Scaling High-Purity o-Nitrobenzaldehyde Production with Advanced Biomimetic Oxidation Technologies
Introduction to Next-Generation Aromatic Aldehyde Synthesis
The global demand for high-purity aromatic aldehydes, particularly o-nitrobenzaldehyde, continues to surge as a critical building block in the synthesis of complex pharmaceutical active ingredients and fine chemicals. Traditional manufacturing routes have long been plagued by inefficiencies, yet a transformative approach detailed in Chinese Patent CN1243717C offers a robust solution through biomimetic catalytic oxygen oxidation. This groundbreaking technology shifts the paradigm from harsh, multi-step chemical oxidations to a sophisticated one-step process that mimics the efficiency of natural enzymatic systems. By utilizing advanced metallophthalocyanine and metalloporphyrin catalysts, this method achieves selective oxidation of o-nitrotoluene under remarkably mild conditions, addressing the longstanding industry challenges of yield optimization, environmental compliance, and operational safety. For R&D directors and procurement strategists, understanding the mechanistic depth of this patent is essential for evaluating its potential to redefine supply chain reliability and cost structures in the production of high-value chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of o-nitrobenzaldehyde has relied on cumbersome multi-step methodologies such as the benzaldehyde method, side-chain methyl chloride hydrolysis, or oxidation using stoichiometric metal compounds. These legacy processes suffer from severe inherent drawbacks, including the requirement for highly acidic environments that cause significant corrosion to reactor vessels and piping infrastructure, leading to frequent maintenance downtime and capital expenditure. Furthermore, the reliance on expensive and environmentally hazardous solvents, such as alkoxyalkylamines disclosed in prior art like CN1111615A, alongside polluting metal salt catalysts, creates a substantial burden for waste treatment and regulatory compliance. The complexity of multi-step syntheses also introduces multiple points of failure, resulting in cumulative yield losses and inconsistent product quality that can disrupt downstream pharmaceutical manufacturing schedules and inflate the total cost of ownership for procurement teams managing global supply chains.
The Novel Approach
In stark contrast, the novel biomimetic approach utilizes molecular oxygen as a clean, abundant, and cost-effective oxidant, driven by structurally sophisticated catalysts that operate efficiently in a methanol-based alkaline medium. This one-step method eliminates the need for corrosive acidic conditions and expensive specialized solvents, replacing them with a system that functions optimally at moderate temperatures between 25°C and 60°C and oxygen pressures ranging from 0.8 to 3.0 MPa. The simplicity of the operation allows for direct conversion of o-nitrotoluene to the target aldehyde with high selectivity, drastically reducing the number of unit operations required and minimizing the formation of unwanted by-products. By streamlining the synthesis into a single catalytic cycle, this technology not only enhances the overall process efficiency but also significantly lowers the barrier for commercial scale-up, offering a compelling value proposition for manufacturers seeking to optimize their production capabilities for aromatic aldehydes.
Mechanistic Insights into Biomimetic Catalytic Oxidation
The core innovation of this technology lies in the deployment of catalysts that structurally and functionally mimic biological enzymes such as hemoglobin, cytochrome C, and cytochrome P-450, which are renowned for their ability to activate molecular oxygen under physiological conditions. The patent specifies the use of metallophthalocyanines (General Formula I) and various metalloporphyrins (General Formulas II, III, and IV), where the central metal atoms—selected from transition metals like Iron (Fe), Cobalt (Co), Manganese (Mn), Copper (Cu), and Zinc (Zn)—play a pivotal role in electron transfer and oxygen activation. These macrocyclic ligands create a protected coordination environment that stabilizes high-valent metal-oxo species, enabling the selective abstraction of hydrogen from the methyl group of o-nitrotoluene to form a reactive carbanion intermediate. This biomimetic mechanism ensures that the oxidation proceeds with high specificity towards the aldehyde, preventing over-oxidation to the corresponding carboxylic acid, which is a common issue in non-catalytic or poorly controlled oxidation processes.
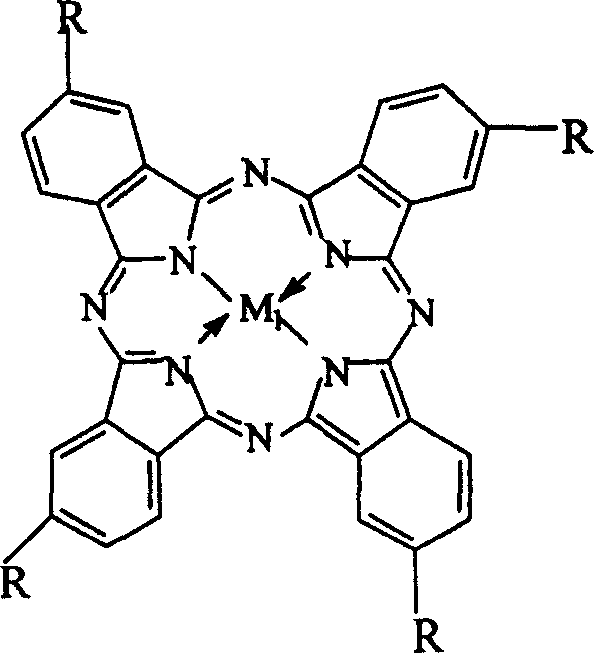
Furthermore, the structural versatility of these catalysts allows for fine-tuning of electronic and steric properties through substitution on the peripheral rings, as illustrated by the variations in General Formulas II, III, and IV. For instance, the introduction of electron-withdrawing groups such as nitro or chloro substituents on the phenyl rings of the porphyrin ligand can modulate the redox potential of the central metal, thereby enhancing catalytic activity and stability under the strongly alkaline reaction conditions (3.0-6.0 mol/L NaOH or KOH). The presence of a strong base acts as a co-catalyst, facilitating the deprotonation of the substrate to generate the nucleophilic species necessary for the oxidation cycle, while the catalyst concentration is kept remarkably low at 0.2% to 1.0% by weight relative to the substrate. This high turnover efficiency underscores the economic viability of the process, as minimal amounts of expensive catalyst are required to drive the reaction to completion.
How to Synthesize o-Nitrobenzaldehyde Efficiently
The practical implementation of this biomimetic oxidation process involves a straightforward sequence of operations that can be readily adapted to existing high-pressure reactor infrastructure commonly found in fine chemical manufacturing facilities. The procedure begins with the preparation of a reaction mixture containing the selected metallophthalocyanine or metalloporphyrin catalyst, a strong alkali base such as sodium hydroxide or potassium hydroxide, and methanol as the solvent, into which o-nitrotoluene is introduced at a controlled concentration. Following the establishment of the liquid phase, the reactor is pressurized with oxygen gas to the specified range, and the temperature is carefully maintained within the optimal window to ensure steady reaction kinetics without triggering thermal runaway or excessive side reactions.
- Prepare the reaction system by dissolving the biomimetic catalyst (0.2-1.0% weight ratio) and strong base (3.0-6.0 mol/L NaOH or KOH) in methanol solvent.
- Introduce o-nitrotoluene substrate into the high-pressure reactor and pressurize with oxygen gas to a range of 0.8-3.0 MPa.
- Maintain reaction temperature between 25-60°C for 6-48 hours, followed by filtration, neutralization, and purification to isolate high-purity o-nitrobenzaldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this biomimetic catalytic technology presents a multitude of strategic advantages that directly impact the bottom line and operational resilience. The shift from multi-step, corrosive processes to a streamlined one-step oxidation significantly reduces the complexity of the manufacturing workflow, thereby lowering labor costs and minimizing the risk of production delays associated with intermediate handling and purification. The use of methanol, a commodity chemical with widespread availability and low cost, replaces expensive and potentially regulated solvents, contributing to a more stable and predictable raw material supply chain that is less susceptible to market volatility. Additionally, the ability to recycle the catalyst multiple times without significant loss of activity means that the effective cost of the catalytic system is amortized over many production batches, leading to substantial long-term savings in material expenditures.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of costly reagents and the reduction of waste disposal fees associated with heavy metal salts and acidic by-products. By utilizing molecular oxygen as the terminal oxidant, the process avoids the purchase of expensive stoichiometric oxidizing agents, while the low catalyst loading and recyclability further decrease the variable cost per kilogram of product. The mild reaction conditions also translate to lower energy consumption for heating and cooling, as the process operates near ambient temperatures compared to the high-energy demands of traditional thermal oxidation methods, collectively resulting in a more cost-competitive manufacturing profile for o-nitrobenzaldehyde.
- Enhanced Supply Chain Reliability: The simplicity and robustness of the one-step synthesis enhance supply chain reliability by reducing the number of critical process parameters that require tight control, thus minimizing the likelihood of batch failures or off-specification products. The use of standard equipment materials that are resistant to alkaline rather than acidic corrosion extends the lifespan of reactor assets and reduces the frequency of unplanned maintenance shutdowns. Furthermore, the reliance on widely available raw materials like o-nitrotoluene, oxygen, and methanol ensures that production can be sustained even during periods of supply chain disruption for specialty chemicals, providing a secure and continuous source of high-purity intermediates for downstream customers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this technology aligns perfectly with modern green chemistry mandates, as it generates significantly less hazardous waste and avoids the release of toxic chlorine-containing by-products common in chlorination-hydrolysis routes. The aqueous workup involving simple filtration and neutralization simplifies the effluent treatment process, reducing the load on wastewater treatment facilities and ensuring compliance with increasingly stringent environmental regulations. The process is inherently scalable, as the reaction kinetics are not limited by mass transfer issues typical of heterogeneous systems, allowing for seamless translation from laboratory optimization to multi-ton commercial production scales without the need for extensive re-engineering of the process flow.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biomimetic oxidation technology, derived directly from the experimental data and specifications outlined in the patent documentation. These insights are intended to clarify the operational parameters and performance expectations for stakeholders evaluating the feasibility of integrating this method into their existing production portfolios.
Q: What are the primary advantages of using biomimetic catalysts over traditional manganese salt catalysts?
A: Biomimetic catalysts such as metallophthalocyanines and metalloporphyrins offer superior selectivity and activity under milder conditions compared to traditional manganese salts. They function similarly to biological enzymes like Cytochrome P-450, allowing for efficient oxygen activation at lower temperatures (25-60°C) and pressures, which significantly reduces equipment corrosion and energy consumption while minimizing environmental pollution associated with heavy metal waste.
Q: Can the catalysts used in this process be recycled for subsequent batches?
A: Yes, a key feature of the technology described in patent CN1243717C is the recyclability of the catalyst. The solid catalyst can be recovered from the reaction mixture via simple suction filtration after the oxidation is complete. This recovery capability allows the catalyst to be reused, which drastically lowers the overall material cost per kilogram of product and reduces the generation of hazardous chemical waste, aligning with green chemistry principles.
Q: What purity levels can be achieved with this one-step oxidation method?
A: Experimental data within the patent indicates that high purity levels are attainable through standard purification techniques. Various examples demonstrate that after filtration, neutralization, and conventional purification steps, the resulting o-nitrobenzaldehyde consistently achieves purity specifications exceeding 98.5%, with many optimized runs reaching purities of 99.0% to 99.7%, making it suitable for demanding pharmaceutical intermediate applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Nitrobenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical and fine chemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the biomimetic catalytic oxidation of o-nitrotoluene can be successfully translated from patent concepts to reliable industrial reality. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch against the highest international standards, guaranteeing consistency and quality for your critical applications.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific supply chain requirements and cost optimization goals. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our advanced manufacturing routes. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a partnership built on transparency, technical excellence, and mutual success in the competitive landscape of chemical intermediates.
