Advanced Photocatalytic Synthesis of Dibenzylamine for Commercial Scale-up and High Purity
Advanced Photocatalytic Synthesis of Dibenzylamine for Commercial Scale-up and High Purity
The chemical industry is currently witnessing a significant paradigm shift towards greener and more efficient synthetic methodologies, particularly in the production of high-value amine intermediates. Patent CN113443996B introduces a groundbreaking approach for the selective hydrogenation of benzonitrile compounds to synthesize dibenzylamine derivatives using a novel photocatalytic system. This technology leverages the unique electronic properties of silicon carbide supported platinum nanoparticles to drive reactions under mild conditions, offering a compelling alternative to traditional thermal hydrogenation processes. For R&D directors and procurement specialists, this patent represents a critical opportunity to optimize supply chains for pharmaceutical intermediates and rubber additives by adopting a route that minimizes energy consumption while maximizing selectivity. The ability to utilize light energy effectively not only accelerates reaction rates but also opens new avenues for sustainable manufacturing practices that align with global environmental compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of dibenzylamine has relied on methods such as the benzyl chloride route or the benzaldehyde ammonification process, both of which present substantial technical and economic challenges for large-scale manufacturers. The benzyl chloride method, while utilizing low-cost raw materials, suffers from significant drawbacks including the generation of chlorine-containing byproducts that are difficult to remove, leading to poor product quality and high three-waste treatment costs. Similarly, the benzaldehyde ammonification method often requires the use of corrosive ammonia gas and large volumes of organic solvents, which escalate equipment maintenance expenses and pose serious safety hazards due to high-pressure operations. These conventional pathways frequently operate under harsh thermal conditions that demand robust infrastructure and result in lower atom economy, making them less attractive for modern cost-sensitive supply chains. Furthermore, the reliance on noble metal catalysts in some traditional hydrogenation methods without efficient recovery systems can lead to substantial metal loss, further eroding profit margins in competitive markets.
The Novel Approach
In stark contrast to these legacy methods, the photocatalytic selective hydrogenation described in the patent utilizes a platinum-loaded silicon carbide catalyst that operates efficiently under visible light irradiation and moderate hydrogen pressure. This innovative route allows for the conversion of benzonitrile compounds into dibenzylamine with exceptional selectivity, effectively bypassing the formation of unwanted byproducts that plague thermal processes. By maintaining reaction temperatures between 10°C and 100°C and hydrogen pressures as low as 0.1MPa, the process significantly reduces the energy footprint and safety risks associated with high-pressure reactors. The integration of light energy as a driving force not only enhances reaction kinetics but also provides a tunable parameter for controlling product distribution, ensuring high-purity outputs suitable for sensitive pharmaceutical applications. This method exemplifies a strategic advancement in cost reduction in pharmaceutical intermediates manufacturing by simplifying downstream purification steps and extending catalyst life through the robustness of the SiC carrier.

Mechanistic Insights into Pt/SiC Photocatalytic Selective Hydrogenation
The core of this technological breakthrough lies in the synergistic interaction between the platinum nanoparticles and the silicon carbide support, which creates a highly active interface for electron transfer under illumination. Silicon carbide, acting as a semiconductor with a narrow forbidden band width and high carrier concentration, facilitates the excitation of electrons when exposed to light sources simulating sunlight or mercury lamps. These excited electrons are then transferred to the platinum active sites, where they activate molecular hydrogen and the nitrile group of the substrate, promoting selective reduction to the secondary amine without over-reduction to the primary amine. The specific surface area of the SiC carrier, ranging from 20 to 100m²/g, ensures a high dispersion of platinum nanoparticles smaller than 200 nanometers, maximizing the number of active sites available for the reaction. This precise control over the catalyst morphology and electronic structure is crucial for achieving the high conversion rates and selectivity reported in the patent data, providing R&D teams with a reliable framework for optimizing reaction conditions for various substituted benzonitriles.
Furthermore, the mechanism demonstrates remarkable tolerance to diverse functional groups on the aromatic ring, allowing for the synthesis of a wide range of dibenzylamine derivatives without compromising yield or purity. The patent data indicates that substituents such as methyl or trifluoromethyl groups do not hinder the photocatalytic process, suggesting that the electron transfer mechanism is robust against steric and electronic variations in the substrate. This versatility is essential for manufacturers producing specialized fine chemicals where batch-to-batch consistency and impurity profiles are strictly regulated. By understanding the impurity control mechanisms inherent in this photocatalytic cycle, quality assurance teams can better predict and manage potential side reactions, ensuring that the final product meets stringent specifications for use in vulcanization accelerators or active pharmaceutical ingredients. The ability to fine-tune the light intensity and reaction time provides an additional layer of process control that is absent in purely thermal systems.
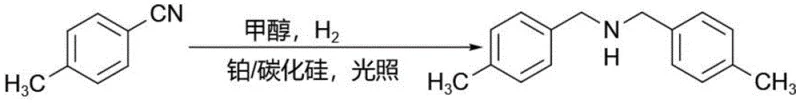
How to Synthesize Dibenzylamine Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for implementing this technology in a pilot or commercial setting, emphasizing the importance of catalyst preparation and reaction parameter control. The process begins with the preparation of the Pt/SiC catalyst, where platinum salts are reduced onto the silicon carbide support under hydrogen pressure to ensure uniform nanoparticle distribution. Once the catalyst is prepared, the benzonitrile substrate is dissolved in a suitable solvent such as ethanol or methanol, and the catalyst is added to form a stable suspension before being transferred to a reactor equipped with a quartz window for light penetration. The reaction is then initiated by purging with hydrogen and maintaining specific pressure and temperature conditions while exposing the mixture to a controlled light source for a defined period. Detailed standardized synthesis steps see the guide below.
- Prepare the Pt/SiC catalyst by loading platinum nanoparticles onto high specific surface area silicon carbide carriers.
- Mix benzonitrile compound with solvent such as ethanol or methanol and add the catalyst to form a suspension.
- React under hydrogen pressure of 0.1-2MPa and light irradiation of 0.01-5W/cm2 at 10-100°C for 0.5-12 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers significant strategic benefits that extend beyond mere technical performance metrics. The elimination of harsh reaction conditions translates directly into reduced operational expenditures, as the need for high-pressure rated equipment and extensive cooling systems is minimized, leading to substantial cost savings in capital investment and maintenance. Additionally, the reusability of the silicon carbide supported catalyst reduces the frequency of catalyst replacement, which is a major cost driver in processes utilizing noble metals, thereby enhancing the overall economic viability of the production line. The simplified workflow also contributes to reducing lead time for high-purity dibenzylamines by shortening the reaction cycle and minimizing the complexity of downstream purification, allowing for faster turnaround times on customer orders. These factors collectively strengthen the supply chain reliability by mitigating risks associated with equipment failure and raw material volatility, ensuring a steady flow of critical intermediates to downstream customers.
- Cost Reduction in Manufacturing: The process achieves cost optimization by eliminating the need for expensive high-pressure infrastructure and reducing energy consumption through the use of light energy instead of thermal heat. By avoiding the use of corrosive ammonia gas and minimizing solvent waste, the method significantly lowers the costs associated with waste treatment and environmental compliance, which are increasingly stringent in the global chemical market. The high selectivity of the reaction reduces the burden on purification units, saving on consumables and labor hours required for distillation or crystallization steps. Furthermore, the efficient use of platinum due to the high dispersion on SiC means less precious metal is tied up in the process, improving cash flow and reducing exposure to metal price fluctuations.
- Enhanced Supply Chain Reliability: The mild operating conditions of the photocatalytic system enhance equipment longevity and reduce the frequency of unplanned shutdowns, ensuring a more consistent production schedule for key intermediates. The use of readily available solvents like ethanol and methanol simplifies logistics and reduces the risk of supply disruptions compared to specialized or hazardous reagents required by older methods. The robustness of the SiC carrier against thermal degradation means the catalyst can be recycled multiple times without significant loss of activity, securing a stable supply of catalytic material and reducing dependency on external catalyst vendors. This stability is crucial for maintaining long-term contracts with pharmaceutical and agrochemical clients who require guaranteed delivery timelines.
- Scalability and Environmental Compliance: The modular nature of photocatalytic reactors allows for easier scale-up from laboratory to commercial production without the exponential increase in safety risks associated with high-pressure thermal reactors. The process generates fewer hazardous byproducts and waste streams, aligning with green chemistry principles and facilitating easier permitting and regulatory approval in environmentally sensitive regions. The ability to operate at lower temperatures reduces the carbon footprint of the manufacturing process, supporting corporate sustainability goals and enhancing the marketability of the final product to eco-conscious consumers. This environmental advantage also future-proofs the production facility against tightening global regulations on industrial emissions and chemical safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis route, based on the detailed data provided in the patent documentation. These answers are designed to clarify the operational feasibility and economic benefits for stakeholders evaluating this technology for adoption in their manufacturing portfolios. Understanding these nuances is critical for making informed decisions about process integration and investment strategies in the competitive fine chemicals sector.
Q: What are the advantages of using SiC as a catalyst carrier in dibenzylamine synthesis?
A: Silicon carbide offers high specific surface area and excellent photocatalytic performance due to its narrow forbidden band width and high electron mobility, enabling efficient electron transfer between metal and carrier.
Q: How does this photocatalytic method compare to traditional high-pressure hydrogenation?
A: This method operates under milder conditions with lower hydrogen pressure and temperature, utilizing light energy to promote reaction rates and selectivity, thereby reducing energy consumption and safety risks.
Q: Is the Pt/SiC catalyst reusable for industrial production?
A: Yes, the silicon carbide carrier is resistant to high temperatures and can be recycled after the reaction, which reduces carbon dioxide emissions and lowers overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibenzylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photocatalytic route for producing high-quality dibenzylamine and are well-positioned to support its industrial implementation. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless and efficient. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required by global pharmaceutical and agrochemical partners. We are committed to leveraging this advanced technology to deliver superior value to our clients through enhanced process efficiency and product consistency.
We invite you to collaborate with us to explore how this innovative synthesis method can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating the tangible economic benefits of switching to this greener alternative. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a competitive edge in the market with reliable, high-purity dibenzylamine supplies.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
