Advanced Halogen-Free Synthesis of p-Nitroaromatic Amides for Commercial Scale-Up
The chemical industry is constantly seeking more sustainable and efficient pathways for synthesizing critical intermediates, and patent CN1098402A presents a significant breakthrough in the preparation of p-nitroaromatic amides. This technology addresses the long-standing environmental and economic challenges associated with traditional amidation processes by introducing a novel halogen-free coupling method. Instead of relying on corrosive acid chlorides that generate substantial halogenated waste, this invention utilizes a direct reaction between amides and nitrobenzene under carefully controlled basic conditions. The strategic manipulation of protic substances within the reaction zone allows for exceptional conversion rates while minimizing downstream purification burdens. For R&D directors and procurement managers alike, this represents a pivotal shift towards greener chemistry that does not compromise on yield or purity. By adopting this methodology, manufacturers can achieve a reliable p-nitroaromatic amide supplier status while significantly reducing the environmental footprint of their operations. The implications for cost reduction in fine chemical intermediates manufacturing are profound, as the elimination of halogen waste treatment translates directly to operational savings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for aromatic amides typically involve the reaction of amines with acid chlorides, a process fraught with significant industrial drawbacks. The primary issue lies in the displacement of halides, which generates corrosive byproducts that necessitate expensive wastewater treatment protocols. Furthermore, the handling of acid chlorides requires stringent safety measures due to their reactivity and toxicity, adding layers of complexity to the supply chain and increasing overall production costs. In many cases, the presence of halogenated impurities can complicate the purification of the final high-purity p-nitroaromatic amides, leading to lower overall yields and extended processing times. These factors collectively create a bottleneck for commercial scale-up of complex pharmaceutical intermediates, making it difficult to maintain consistent quality and competitive pricing. The environmental regulations surrounding halogenated waste disposal are becoming increasingly stringent, forcing manufacturers to seek alternative pathways that avoid these liabilities entirely. Consequently, the industry is in urgent need of a method that bypasses these halogen-dependent steps while maintaining high efficiency.
The Novel Approach
The method disclosed in the patent offers a transformative solution by enabling the direct coupling of amides with nitrobenzene in the presence of a suitable base and controlled protic substances. This approach completely circumvents the need for acid chlorides, thereby eliminating the generation of halogenated waste at the source. By utilizing nitrobenzene not only as a reactant but potentially as a solvent, the process simplifies the reaction mixture and enhances the atom economy of the synthesis. The careful regulation of water and other protic materials ensures that the reaction equilibrium is driven strongly towards the formation of the desired p-nitroaromatic amide product. This results in a cleaner reaction profile with fewer side products, such as azoxybenzene, which are common in less controlled nitro-reduction or coupling scenarios. For supply chain heads, this translates to reducing lead time for high-purity aromatic amines and their precursors, as the downstream workup is significantly streamlined. The robustness of this chemistry allows for flexible scaling, making it an ideal candidate for large-volume production runs required by global agrochemical and pharmaceutical markets.
Mechanistic Insights into Base-Catalyzed Nitrobenzene Coupling
The core of this innovation lies in the precise mechanistic control of the reaction environment, specifically regarding the role of the base and the management of protic species. The reaction proceeds through a nucleophilic attack where the amide, activated by a strong base such as a tetra-alkyl ammonium hydroxide, interacts with the nitrobenzene ring. The presence of the base is crucial for deprotonating the amide nitrogen, thereby increasing its nucleophilicity and facilitating the coupling event. However, the success of this transformation is heavily dependent on maintaining a low concentration of protic substances like water, which can otherwise quench the active species or reverse the reaction. The patent details methods for controlling these levels, such as the use of drying agents like molecular sieves or continuous azeotropic distillation to remove water as it forms. This dynamic control ensures that the catalytic cycle remains active and efficient throughout the process. Understanding this mechanism is vital for R&D teams aiming to optimize the process for specific substrates, as the balance between base strength and protic tolerance can vary depending on the electronic nature of the amide substituents.
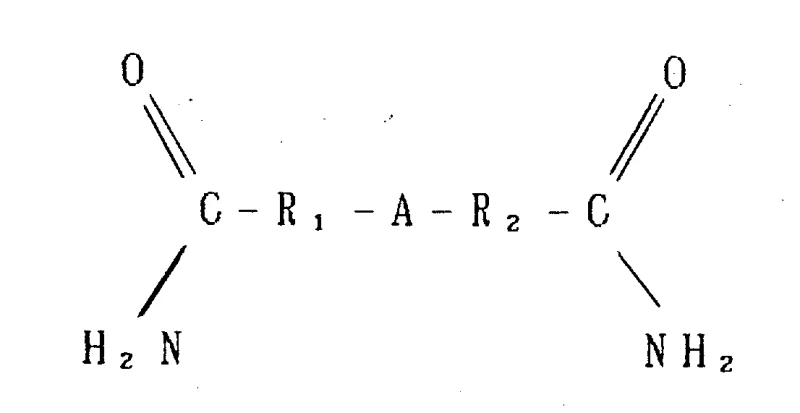
Impurity control is another critical aspect of this mechanistic pathway, particularly in the context of producing high-purity p-nitroaromatic amides for sensitive applications. The formation of byproducts like azoxybenzene is a known risk in reactions involving nitro compounds and amines, but this method mitigates that risk through aerobic conditions and precise temperature control. By conducting the reaction under aerobic conditions, the formation of certain reduction byproducts is suppressed, leading to a cleaner product profile. Additionally, the choice of solvent plays a significant role in impurity management; polar aprotic solvents like DMSO or nitrobenzene itself provide a medium that stabilizes the transition states without participating in unwanted side reactions. The ability to recycle starting materials, such as recovering unreacted amides or nitrobenzene, further enhances the purity of the final isolate. For quality assurance teams, this means that the specification limits for critical impurities can be met more consistently, ensuring that the material is suitable for downstream reduction to p-aminoaromatic amides without requiring extensive chromatographic purification.
How to Synthesize p-Nitroaromatic Amides Efficiently
Implementing this synthesis route requires a systematic approach to reagent preparation and reaction monitoring to ensure optimal yields and safety. The process begins with the selection of high-quality starting materials, specifically the amide and nitrobenzene, which should be dried to minimize initial protic content. The reaction is typically initiated by mixing the amide with a suitable base, such as tetramethylammonium hydroxide, in a solvent system that supports the coupling mechanism. Temperature control is paramount, with the reaction zone maintained between 60°C to 80°C to balance reaction kinetics with thermal stability. Throughout the process, continuous removal of water via distillation or chemical drying agents is essential to drive the equilibrium forward. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety precautions.
- Contact the amide with nitrobenzene in a suitable polar aprotic solvent system such as nitrobenzene itself or DMSO.
- Add a suitable base, preferably a tetra-alkyl ammonium hydroxide, while strictly controlling the amount of protic substances.
- Maintain the reaction temperature between 60°C to 80°C and remove water continuously to drive the equilibrium towards the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this halogen-free synthesis method offers substantial strategic advantages for procurement and supply chain management. The elimination of acid chlorides and the associated halogenated waste streams significantly reduces the cost burden related to hazardous waste disposal and regulatory compliance. This shift not only lowers the direct operational expenses but also mitigates the risk of supply chain disruptions caused by environmental audits or waste treatment bottlenecks. Furthermore, the use of readily available and cost-effective reagents like nitrobenzene and common quaternary ammonium bases enhances the reliability of the raw material supply. For procurement managers, this means a more stable pricing structure and reduced vulnerability to market fluctuations associated with specialized halogenated reagents. The overall simplification of the process flow also contributes to cost reduction in fine chemical intermediates manufacturing by shortening the production cycle and reducing energy consumption associated with extensive purification steps.
- Cost Reduction in Manufacturing: The primary driver for cost savings in this process is the complete removal of halogenated reagents and the subsequent waste treatment costs. Traditional methods require expensive neutralization and disposal of halide salts, which can account for a significant portion of the total manufacturing cost. By avoiding these steps, the process inherently lowers the variable cost per kilogram of the final product. Additionally, the ability to use nitrobenzene as both a reactant and a solvent reduces the volume of solvents required, further cutting down on material costs and solvent recovery expenses. The high conversion rates achieved through protic control mean that less raw material is wasted, improving the overall material efficiency of the plant. These factors combine to create a leaner manufacturing model that is highly competitive in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Supply chain reliability is significantly improved by the use of commodity chemicals that are widely available from multiple global suppliers. Unlike specialized acid chlorides which may have limited sources or long lead times, nitrobenzene and quaternary ammonium hydroxides are standard industrial chemicals with robust supply networks. This diversity in sourcing options reduces the risk of single-supplier dependency and ensures continuity of supply even during market disruptions. The simplified process also reduces the complexity of the manufacturing schedule, allowing for more flexible production planning and faster response to customer demand fluctuations. For supply chain heads, this translates to a more resilient operation capable of maintaining consistent delivery schedules for high-purity p-nitroaromatic amides. The reduced need for specialized waste handling infrastructure also means that production can be scaled or relocated with greater ease.
- Scalability and Environmental Compliance: Scalability is a key strength of this technology, as the reaction conditions are compatible with standard industrial reactor setups and do not require exotic equipment. The ability to operate under aerobic conditions and at moderate temperatures simplifies the engineering requirements for large-scale production. From an environmental compliance standpoint, the halogen-free nature of the process aligns perfectly with increasingly strict global regulations regarding hazardous waste and emissions. This proactive approach to green chemistry future-proofs the manufacturing asset against tightening environmental laws, avoiding potential fines or shutdowns. The reduction in hazardous waste generation also improves the company's sustainability profile, which is becoming a critical factor in supplier selection for major pharmaceutical and agrochemical corporations. This makes the technology not just a chemical improvement, but a strategic asset for long-term business growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this halogen-free synthesis technology. These answers are derived directly from the technical specifications and experimental data provided in the patent literature to ensure accuracy and relevance. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The insights provided here cover aspects ranging from reaction mechanics to downstream processing, offering a comprehensive overview of the technology's capabilities.
Q: How does this method avoid halogenated waste?
A: Unlike conventional methods using acid chlorides which generate corrosive halide waste, this patent describes a direct coupling of amides with nitrobenzene, eliminating halogen displacement byproducts entirely.
Q: What is the role of protic substance control?
A: Controlling protic substances like water is critical. Excess water suppresses the reaction. The process uses drying agents or azeotropic distillation to maintain low protic levels, ensuring high conversion rates.
Q: Can this process be scaled for industrial production?
A: Yes, the use of common solvents like nitrobenzene and robust bases like tetra-alkyl ammonium hydroxides, combined with standard distillation for water removal, makes this highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Nitroaromatic Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to meet the evolving demands of the global pharmaceutical and agrochemical industries. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN1098402A can be successfully translated into robust industrial processes. We are committed to delivering high-purity p-nitroaromatic amides that meet stringent purity specifications through our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging our CDMO expertise, we help clients navigate the complexities of process optimization, impurity control, and regulatory compliance, ensuring a seamless transition from development to commercial supply. Our dedication to quality and reliability makes us a trusted partner for companies seeking to enhance their supply chain resilience.
We invite you to collaborate with us to explore how this halogen-free technology can optimize your specific production requirements and drive significant value for your organization. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing setup, highlighting potential efficiencies and ROI. We encourage you to contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a wealth of technical knowledge and production capacity dedicated to advancing the frontiers of fine chemical manufacturing. Let us help you achieve your supply chain goals with precision and efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
