Advanced Room-Temperature Hydrolysis for Commercial Scale Scutellarein Production
Advanced Room-Temperature Hydrolysis for Commercial Scale Scutellarein Production
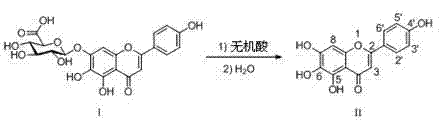
The pharmaceutical industry continuously seeks efficient pathways to transform low-bioavailability natural extracts into potent therapeutic agents, and the conversion of scutellarin to scutellarein represents a critical value-addition step in cardiovascular and cerebrovascular drug development. A groundbreaking approach detailed in patent CN103102337A introduces a novel preparation method that utilizes direct acid hydrolysis under remarkably mild conditions to achieve this transformation with exceptional efficiency. This technology leverages concentrated inorganic acids as the sole reaction medium at room temperature, effectively cleaving the glucuronic acid moiety from the flavonoid backbone without the need for energy-intensive heating or hazardous organic solvents. By shifting the paradigm from traditional reflux protocols to a rapid, ambient-temperature process, this innovation addresses long-standing challenges regarding reaction time, energy consumption, and environmental safety in the manufacturing of high-purity pharmaceutical intermediates. For global procurement teams and R&D directors, understanding the mechanistic superiority of this route is essential for securing a reliable scutellarein supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of scutellarein from scutellarin has been plagued by inefficient processes that rely heavily on harsh reaction conditions and extensive solvent usage. Traditional methodologies often involve heating the reaction mixture to reflux temperatures for extended periods, ranging from several hours to nearly two days, which not only consumes substantial energy but also increases the risk of thermal degradation of the sensitive flavonoid structure. Furthermore, many existing protocols necessitate the use of organic solvents to facilitate the hydrolysis or subsequent extraction steps, introducing significant complexities in terms of solvent recovery, waste disposal, and residual solvent control in the final active pharmaceutical ingredient. These legacy methods frequently suffer from inconsistent yields and purity profiles, as the prolonged exposure to heat and aggressive chemical environments can generate difficult-to-remove impurities that compromise the therapeutic efficacy of the final product. Consequently, manufacturers relying on these outdated techniques face elevated production costs and supply chain vulnerabilities that hinder their ability to compete in the fast-paced market for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy systems, the innovative method described in the patent data employs a streamlined direct acid hydrolysis strategy that operates entirely at room temperature, thereby eliminating the need for external heating sources. By carefully controlling the concentration of scutellarin within a specific range in concentrated inorganic acid solutions, such as sulfuric, hydrochloric, or phosphoric acid, the process ensures rapid and complete cleavage of the glycosidic bond within minutes rather than hours. The absence of organic solvents during the primary reaction phase drastically simplifies the downstream processing workflow, as the product can be directly precipitated by diluting the reaction mixture with water, avoiding complex extraction and distillation units. This approach not only accelerates the overall production cycle but also inherently enhances the safety profile of the manufacturing facility by removing volatile organic compounds from the immediate process environment. For stakeholders focused on cost reduction in pharmaceutical intermediates manufacturing, this transition represents a fundamental shift towards leaner, greener, and more economically viable production models.
Mechanistic Insights into Acid-Catalyzed Glycosidic Bond Cleavage
The core chemical transformation driving this process is the acid-catalyzed hydrolysis of the glycosidic linkage connecting the glucuronic acid group to the flavonoid aglycone skeleton of scutellarin. Under the influence of strong proton donors provided by concentrated inorganic acids, the oxygen atom in the glycosidic bond becomes protonated, weakening the bond and facilitating its heterolytic cleavage to release the glucose derivative and the free hydroxyl group on the scutellarein molecule. The precise control of acid concentration and the specific ratio of water addition are critical parameters that govern the kinetics of this reaction, ensuring that the hydrolysis proceeds to completion without inducing unwanted side reactions on the phenolic rings of the flavonoid structure. Operating at room temperature is particularly advantageous for preserving the integrity of the polyphenolic system, as elevated temperatures could otherwise promote oxidation or rearrangement reactions that would degrade the quality of the target molecule. This mechanistic precision allows for the generation of scutellarein with a clean impurity profile, which is paramount for meeting the stringent regulatory requirements of modern drug substance manufacturing.
Furthermore, the impurity control mechanism inherent in this low-temperature protocol significantly reduces the formation of polymeric byproducts or decomposition fragments that are commonly observed in thermal hydrolysis processes. The rapid precipitation step, triggered by pouring the acid reaction mixture into a larger volume of water, serves as an effective purification stage where the less soluble scutellarein aggregates and separates from the aqueous acid phase containing the hydrophilic sugar byproducts. This physical separation is highly efficient and minimizes the loss of product to the mother liquor, contributing to the high overall yields reported in the experimental data. For R&D teams evaluating process robustness, the ability to tune the precipitation conditions by adjusting water temperature and stirring speed offers an additional layer of control over particle size distribution and crystal morphology, which are critical attributes for downstream formulation and bioavailability. Understanding these mechanistic nuances is vital for any organization aiming to establish a commercial scale-up of complex pharmaceutical intermediates with consistent quality attributes.
How to Synthesize Scutellarein Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters regarding acid concentration, mixing ratios, and precipitation dynamics to ensure maximum yield and purity. The process begins with the dissolution of the raw material in concentrated acid, followed by a controlled quenching step with water that must be executed with precision to avoid localized overheating or incomplete precipitation. Detailed standard operating procedures for this transformation are critical for maintaining batch-to-batch consistency, especially when scaling from laboratory benchtop quantities to multi-ton industrial production campaigns. The following guide outlines the critical operational phases derived from the patent specifications to assist technical teams in replicating this high-efficiency pathway.
- Dissolve scutellarin in concentrated inorganic acid (such as sulfuric, hydrochloric, or phosphoric acid) to achieve a concentration between 0.03g/mL and 0.08g/mL, stirring at room temperature for 5 to 20 minutes.
- Add distilled water dropwise to the acid solution while stirring, maintaining a water-to-scutellarin mass ratio between 1: 1 and 15:1, then allow the mixture to stand and cool.
- Pour the cooled reaction mixture into stirred water (5°C to 60°C) at a volume ratio of 1: 1 to 1:10 to precipitate the product, followed by filtration and optional recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this room-temperature hydrolysis technology offers profound advantages that extend far beyond simple chemical yield improvements, directly impacting the bottom line and operational resilience of the supply chain. By eliminating the requirement for organic solvents during the reaction phase, manufacturers can drastically reduce the capital expenditure associated with solvent storage, recovery infrastructure, and hazardous waste treatment facilities, leading to substantial cost savings in pharmaceutical intermediates manufacturing. The removal of heating requirements further translates into a significant reduction in utility consumption, lowering the carbon footprint of the production process and aligning with increasingly rigorous global environmental compliance standards. These operational efficiencies enable suppliers to offer more competitive pricing structures while maintaining healthy margins, providing a distinct advantage in price-sensitive markets where cost leadership is a key differentiator.
- Cost Reduction in Manufacturing: The elimination of organic solvents and heating equipment removes major cost centers from the production budget, allowing for a leaner operational model that is less susceptible to fluctuations in energy and solvent prices. Without the need for complex distillation columns or solvent recycling loops, the fixed asset investment required for setting up production lines is significantly lowered, improving the return on investment for new capacity installations. Additionally, the simplified workup procedure reduces labor hours and processing time per batch, enhancing overall plant throughput and asset utilization rates without compromising product quality. These cumulative efficiencies create a robust economic framework that supports long-term price stability for buyers seeking a reliable scutellarein supplier.
- Enhanced Supply Chain Reliability: The reliance on readily available inorganic acids and water as primary reagents mitigates the risk of supply disruptions often associated with specialized organic solvents or expensive enzymatic catalysts. Since the reaction proceeds rapidly at ambient conditions, production lead times are drastically shortened, enabling manufacturers to respond more agilely to sudden spikes in demand or urgent order requirements from downstream pharmaceutical clients. The robustness of the chemistry also means that production can be easily transferred between different manufacturing sites with minimal requalification effort, ensuring continuity of supply even in the face of regional logistical challenges or facility maintenance schedules. This flexibility is crucial for maintaining a steady flow of high-purity pharmaceutical intermediates to support uninterrupted drug formulation and clinical trial activities.
- Scalability and Environmental Compliance: The inherent safety of operating at room temperature without volatile organic compounds makes this process exceptionally easy to scale from pilot plant studies to full commercial production volumes. Regulatory bodies increasingly favor green chemistry principles, and a solvent-free reaction pathway significantly simplifies the environmental impact assessment and permitting processes required for new manufacturing facilities. The reduction in hazardous waste generation lowers disposal costs and minimizes the environmental liability of the manufacturing entity, fostering a more sustainable partnership between chemical producers and eco-conscious pharmaceutical companies. This alignment with sustainability goals not only future-proofs the supply chain against tightening regulations but also enhances the brand reputation of all stakeholders involved in the value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced hydrolysis technology, providing clarity for decision-makers evaluating its potential integration into their supply networks. These insights are derived directly from the patented methodology and reflect the practical realities of scaling this chemistry for industrial applications, ensuring that all stakeholders have a clear understanding of the process capabilities and limitations. Addressing these points proactively helps to mitigate risks and accelerate the adoption of this superior manufacturing route.
Q: Does this hydrolysis method require organic solvents?
A: No, the patented process specifically eliminates the need for organic solvents during the reaction phase, utilizing only concentrated inorganic acid and water, which significantly reduces environmental impact and solvent recovery costs.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction proceeds under mild conditions at room temperature without external heating, typically requiring only 5 to 20 minutes of stirring time before precipitation, contrasting sharply with traditional reflux methods.
Q: How does the purity of the final product compare to traditional methods?
A: The method yields scutellarein with high purity, often exceeding 95% as detected by HPLC after simple filtration or recrystallization, due to the minimized formation of thermal degradation byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Scutellarein Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this room-temperature acid hydrolysis route for producing high-quality scutellarein and possess the technical expertise to bring this innovation to life on a global scale. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of scutellarein meets the exacting standards required for pharmaceutical applications, thereby supporting our partners in achieving their regulatory and therapeutic goals with confidence.
We invite forward-thinking organizations to collaborate with us to leverage these process advancements for their specific product portfolios, driving mutual growth through technological excellence. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements, and ask for specific COA data and route feasibility assessments to validate the fit for your supply chain needs. Together, we can redefine the standards of efficiency and quality in the production of critical pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
