Optimizing Diaryl Ether Production: A Breakthrough in Cesium-Catalyzed Convergent Synthesis
Introduction to Advanced Diaryl Ether Synthesis
The pharmaceutical industry continuously seeks robust synthetic routes for complex molecular scaffolds, particularly diaryl ether compounds which serve as critical building blocks for various therapeutic agents. Patent CN116082335A introduces a significant advancement in this domain, detailing a preparation method that achieves substantially higher yields through a convergent synthetic strategy. This innovation addresses long-standing challenges in organic synthesis regarding efficiency and scalability. The core of this technology lies in the strategic coupling of an aminophenol derivative with a halogenated heterocyclic compound under optimized conditions. By leveraging specific alkali reagents and solvent systems, the process ensures high conversion rates while maintaining exceptional product purity. This development is particularly relevant for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials for drug development pipelines.
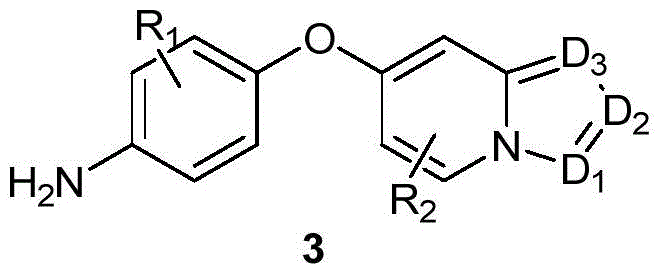
The general reaction pathway illustrates the versatility of this method, accommodating various substituents on both aromatic rings. The ability to tolerate diverse functional groups such as cyano, nitro, and alkoxy moieties expands the applicability of this synthesis across different drug classes. Furthermore, the process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to industrial-scale production. This scalability is essential for meeting the rigorous demands of global supply chains where consistency and volume are paramount. As we delve deeper into the technical specifics, it becomes evident that this patent represents a pivotal shift from traditional linear syntheses to more efficient convergent approaches.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of target diaryl ether compounds, such as Compound 3a, has relied heavily on linear synthetic routes. As documented in prior art like WO2022006386A, these traditional methods involve multiple sequential steps to construct the final molecular architecture. Each step in a linear sequence inherently carries a risk of yield loss, impurity accumulation, and increased operational complexity. For instance, the conventional pathway often requires the separate preparation of distinct fragments followed by a final coupling step that may suffer from poor reactivity or selectivity issues. These inefficiencies compound over the course of the synthesis, leading to an overall process that is economically burdensome and environmentally taxing. The cumulative effect of low yields at each stage significantly restricts the total production output, creating bottlenecks in the supply of critical intermediates.
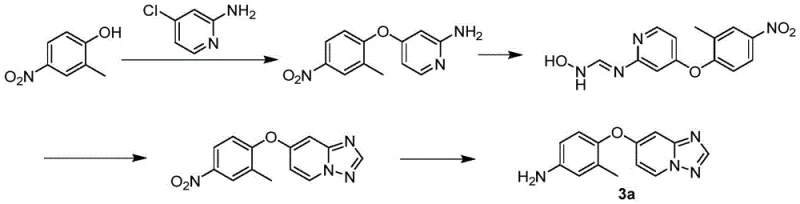
Moreover, traditional methods frequently employ transition metal catalysts, such as copper, to facilitate the etherification reaction. While effective in some contexts, these metals introduce significant downstream processing challenges. The removal of trace heavy metals to meet stringent pharmaceutical specifications often requires additional purification steps, such as specialized scavenging or extensive chromatography. These extra operations not only increase manufacturing costs but also extend production lead times. Additionally, the disposal of metal-contaminated waste streams poses environmental compliance hurdles. Consequently, there is a pressing need for alternative methodologies that can bypass these limitations while delivering superior performance metrics in terms of yield and purity.
The Novel Approach
In stark contrast to the cumbersome linear strategies, the novel approach disclosed in CN116082335A utilizes a convergent synthesis design that dramatically streamlines the production process. This method directly couples a pre-functionalized aminophenol with a halogenated heterocycle in a single, highly efficient step. The key to this breakthrough lies in the meticulous optimization of reaction conditions, specifically the selection of the solvent and the base system. By employing N,N-dimethylacetamide (DMAc) as the reaction medium, the process achieves a solubility profile that favors the formation of the desired ether linkage. More importantly, the introduction of cesium salts, either as the primary base or as a catalytic additive, acts as a powerful promoter for the nucleophilic aromatic substitution reaction. This catalytic effect allows the reaction to proceed rapidly and to completion, even at moderate temperatures.
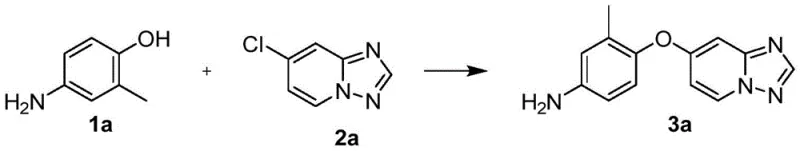
The impact of this novel approach on process efficiency is profound. Experimental data indicates that the use of cesium carbonate as a catalyst can boost yields to over 90%, a significant improvement over the sub-50% yields often observed with traditional copper catalysts or less effective bases. This leap in efficiency translates directly into reduced raw material consumption and lower waste generation. Furthermore, the avoidance of transition metals simplifies the workup procedure, eliminating the need for complex metal removal protocols. This streamlined workflow not only enhances the economic viability of the process but also aligns with green chemistry principles by reducing the environmental footprint. For procurement and supply chain teams, this means a more reliable source of high-purity intermediates with a shorter time-to-market.
Mechanistic Insights into Cesium-Promoted Nucleophilic Substitution
The superior performance of the cesium-promoted synthesis can be attributed to the unique physicochemical properties of the cesium cation. In the context of nucleophilic aromatic substitution, the reaction rate is largely determined by the nucleophilicity of the phenoxide anion generated from the aminophenol starting material. Cesium, being the largest stable alkali metal cation, possesses a low charge density and weak coordination ability compared to smaller cations like sodium or potassium. This characteristic allows the cesium ion to loosely associate with the phenoxide oxygen, effectively leaving the oxygen atom more 'naked' and thus more nucleophilic. This enhanced nucleophilicity facilitates a more aggressive attack on the electron-deficient carbon of the halogenated heterocycle, lowering the activation energy of the rate-determining step. Consequently, the reaction proceeds faster and with greater specificity towards the desired product.
Furthermore, the choice of solvent plays a critical role in stabilizing the transition state and solvating the ionic species involved. DMAc, a polar aprotic solvent, is exceptionally well-suited for this transformation. It effectively solvates the cation without strongly hydrogen-bonding to the anionic nucleophile, thereby preserving its reactivity. Screening results presented in the patent demonstrate that switching from solvents like DMSO or NMP to DMAc results in a marked increase in yield. This solvent effect, combined with the cesium promotion, creates a synergistic environment that drives the equilibrium towards product formation. Additionally, the absence of copper catalysts eliminates potential side reactions such as Ullmann-type couplings or oxidative degradations that can generate difficult-to-remove impurities. This mechanistic clarity provides a solid foundation for scaling the process with confidence in its reproducibility and robustness.
How to Synthesize Diaryl Ether Compound 3 Efficiently
Implementing this high-yield synthesis requires careful attention to stoichiometry and reaction parameters to ensure optimal outcomes. The process begins with the precise weighing of the aminophenol derivative and the halogenated heterocycle, typically in a molar ratio ranging from 1.2:1 to 1.5:1 to drive the reaction to completion. These reactants are suspended in DMAc, and the base system is introduced. If utilizing the catalytic method, a small molar fraction of cesium carbonate (0.02 to 0.3 equivalents) is added alongside a stoichiometric base like potassium carbonate. The mixture is then heated under an inert atmosphere to prevent oxidative degradation of the sensitive amine functionality. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this success.
- Prepare the reaction mixture by combining Compound 1 (aminophenol derivative), Compound 2 (halogenated heterocycle), and a solvent such as N,N-dimethylacetamide (DMAc).
- Add an alkaline reagent like potassium carbonate and a catalytic amount of a cesium salt (e.g., cesium carbonate) to the mixture under an inert atmosphere.
- Heat the reaction to 120°C-160°C for 6-48 hours, then cool, quench with water, and isolate the product via filtration or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cesium-catalyzed synthesis offers tangible strategic benefits that extend beyond simple yield improvements. The primary advantage lies in the significant cost reduction in pharmaceutical intermediate manufacturing driven by process intensification. By consolidating multiple synthetic steps into a single convergent coupling, the overall material throughput is maximized. This efficiency reduces the quantity of raw materials required per kilogram of final product, directly lowering the cost of goods sold (COGS). Moreover, the elimination of expensive and toxic transition metal catalysts removes the need for costly metal scavengers and specialized waste treatment processes. This simplification of the downstream processing train results in substantial operational savings and a leaner manufacturing footprint.
- Cost Reduction in Manufacturing: The shift from linear to convergent synthesis fundamentally alters the economics of production. Traditional linear routes suffer from geometric yield losses, where a 90% yield in each of five steps results in an overall yield of roughly 59%. In contrast, this convergent approach minimizes the number of isolation steps, preserving mass efficiency. The use of inexpensive inorganic bases like potassium carbonate, supplemented by catalytic amounts of cesium salts, avoids the high costs associated with proprietary ligands or precious metal catalysts. This cost structure allows for more competitive pricing models without sacrificing margin, providing a distinct advantage in price-sensitive markets.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and stability of raw materials. The reagents used in this process, including DMAc, potassium carbonate, and cesium carbonate, are commodity chemicals with robust global supply networks. Unlike specialized catalysts that may face sourcing bottlenecks or long lead times, these materials are readily accessible from multiple vendors. This diversity of supply sources mitigates the risk of production stoppages due to material shortages. Furthermore, the robustness of the reaction conditions (tolerance to slight variations in temperature and stoichiometry) ensures consistent batch-to-batch quality, reducing the incidence of out-of-specification batches that can disrupt inventory planning.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges related to heat transfer and mixing, but this methodology is inherently scalable. The reaction operates at moderate temperatures (120°C-160°C) which are easily achievable in standard stainless steel reactors without requiring exotic cryogenic or high-pressure equipment. From an environmental perspective, the absence of heavy metals simplifies effluent treatment. Wastewater streams do not require complex precipitation or ion-exchange steps to remove copper residues, reducing the load on wastewater treatment facilities. This alignment with environmental, social, and governance (ESG) goals is increasingly important for multinational corporations seeking to minimize their ecological impact while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diaryl ether synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for quality assurance personnel establishing control strategies. The insights provided here aim to clarify the operational boundaries and potential advantages of adopting this novel synthetic route.
Q: Why is cesium salt preferred over copper catalysts for this diaryl ether synthesis?
A: Cesium salts act as effective phase-transfer-like catalysts that enhance the nucleophilicity of the phenoxide without introducing toxic heavy metals. Unlike copper catalysts which showed inhibitory effects and low yields (11.2%-15.6%) in screening, cesium carbonate achieved yields up to 94.5% while simplifying downstream purification.
Q: What are the critical reaction parameters for maximizing yield in this process?
A: The patent identifies N,N-dimethylacetamide (DMAc) as the optimal solvent, significantly outperforming DMSO and NMP. Additionally, maintaining a reaction temperature between 130°C and 150°C and using a molar ratio of base to substrate around 2.5:1 are crucial for achieving yields above 90%.
Q: How does this convergent synthesis improve supply chain reliability compared to linear methods?
A: By coupling two advanced intermediates directly rather than building the molecule step-by-step, this method reduces the total number of unit operations. This convergence minimizes cumulative yield losses associated with linear synthesis and reduces the overall production lead time for complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Ether Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has thoroughly analyzed the technological advancements presented in CN116082335A and is fully prepared to leverage this knowledge for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of diaryl ether compound meets the highest international standards. We are committed to delivering high-purity pharmaceutical intermediates that empower your drug development programs.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By partnering with us, you gain access to our deep expertise in process chemistry and our robust production capabilities. Please contact us today to request specific COA data and route feasibility assessments for your target molecules. Let us help you navigate the complexities of chemical manufacturing with confidence and precision.
