without complex purification trains means that inventory can be turned over more quickly, enhancing the overall agility of the supply chain.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is straightforward due to the use of standard reaction engineering principles. The exothermic nature of the reactions is manageable with conventional cooling systems, and the phase separation steps are well-understood in industrial settings. From an environmental perspective, the reduction in solvent usage and energy consumption aligns with green chemistry principles. The minimization of waste streams simplifies effluent treatment, ensuring compliance with increasingly stringent environmental regulations. This scalability ensures that the technology can meet the growing demand for complex polymer additives and specialty chemicals without compromising on sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this technology for their specific production needs. The focus is on clarity regarding yield improvements, operational safety, and product quality assurance.
Q: Why is distillation avoided in this synthesis route?
A: Distillation of thermally unstable furfural derivatives leads to decomposition and significant yield loss. This process keeps the intermediate in solution.
Q: What is the overall yield advantage of this method?
A: By eliminating isolation steps, the overall yield based on starting furan derivatives is significantly improved compared to conventional multi-step isolation methods.
Q: Is this process scalable for industrial production?
A: Yes, the use of common solvents like toluene and standard reagents like zinc powder facilitates easy commercial scale-up of complex fine chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Haloallyl Furfuryl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthesis routes play in the competitiveness of the global fine chemical market. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN101108837A can be successfully translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, providing our partners with the confidence they need to integrate our intermediates into their own complex synthesis chains. Our capability to handle hazardous reagents safely and efficiently makes us an ideal partner for the production of sensitive furan derivatives.
We invite you to collaborate with us to optimize your supply chain and achieve significant operational efficiencies. By leveraging our technical expertise, you can access a Customized Cost-Saving Analysis tailored to your specific production requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can drive innovation and value creation in the pharmaceutical and agrochemical sectors, ensuring a stable and cost-effective supply of high-quality intermediates for your most critical applications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
