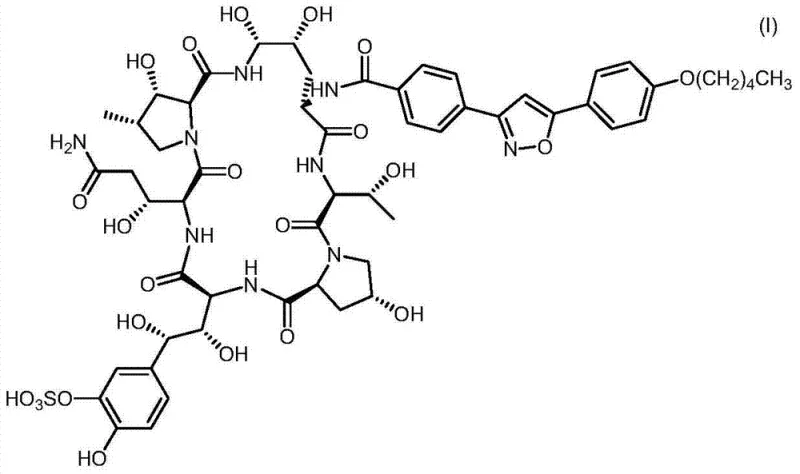
Advanced One-Pot Synthesis of Micafungin: Technical Breakthroughs for Commercial Scale-Up and Supply Chain Stability
The pharmaceutical landscape for antifungal agents is continuously evolving, driven by the need for more efficient manufacturing processes that ensure supply security and cost-effectiveness. A pivotal development in this domain is documented in patent CN103492409A, which discloses an improved process for the preparation of Micafungin, a critical echinocandin antifungal agent. This technical insight report analyzes the proprietary one-pot synthesis method described therein, focusing on its implications for industrial scalability and impurity control. Micafungin, known chemically as a complex lipopeptide, presents significant synthetic challenges due to its multifunctional structure and the sensitivity of its peptide backbone. The traditional approaches often involve multiple isolation and purification steps that increase production costs and extend lead times. By leveraging the innovations outlined in this patent, manufacturers can achieve a more streamlined workflow that directly addresses the pain points of R&D directors and supply chain managers alike. The following analysis dissects the chemical mechanisms and commercial advantages of this novel approach, providing a comprehensive view for stakeholders in the global pharmaceutical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of Micafungin has relied on methods that necessitate the isolation of the activated side chain prior to its coupling with the polypeptide core. This conventional wisdom, as seen in earlier patents and literature, dictates that the micafungin side chain must be activated, purified, and isolated as a stable intermediate before it can be reacted with the micafungin polypeptide core. This multi-step separation process introduces several critical inefficiencies into the manufacturing workflow. Firstly, the isolation of the activated acid intermediate often results in significant material loss, thereby reducing the overall yield of the final active pharmaceutical ingredient. Secondly, the requirement for separate purification steps increases the consumption of solvents and reagents, which not only drives up the cost of goods sold but also complicates waste management and environmental compliance. Furthermore, handling unstable activated intermediates poses risks to product quality, as prolonged storage or additional processing can lead to degradation or the formation of unwanted by-products. These factors collectively create a bottleneck in production capacity, making it difficult to respond rapidly to market demand fluctuations.
The Novel Approach
In stark contrast to the established norms, the method described in patent CN103492409A introduces a groundbreaking one-pot strategy that fundamentally alters the synthesis landscape. This novel approach involves the activation of the micafungin side chain and its subsequent connection to the micafungin polypeptide core within a single reaction mixture, effectively rendering the isolation of the active side chain superfluous. By eliminating the intermediate isolation step, the process achieves a higher degree of operational efficiency and atom economy. The inventors have surprisingly discovered that the activated side chain can be generated in situ and immediately consumed by the polypeptide core without the need for purification, yet still resulting in a product with minimal impurities. This telescoping of reactions not only shortens the overall processing time but also enhances the utilization of the active side chain, leading to substantially higher overall yields. The ability to perform this complex coupling in a single vessel simplifies the equipment requirements and reduces the operational footprint, offering a compelling value proposition for large-scale commercial manufacturing.
Mechanistic Insights into One-Pot Peptide Coupling
The core of this technological advancement lies in the precise orchestration of coupling reagents and additives within the reaction matrix. The process initiates with the mixing of the micafungin side chain, specifically the compound of Formula III, with a coupling additive in a polar aprotic solvent such as dimethylformamide (DMF). The choice of coupling additive is critical; the patent highlights the efficacy of hydroxybenzotriazole (HOBt) or ethyl 2-cyano-2-(oximino)acetate (Oxyma). These additives function by enhancing the reactivity of the activated carboxylic acid and suppressing racemization or other side reactions that are common in peptide synthesis. Following this, a carbodiimide coupling agent, preferably 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) or its hydrochloride salt, is introduced to the mixture. This reagent activates the carboxylic acid group of the side chain, forming an active ester intermediate in situ. The presence of the coupling additive stabilizes this intermediate, preventing it from rearranging into unreactive N-acylurea by-products, which is a common failure mode in carbodiimide-mediated couplings.
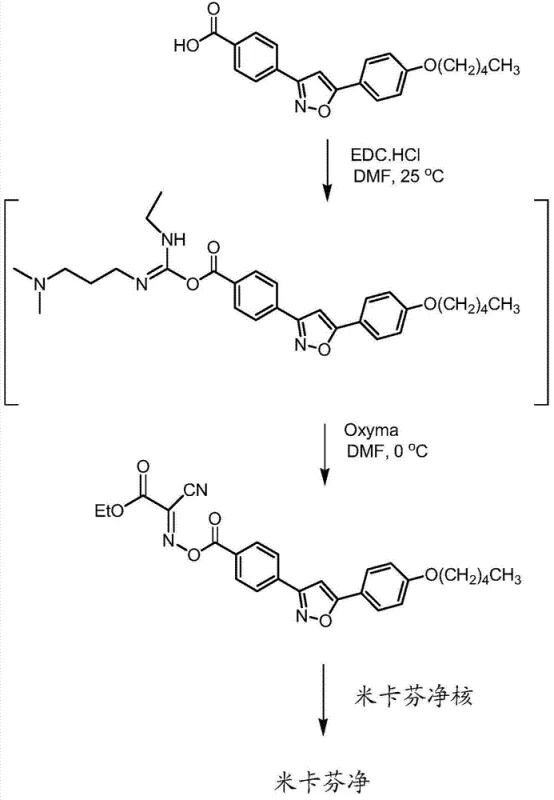
Once the side chain is activated within the reaction vessel, the micafungin polypeptide core, represented by Formula II, is introduced along with an excipient such as diisopropylethylamine (DIPEA). The excipient serves to deprotonate the amine group on the polypeptide core, facilitating the nucleophilic attack on the activated ester of the side chain. This step forms the crucial amide bond that links the side chain to the core, completing the assembly of the Micafungin molecule. The control of temperature during this phase is paramount; the activation typically occurs at temperatures between 0°C and 40°C, while the coupling is often optimized at lower temperatures, around 0°C, to minimize thermal degradation and impurity formation. The mechanistic elegance of this one-pot method ensures that the reactive intermediates are consumed as soon as they are formed, thereby maintaining a low concentration of potentially unstable species and driving the reaction towards the desired product with high fidelity. This precise control over the reaction environment is what allows the process to achieve high purity without the need for intermediate chromatographic purification.
How to Synthesize Micafungin Efficiently
Implementing this synthesis route requires strict adherence to the sequence of reagent addition and reaction conditions to maximize yield and purity. The process begins with the suspension of the micafungin side chain and the coupling additive in the solvent, followed by the controlled addition of the coupling agent to initiate activation. Once the activation phase is complete, the reaction mixture is cooled, and the polypeptide core is added in the presence of the base. The detailed standardized synthesis steps, including specific molar ratios, stirring times, and quenching procedures, are critical for reproducibility and are outlined in the technical guide below. This structured approach ensures that the complex chemical transformations proceed smoothly, minimizing the risk of batch failure.
- Mix micafungin side chain (Formula III) with a coupling additive like HOBt or Oxyma in a polar aprotic solvent such as DMF.
- Add a carbodiimide coupling agent, preferably EDC hydrochloride, to activate the side chain carboxylic acid in situ.
- Introduce the micafungin polypeptide core (Formula II) and an excipient like DIPEA to the mixture to complete the amide bond formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this one-pot synthesis method represents a strategic opportunity to optimize the supply of high-purity antifungal agents. The elimination of the isolation step for the activated side chain directly translates into a reduction in manufacturing complexity and resource consumption. By removing a unit operation that typically involves filtration, drying, and re-dissolution, the process significantly reduces the demand for solvents and energy. This reduction in material usage aligns with broader sustainability goals and contributes to a lower environmental footprint, which is increasingly important for regulatory compliance and corporate social responsibility initiatives. Furthermore, the simplified workflow reduces the potential for human error and cross-contamination, enhancing the overall robustness of the manufacturing process.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substantial cost savings achieved through process intensification. By avoiding the isolation of the intermediate activated side chain, manufacturers eliminate the costs associated with additional purification equipment, labor, and time. The higher overall yield reported in the patent examples indicates better material utilization, meaning less raw material is wasted to produce the same amount of final product. This efficiency gain allows for a more competitive pricing structure without compromising on quality. Additionally, the reduced solvent consumption lowers waste disposal costs, further enhancing the economic viability of the process. These factors combine to create a leaner manufacturing model that is resilient to fluctuations in raw material prices.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex manufacturing processes that have multiple failure points. The one-pot method reduces the number of process steps, thereby decreasing the probability of batch failures and delays. The ability to produce the active ingredient in a shorter timeframe allows for more responsive inventory management and faster turnaround on customer orders. This agility is crucial in the pharmaceutical sector, where demand for antifungal agents can spike unexpectedly due to seasonal infections or outbreaks. By adopting a more streamlined synthesis route, suppliers can maintain higher safety stock levels and ensure consistent availability of the product, mitigating the risk of supply disruptions that could impact downstream drug formulation.
- Scalability and Environmental Compliance: Scaling up peptide couplings is notoriously difficult due to heat transfer and mixing challenges. However, the one-pot nature of this reaction simplifies the scale-up trajectory, as there are fewer transfer operations between vessels. The use of established reagents like EDC and HOBt ensures that the chemistry is well-understood and manageable at large scales. Moreover, the reduction in solvent waste and the avoidance of intermediate isolation steps contribute to a greener manufacturing process. This aligns with stringent environmental regulations and helps manufacturers meet sustainability targets. The process is designed to be robust enough for commercial scale-up of complex pharmaceutical intermediates, ensuring that quality remains consistent from pilot plant to full-scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical feasibility of the one-pot approach. Understanding these details is essential for technical teams evaluating the adoption of this process for their own manufacturing lines.
Q: What is the primary advantage of the one-pot method for Micafungin synthesis?
A: The primary advantage is the elimination of the isolation step for the activated micafungin side chain. This reduces processing time, minimizes product loss during purification, and significantly lowers solvent consumption compared to conventional batch methods.
Q: Which coupling additives are recommended for minimizing impurities in this process?
A: The patent specifically highlights 1-hydroxybenzotriazole (HOBt) and ethyl 2-cyano-2-(oximino)acetate (Oxyma) as effective coupling additives. These reagents enhance reactivity and suppress side reactions, leading to higher crude purity.
Q: How does this process impact the scalability of Micafungin production?
A: By simplifying the workflow into a single reaction vessel for the coupling step, the process reduces operational complexity. This facilitates easier commercial scale-up from laboratory to industrial production, ensuring better supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Micafungin Supplier
The technical potential of the one-pot Micafungin synthesis route is immense, offering a pathway to more efficient and cost-effective production of this vital antifungal agent. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovative processes to fruition. Our facility is equipped with stringent purity specifications and rigorous QC labs to ensure that every batch meets the highest international standards. We understand the complexities of peptide coupling and the critical nature of impurity control in antifungal manufacturing. Our team is dedicated to translating laboratory-scale breakthroughs into robust industrial processes that deliver value to our partners.
We invite you to engage with our technical procurement team to discuss how we can support your supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how this optimized synthesis route can benefit your specific project. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your requirements. Let us collaborate to enhance the efficiency and reliability of your pharmaceutical supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
