Revolutionizing Ionic Liquid Precursors: Safe Alkylation for Commercial Scale-Up
The landscape of advanced electronic materials is constantly evolving, driven by the relentless demand for higher performance energy storage solutions. A pivotal development in this sector is detailed in Chinese Patent CN102365266B, which introduces a groundbreaking manufacturing method for fluorine-containing N-alkylsulfonylimide compounds. These compounds serve as critical precursors for next-generation ionic liquids, which are increasingly utilized as electrolytes in high-voltage lithium-ion batteries and supercapacitors. The patent outlines a novel alkylation strategy that replaces hazardous and inefficient legacy processes with a robust, scalable, and safe industrial protocol. By utilizing dialkyl sulfates or dialkyl carbonates as alkylating agents, this technology enables the production of high-purity imides with exceptional yields, addressing long-standing purity challenges that have hindered the widespread adoption of fluorinated ionic liquids in commercial battery applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-alkylsulfonylimides has been plagued by significant technical and safety hurdles that impede large-scale commercialization. Traditional approaches often relied on salt exchange reactions where a fluorosulfonylimide salt reacts with a quaternary ammonium halide, such as an imidazolium bromide. As illustrated in the reaction scheme below, this pathway inevitably generates stoichiometric amounts of inorganic salt byproducts (MX) which are notoriously difficult to separate completely from the target ionic liquid.
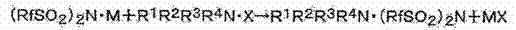
Furthermore, alternative methylation routes described in prior art frequently employed diazomethane, a highly toxic and explosive gas that necessitates extreme safety measures and specialized equipment, drastically inflating capital expenditure. Other methods utilized expensive silver salts of the imide acid, which not only increased raw material costs but also introduced heavy metal contamination risks that are unacceptable for high-purity electronic grade chemicals. These legacy limitations resulted in low overall yields, complex purification workflows, and inconsistent product quality, creating a bottleneck for supply chains requiring reliable volumes of battery-grade electrolytes.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology disclosed in CN102365266B employs a direct alkylation strategy using industrially available dialkyl sulfates or dialkyl carbonates. This approach fundamentally shifts the reaction paradigm by avoiding the formation of inorganic halide salts entirely. Instead of a metathesis reaction, the process utilizes a nucleophilic substitution where the sulfonylimide anion attacks the alkyl group of the sulfate or carbonate. This results in the formation of the target N-alkylsulfonylimide alongside benign byproducts such as sulfuric acid, carbon dioxide, or alcohol, which are far easier to manage and separate than inorganic salts. The elimination of halide byproducts ensures that the resulting ionic liquids are inherently free from corrosive halogen contaminants, a critical specification for maintaining the longevity and stability of electrochemical cells.
Mechanistic Insights into Direct N-Alkylation Chemistry
The core innovation lies in the precise control of reaction thermodynamics and kinetics through solvent engineering and reagent selection. When using dialkyl carbonates, such as dimethyl carbonate, the reaction proceeds under mild thermal conditions to release carbon dioxide and alcohol as volatile byproducts, effectively driving the equilibrium towards completion without generating solid waste. Similarly, the use of dialkyl sulfates, like dimethyl sulfate, allows for rapid alkylation at elevated temperatures. A key mechanistic advantage identified in the patent is the solubility profile of the product; the resulting N-alkylsulfonylimide often exhibits low solubility in the excess alkylating agent or the reaction medium.
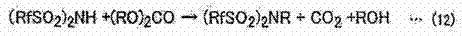
This phenomenon induces spontaneous precipitation of the product as it forms, which serves a dual purpose: it simplifies downstream isolation via filtration and prevents reverse reactions, thereby pushing the conversion rate to near-quantitative levels. Additionally, when starting from sulfonylimide salts (such as potassium or sodium salts), the addition of a catalytic amount of acid can further accelerate the alkylation rate. The process is versatile enough to accommodate various perfluoroalkyl chain lengths, including pentafluoroethyl and heptafluoropropyl groups, enabling the fine-tuning of the final ionic liquid's physical properties, such as viscosity and melting point, to match specific battery operating requirements without compromising on chemical purity.
Impurity control is inherently built into this synthetic design. Unlike silver-mediated routes that risk trace metal contamination, this metal-free alkylation ensures the final product contains no alkali metals or transition metals. The patent specifies that post-reaction water treatment can effectively decompose any unreacted dialkyl sulfate into alcohol and sulfuric acid, which can then be washed away, leaving behind a highly pure organic imide. This rigorous control over the impurity profile is essential for R&D directors who require materials with consistent electrochemical windows and minimal side reactions during cell cycling. The ability to produce compounds like N-methylbis(pentafluoroethanesulfonyl)imide with yields exceeding 90% demonstrates the robustness of this chemical architecture.
How to Synthesize N-Alkylsulfonylimide Efficiently
Implementing this synthesis route requires careful attention to molar ratios and thermal profiles to maximize efficiency. The patent recommends using a molar excess of the alkylating agent, typically ranging from 1 to 50 times the amount of the sulfonylimide substrate, to ensure complete conversion. The reaction is generally conducted at temperatures between 50°C and 150°C, depending on the specific reactivity of the alkylating agent chosen. For R&D teams looking to replicate or scale this process, the key is leveraging the precipitation effect for isolation.
- Charge fluorine-containing sulfonylimide acid or its salt into a reaction vessel equipped with stirring and temperature control.
- Add dialkyl sulfate or dialkyl carbonate in a molar ratio of 1: 1 to 1:50 relative to the substrate to initiate alkylation.
- Maintain temperature between 50°C and 150°C, allowing the product to precipitate for easy filtration and high-purity isolation.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this alkylation technology offers transformative benefits that extend far beyond simple chemical yield improvements. The shift away from exotic reagents like diazomethane or silver salts to commodity chemicals like dimethyl sulfate and dimethyl carbonate fundamentally alters the cost structure of production. These alkylating agents are produced on a massive global scale for other industries, ensuring a stable and predictable supply chain that is not subject to the volatility often seen with specialized fine chemical intermediates. This stability allows procurement managers to negotiate long-term contracts with greater confidence, securing consistent pricing and availability for critical battery material precursors.
- Cost Reduction in Manufacturing: The elimination of expensive silver salts and the avoidance of complex salt-removal purification steps lead to substantial cost savings. Traditional methods often require multiple recrystallizations or ion-exchange chromatography to remove metal ions and halides, which are both time-consuming and resource-intensive. By contrast, the precipitation-based isolation described in this patent allows for simple filtration and washing, drastically reducing solvent consumption and processing time. Furthermore, the absence of heavy metal catalysts removes the need for costly scavenging resins or activated carbon treatments, streamlining the entire manufacturing workflow and lowering the total cost of ownership for the final electronic chemical product.
- Enhanced Supply Chain Reliability: Relying on hazardous gases like diazomethane creates significant logistical bottlenecks, as few suppliers are willing or able to handle such dangerous materials safely. By transitioning to stable liquid alkylating agents, manufacturers can utilize standard chemical logistics networks, reducing lead times and minimizing the risk of shipment delays due to safety regulations. This enhanced logistical flexibility ensures a more resilient supply chain, capable of responding quickly to surges in demand from the rapidly growing electric vehicle and energy storage sectors without being constrained by specialized handling requirements.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids exothermic runaways associated with explosive reagents and generates benign byproducts like carbon dioxide and alcohols that are easier to treat than heavy metal waste streams. This aligns perfectly with increasingly stringent environmental regulations regarding industrial emissions and waste disposal. The ability to scale from laboratory gram quantities to multi-ton commercial production without changing the fundamental chemistry reduces the risk associated with technology transfer, allowing for faster time-to-market for new battery formulations while maintaining a smaller environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkylation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on how this method compares to industry standards. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating these precursors into their existing supply chains.
Q: Why is the direct alkylation method superior to traditional salt exchange?
A: Traditional salt exchange methods often leave behind difficult-to-remove halide byproducts (MX), compromising the purity of the ionic liquid. The direct alkylation method described in CN102365266B avoids halide salts entirely, yielding products free of alkali metals and halogens, which is critical for battery electrolyte performance.
Q: What safety advantages does this process offer over diazomethane routes?
A: Conventional methods using diazomethane involve handling explosive gases, requiring specialized and costly safety infrastructure. This patented process utilizes stable dialkyl sulfates or carbonates, significantly reducing operational risk and eliminating the need for explosive gas handling protocols.
Q: How does the process ensure high yield without expensive catalysts?
A: The process leverages the low solubility of the product in the reaction medium (often the alkylating agent itself acts as a poor solvent). This causes the product to precipitate out as it forms, driving the equilibrium forward without needing expensive silver salts or transition metal catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alkylsulfonylimide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced battery chemistries requires partners who can deliver not just molecules, but validated, scalable processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN102365266B can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities down to the ppm level, guaranteeing that every batch of N-alkylsulfonylimide meets the exacting standards required for high-performance electronic materials.
We invite you to collaborate with us to optimize your electrolyte supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced alkylation capabilities can enhance the performance and reliability of your energy storage solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
