Advanced Biomimetic Catalysis for Commercial Scale O-Bromoacetophenone Production
Introduction to Patent CN101747168B Technology
The chemical industry is constantly seeking more efficient and environmentally benign pathways for synthesizing critical aromatic ketones, and Patent CN101747168B represents a significant breakthrough in this domain. This intellectual property details a novel method for preparing o-bromoacetophenone through the biomimetic catalytic oxidation of o-bromoethylbenzene using molecular oxygen. Unlike traditional processes that rely on harsh conditions and toxic reagents, this invention utilizes highly active metalloporphyrin catalysts to achieve superior conversion rates under mild, solvent-free conditions. The technology addresses long-standing challenges in fine chemical synthesis, specifically targeting the reduction of energy consumption and the elimination of hazardous waste streams. For R&D directors and process engineers, this patent offers a compelling alternative to legacy oxidation methods, promising not only higher purity profiles but also a fundamentally safer operational framework. By leveraging the unique electronic properties of iron, manganese, and cobalt porphyrins, the process mimics natural enzymatic oxidation, resulting in exceptional selectivity that minimizes downstream purification burdens.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of o-bromoacetophenone has been plagued by significant technical and economic inefficiencies inherent to conventional oxidation technologies. Traditional methods, such as the Friedel-Crafts acylation or chemical oxidations using stoichiometric oxidants like sodium periodate or chloramine-T, generate substantial amounts of toxic waste and require complex separation procedures. Furthermore, prior art involving molecular oxygen oxidation often necessitates high-pressure reactors operating at elevated temperatures, such as 140°C and 0.8MPa, which introduces severe safety risks and exponentially increases capital expenditure for pressure-rated equipment. The use of solvents like benzene or chlorobenzene in older protocols further complicates the regulatory landscape due to their carcinogenic nature and the energy-intensive requirements for solvent recovery and recycling. Additionally, conventional cobalt-based catalyst systems typically require high loadings, often around 400ppm, leading to difficult product purification and potential heavy metal contamination in the final API intermediate. These cumulative factors result in a manufacturing process that is not only costly but also environmentally unsustainable and operationally hazardous.
The Novel Approach
In stark contrast, the methodology disclosed in CN101747168B revolutionizes the synthesis landscape by employing a biomimetic catalytic system that operates under atmospheric pressure and solvent-free conditions. This innovative approach utilizes extremely low concentrations of metalloporphyrin catalysts, ranging from just 1 to 30ppm, to drive the oxidation of o-bromoethylbenzene with molecular oxygen. The process features a unique two-stage temperature profile, initiating rapidly at 140-170°C to overcome induction periods and then sustaining the reaction at a milder 80-120°C. This strategic thermal management drastically shortens reaction times while maintaining high selectivity, effectively eliminating the long induction phases seen in previous heterogeneous systems. By removing the need for organic solvents and high-pressure vessels, the novel approach significantly lowers both the operational complexity and the safety risks associated with large-scale production. The result is a streamlined workflow that delivers o-bromoacetophenone with conversion rates reaching up to 95.2% and selectivity exceeding 91%, far surpassing the performance metrics of legacy technologies.
Mechanistic Insights into Metalloporphyrin-Catalyzed Oxidation
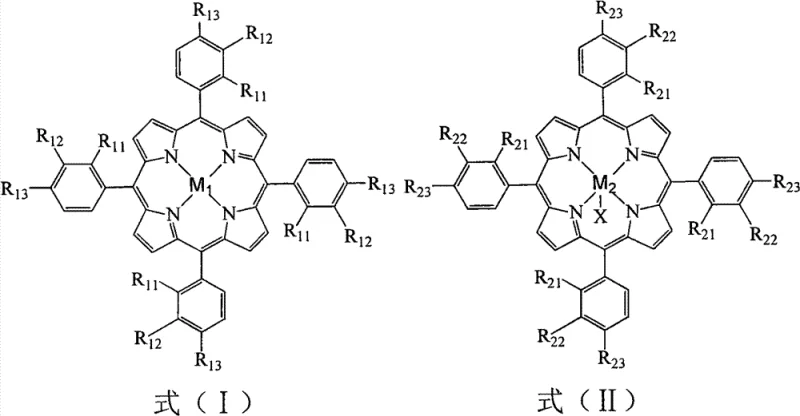
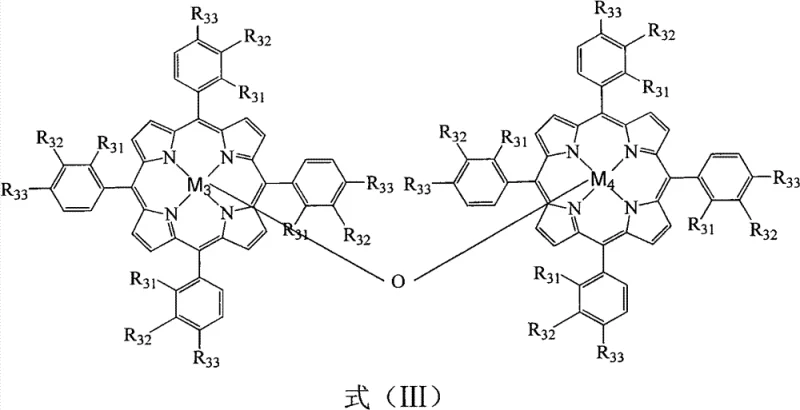
The core of this technological advancement lies in the sophisticated design of the metalloporphyrin catalysts, which function as artificial enzymes to activate molecular oxygen with high precision. As illustrated in the structural diagrams, the catalysts include mononuclear species defined by Formula (I) and (II), as well as mu-oxo binuclear complexes represented by Formula (III). These structures feature central metal ions such as iron, manganese, cobalt, or copper coordinated within a porphyrin macrocycle, often substituted with electron-withdrawing or electron-donating groups like halogens or alkoxy groups to tune their redox potentials. The mechanism involves the formation of high-valent metal-oxo intermediates that selectively abstract hydrogen atoms from the benzylic position of o-bromoethylbenzene. This biomimetic pathway ensures that the oxidation proceeds directly to the ketone stage with minimal over-oxidation to carboxylic acids or other by-products. The synergy between different metal centers, particularly when using combinations of iron or manganese porphyrins with cobalt porphyrins, creates a cooperative catalytic effect that enhances the turnover frequency and stability of the active species throughout the reaction cycle.
From an impurity control perspective, the homogeneous nature of this catalytic system plays a pivotal role in ensuring product quality. Unlike supported or polymerized catalysts which suffer from mass transfer limitations and heterogeneous active sites, these soluble metalloporphyrins provide uniform access to the substrate, leading to a consistent reaction environment. This uniformity suppresses the formation of radical-induced side products that typically arise in non-selective free-radical oxidations. Furthermore, the ability to operate at lower temperatures (100-110°C) kinetically favors the formation of the desired ketone over thermal degradation products. The absence of solvent also removes a major source of potential impurities and side reactions, simplifying the crude reaction profile. Consequently, the downstream purification process, primarily involving reduced pressure distillation, becomes highly efficient, yielding o-bromoacetophenone with purity levels consistently above 99%, which is critical for meeting the stringent specifications required in pharmaceutical intermediate supply chains.
How to Synthesize O-Bromoacetophenone Efficiently
Implementing this synthesis route requires precise control over catalyst loading and thermal parameters to maximize the benefits of the biomimetic system. The process begins by charging o-bromoethylbenzene into a reactor followed by the addition of the selected metalloporphyrin catalyst combination. Oxygen is then introduced at a controlled flow rate, typically between 10 to 60mL/min, to maintain an optimal concentration of the oxidant without creating safety hazards. The reaction is initiated at a higher temperature range of 140-170°C to rapidly activate the catalyst and consume any induction period, after which the temperature is lowered to 80-120°C for the main conversion phase lasting 6 to 16 hours. This protocol ensures that the reaction proceeds with maximum efficiency and safety, leveraging the high activity of the metalloporphyrin complexes to achieve near-quantitative conversion.
- Load o-bromoethylbenzene into a reactor and add 1-30ppm of mononuclear or mu-oxo binuclear metalloporphyrin catalyst.
- Initiate the reaction at 140-170°C with oxygen flow, then maintain at 80-120°C for 6-16 hours.
- Perform reduced pressure distillation on the reaction mixture to isolate high-purity o-bromoacetophenone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this biomimetic catalytic technology translates into tangible strategic advantages regarding cost structure and supply reliability. The most immediate impact is observed in the drastic reduction of raw material and utility costs driven by the elimination of solvents and the minimization of catalyst usage. By operating without solvents, the process removes the entire unit operation of solvent recovery and distillation, which is traditionally one of the most energy-intensive steps in fine chemical manufacturing. Additionally, the catalyst loading is reduced from hundreds of ppm in conventional methods to single-digit or low double-digit ppm levels, which not only lowers the direct cost of the catalyst but also simplifies the removal of metal residues from the final product. These efficiencies collectively contribute to a significantly leaner cost base, allowing for more competitive pricing in the global market for pharmaceutical intermediates while maintaining healthy margins.
- Cost Reduction in Manufacturing: The economic benefits of this process are profound, primarily stemming from the simplified operational workflow and reduced energy intensity. The elimination of high-pressure equipment requirements means that standard glass-lined or stainless steel reactors can be utilized, avoiding the massive capital investment associated with autoclaves rated for high-pressure oxygen service. Furthermore, the solvent-free nature of the reaction removes the need for purchasing, storing, and disposing of large volumes of organic solvents, which are subject to volatile pricing and strict environmental regulations. The low catalyst loading also means that the cost contribution of the precious metal or complex ligand systems is negligible on a per-kilogram basis. These factors combine to create a manufacturing process that is inherently more cost-effective, providing a buffer against raw material price fluctuations and enabling substantial cost savings in the production of high-purity o-bromoacetophenone.
- Enhanced Supply Chain Reliability: Supply chain continuity is greatly improved by the robustness and simplicity of this synthetic route. Traditional methods that rely on hazardous solvents or high-pressure conditions are prone to unplanned shutdowns due to safety incidents or regulatory inspections. In contrast, the atmospheric pressure and solvent-free conditions of this biomimetic process significantly lower the operational risk profile, ensuring more consistent uptime and production throughput. The use of molecular oxygen as the sole oxidant eliminates dependence on specialized chemical oxidants like sodium periodate or chloramine-T, which can face supply constraints or quality variability. This self-sufficiency in reagents, combined with the ease of scaling the reaction from laboratory to industrial volumes, ensures that suppliers can meet demanding delivery schedules reliably. For buyers, this translates to reduced lead times and a more secure supply of critical intermediates for their own API synthesis campaigns.
- Scalability and Environmental Compliance: Scaling this technology to commercial production is straightforward due to the absence of complex engineering constraints associated with high-pressure or cryogenic operations. The reaction exotherm is manageable through the controlled oxygen feed and the two-stage temperature profile, making it suitable for large-scale batch or continuous processing. From an environmental compliance standpoint, the process is exemplary, generating minimal waste as there are no solvent effluents to treat and the catalyst residues are biodegradable or easily removed. This aligns perfectly with the increasing global pressure on chemical manufacturers to adopt green chemistry principles and reduce their carbon footprint. By minimizing the E-factor (mass of waste per mass of product), companies utilizing this technology can achieve better sustainability ratings and avoid the escalating costs associated with waste disposal and environmental remediation, future-proofing their supply chains against tightening regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biomimetic oxidation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines or for procurement specialists assessing the quality implications of the new method. These answers reflect the proven capabilities of the metalloporphyrin catalyst system in delivering high-quality intermediates efficiently.
Q: What are the primary advantages of the metalloporphyrin catalyst system over traditional methods?
A: The metalloporphyrin system operates at atmospheric pressure and lower temperatures (100-110°C) compared to traditional high-pressure methods, significantly enhancing safety and reducing energy consumption while achieving conversion rates up to 95.2%.
Q: Is solvent recovery required in this biomimetic oxidation process?
A: No, the process is designed to be solvent-free. This eliminates the need for solvent recovery systems, reduces waste generation, and lowers overall operational costs associated with solvent handling and disposal.
Q: How does the catalyst loading compare to conventional cobalt catalyst systems?
A: This method requires only 1-30ppm of catalyst, which is drastically lower than the 400ppm typically needed for conventional cobalt complexes, thereby minimizing metal residue and purification costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Bromoacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN101747168B for optimizing the production of essential pharmaceutical intermediates. Our team of expert chemists and process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of biomimetic catalysis are fully realized in an industrial setting. We are committed to delivering high-purity o-bromoacetophenone that meets stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest industry standards. Our capability to adapt and implement such cutting-edge solvent-free and atmospheric pressure processes allows us to offer a product that is not only chemically superior but also produced with a significantly reduced environmental impact.
We invite global partners to collaborate with us to optimize their supply chains and reduce manufacturing costs through the adoption of this efficient synthesis route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out for specific COA data and route feasibility assessments to understand how our advanced manufacturing capabilities can support your long-term strategic goals. Let us help you secure a reliable supply of high-quality intermediates while driving sustainability and efficiency in your operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
