Advanced Isomerization Technology for High-Purity Trans-Oxane Liquid Crystal Monomers
Advanced Isomerization Technology for High-Purity Trans-Oxane Liquid Crystal Monomers
The rapid evolution of the liquid crystal display (LCD) industry has placed immense pressure on the supply chain for high-performance monomeric materials, specifically regarding the purity and stereochemical configuration of oxane compounds. Patent CN102633768B introduces a groundbreaking synthetic methodology that addresses a critical bottleneck in the production of trans-oxane liquid crystal monomers, which are essential components for TN, STN, and TFT display technologies. Historically, the synthesis of these vital materials has been plagued by the formation of undesirable cis-isomers, leading to significant material loss and environmental waste. This patent discloses a robust catalytic isomerization process that effectively transforms low-value cis-configuration oxane compounds into the highly sought-after trans-configuration, thereby optimizing raw material utilization and enhancing the economic viability of liquid crystal manufacturing. For R&D directors and procurement specialists, understanding this technological shift is paramount for securing a competitive edge in the electronic chemicals market.
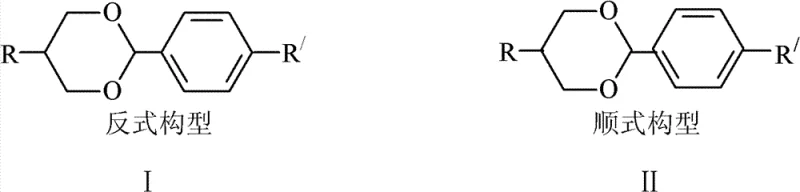
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing oxane-based liquid crystal monomers typically involve the condensation reaction of substituted benzaldehydes with propylene glycol derivatives. While chemically straightforward, this conventional approach suffers from a fundamental stereochemical limitation: it invariably generates a mixture of both cis and trans isomers. In many standard protocols, the unwanted cis-isomer can account for approximately 30% of the crude product mixture. Since only the pure trans-configuration possesses the necessary mesogenic properties for effective liquid crystal alignment in displays, this substantial fraction of cis-isomer is traditionally viewed as waste. Consequently, manufacturers face the dual burden of low overall yields, often hovering around 50% for the target trans-product, and the logistical challenge of disposing of large volumes of chemical waste mother liquor. This inefficiency not only inflates the cost of goods sold but also creates significant environmental compliance hurdles regarding waste treatment and disposal.
The Novel Approach
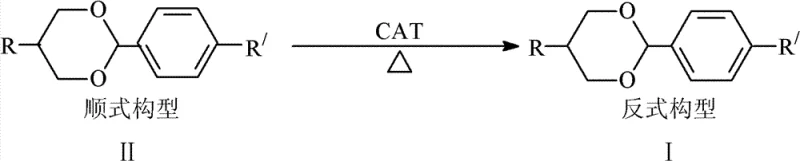
The innovative method detailed in the patent data circumvents these historical inefficiencies by treating the cis-isomer not as waste, but as a valuable feedstock for further conversion. By employing p-toluenesulfonic acid as a catalyst within an organic solvent medium, the process facilitates a stereoselective transformation where the thermodynamically less stable cis-oxane compounds are converted into the more stable trans-configuration. This approach allows manufacturers to recover useful products from what was previously discarded mother liquor, effectively closing the loop on material usage. The reaction conditions are remarkably flexible, operating effectively across a broad temperature range of 35-200°C, with preferred embodiments utilizing moderate heating between 60-100°C. This flexibility ensures that the process can be adapted to various reactor configurations and scale-up requirements without necessitating exotic or prohibitively expensive equipment, making it an ideal candidate for industrial adoption.
Mechanistic Insights into Acid-Catalyzed Isomerization
The core of this technological advancement lies in the acid-catalyzed equilibration of the oxane ring system. Under the influence of the tosic acid catalyst, the acetal linkage within the oxane ring becomes susceptible to reversible cleavage and reformation. This dynamic equilibrium allows the molecule to explore different conformational states, eventually settling into the thermodynamically favored trans-configuration where steric hindrance between the substituents at the 2 and 5 positions is minimized. The presence of the aromatic solvent, such as toluene or xylene, plays a crucial role not just as a medium for heat transfer, but potentially in stabilizing the transition states through solvation effects. By maintaining the reaction for a duration of 2-12 hours, the system is given sufficient time to reach an equilibrium that heavily favors the trans-isomer, driven by the removal of the product or the inherent stability difference. This mechanistic pathway avoids the need for harsh reagents or complex multi-step sequences, relying instead on fundamental thermodynamic principles to drive the reaction towards the desired high-purity product.
From an impurity control perspective, this method offers a distinct advantage by simplifying the purification profile. In traditional synthesis, separating cis and trans isomers often requires difficult chromatographic separations or multiple recrystallizations that sacrifice yield. However, by chemically converting the cis-impurity into the trans-product, the burden on downstream purification is significantly reduced. The patent data indicates that after standard workup procedures involving washing, extraction, and recrystallization, the resulting trans-oxane compounds achieve a purity (omega) of greater than 99%. This high level of purity is critical for liquid crystal applications, where even trace impurities can degrade the voltage holding ratio and response time of the final display panel. The ability to consistently achieve such high purity standards through a simple isomerization step represents a major leap forward in process chemistry for electronic materials.
How to Synthesize Trans-Oxane Compounds Efficiently
The operational protocol for implementing this isomerization technology is designed for ease of execution and scalability, making it accessible for both pilot-scale optimization and full commercial production. The process begins with the charging of the cis-oxane starting material, which may be sourced from recycled mother liquors or crude reaction mixtures, into a standard reactor equipped with mechanical stirring and reflux capabilities. A catalytic amount of p-toluenesulfonic acid is introduced, typically ranging from 1% to 20% by mass relative to the substrate, followed by the addition of the organic solvent. The detailed standardized synthesis steps, including specific molar ratios, precise temperature ramping rates, and quenching procedures, are outlined in the technical guide below to ensure reproducibility and safety during operation.
- Prepare the reaction mixture by combining cis-oxane compounds with p-toluenesulfonic acid catalyst in an organic solvent such as toluene.
- Heat the reaction mixture to a temperature between 35-200°C, preferably 60-100°C, and maintain stirring for 2-12 hours to facilitate isomerization.
- Upon completion, purify the resulting trans-oxane compounds through washing, extraction, and recrystallization to achieve purity greater than 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this isomerization technology translates directly into enhanced operational resilience and cost efficiency. By unlocking the value trapped in cis-isomer waste streams, manufacturers can drastically reduce their reliance on fresh raw materials, effectively increasing the output from the same input of starting benzaldehydes and glycols. This improvement in atom economy means that the effective cost per kilogram of the final high-purity trans-oxane monomer is significantly lowered, providing a buffer against volatility in upstream petrochemical pricing. Furthermore, the reduction in waste volume simplifies environmental compliance and lowers the overhead costs associated with hazardous waste disposal, contributing to a more sustainable and economically robust manufacturing footprint.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the valorization of by-products. In conventional processing, the 30% cis-fraction represents a total loss of capital invested in raw materials and energy. By converting this fraction into saleable product, the effective yield of the entire production line is boosted substantially without proportional increases in raw material procurement. Additionally, the use of common, commodity-grade solvents like toluene and inexpensive catalysts like tosic acid ensures that the variable costs of the reaction remain low. The elimination of complex separation steps further reduces utility consumption and labor hours, driving down the overall cost of goods sold for these high-value electronic chemicals.
- Enhanced Supply Chain Reliability: Supply continuity for critical LCD materials is often threatened by the complexity of synthesis and the scarcity of high-purity intermediates. This method enhances reliability by creating an internal feedback loop; even if the initial condensation step underperforms or generates excess cis-isomer, the supply of the final trans-product remains secure through the isomerization recovery step. This redundancy mitigates the risk of batch failures and ensures a steady flow of material to downstream formulation teams. Moreover, the robustness of the reaction conditions, which tolerate a wide temperature range, reduces the likelihood of process upsets due to minor equipment fluctuations, ensuring consistent delivery schedules for global customers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but this isomerization reaction is inherently scalable due to its homogeneous nature and moderate thermal requirements. The ability to operate at temperatures as low as 60°C reduces the energy intensity of the process compared to high-temperature alternatives. From an environmental standpoint, the drastic reduction in waste mother liquor discharge aligns with increasingly stringent global regulations on industrial effluent. By minimizing the environmental footprint, manufacturers can avoid regulatory fines and maintain their social license to operate, which is increasingly important for suppliers serving major multinational electronics corporations with strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cis-to-trans isomerization technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is essential for making informed decisions about process integration and supplier qualification.
Q: What is the primary advantage of this isomerization method over traditional synthesis?
A: Traditional synthesis often yields a mixture containing approximately 30% unwanted cis-isomers which are typically discarded as waste. This patented method converts that waste cis-isomer back into the valuable trans-isomer, significantly improving overall yield and reducing environmental impact.
Q: What catalysts and solvents are utilized in this process?
A: The process utilizes p-toluenesulfonic acid (tosic acid) as the catalyst. Suitable organic solvents include toluene, xylene, n-heptane, and n-hexane, with toluene being a preferred embodiment for optimal reaction control.
Q: What purity levels can be achieved with this recycling technique?
A: Through the described isomerization and subsequent purification steps including recrystallization, the process achieves a trans-oxane compound purity (omega) of greater than 99%, meeting the stringent requirements for liquid crystal display applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-Oxane Compounds Supplier
As the demand for high-performance liquid crystal materials continues to surge, partnering with a technically proficient manufacturer is critical for success. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced catalytic technologies like the one described in CN102633768B to deliver superior products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of trans-oxane compounds meets the exacting standards required for next-generation display applications.
We invite you to engage with our technical procurement team to discuss how our optimized isomerization processes can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to our recovered-material workflow. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to serve as your long-term strategic partner in the electronic chemicals market. Contact us today to secure a reliable supply of high-purity liquid crystal monomers.
