Advanced Catalytic Synthesis of 2,4,4,4-Tetrachlorobutyronitrile for Commercial Scale-Up and High-Purity Supply
The chemical industry is constantly evolving, driven by the need for more efficient and sustainable synthetic routes for critical intermediates. Patent CN109535029B introduces a groundbreaking method for the synthesis of 2,4,4,4-tetrachlorobutyronitrile, a vital building block in the production of agrochemicals and pharmaceuticals. This innovative approach utilizes a telomerization reaction between carbon tetrachloride and acrylonitrile, catalyzed by a sophisticated system comprising copper carboxylate and imidazole ligands. Unlike traditional methods that struggle with solubility and side reactions, this patent details a process that achieves a remarkable product purity of 99.64% and a single-pass reaction yield of 90.6%. For R&D Directors and Procurement Managers seeking a reliable agrochemical intermediate supplier, this technology represents a significant leap forward in process chemistry. The method eliminates the need for hazardous initiators and operates under mild conditions, ensuring not only high quality but also enhanced safety and environmental compliance. By leveraging this advanced catalytic system, manufacturers can overcome the limitations of conventional synthesis, paving the way for cost reduction in fine chemical manufacturing and more robust supply chains for high-purity intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,4,4,4-tetrachlorobutyronitrile has relied on Atom Transfer Radical Addition (ATRA) using inorganic copper salts such as copper chloride or oxides. These conventional catalysts suffer from inherently poor solubility in organic solvents, which severely limits their dispersion within the reaction phase and inhibits catalytic activity. To compensate for this inefficiency, prior art methods often require high reaction temperatures or the addition of radical initiators like AIBN, which introduce significant safety risks and complicate the purification process. Furthermore, the inorganic nature of these catalysts makes recovery and recycling extremely difficult, leading to increased waste generation and higher operational costs. The use of high-boiling solvents like polyethylene glycol in some existing processes further exacerbates energy consumption during product separation. Consequently, these traditional routes often result in moderate yields, complex by-product profiles, and significant challenges in scaling up for commercial production, making them less attractive for modern industrial applications focused on sustainability and efficiency.
The Novel Approach
In stark contrast, the novel method disclosed in patent CN109535029B employs a homogeneous catalytic system based on copper carboxylate and/or cuprous carboxylate combined with specific imidazole ligands. This strategic combination addresses the solubility issues of inorganic salts by utilizing the unique 'Chinese lantern' dimeric structure of copper carboxylates, which dissolves readily in organic phases. The addition of imidazole ligands further stabilizes the copper complex, enhancing its ability to activate carbon tetrachloride and generate chlorine radicals efficiently without the need for external initiators. This results in a reaction that proceeds smoothly under mild temperatures, significantly reducing energy consumption and minimizing the formation of polymeric tars and other by-products. The simplicity of the process operation, coupled with the ease of catalyst recovery and reuse, offers a compelling advantage for industrial adoption. By shifting from heterogeneous inorganic catalysts to this tailored organic-metallic complex, the process achieves superior selectivity and yield, establishing a new standard for the commercial scale-up of complex organic synthesis.
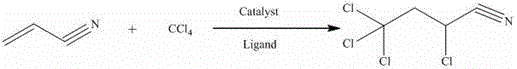
Mechanistic Insights into Cu-Carboxylate and Imidazole Catalyzed Telomerization
The core of this technological breakthrough lies in the intricate mechanistic interplay between the copper catalyst and the imidazole ligand. The copper carboxylate, possessing a dimeric structure, acts as the primary source of copper ions that cycle between cuprous and cupric oxidation states during the reaction. When coordinated with the imidazole ligand, the copper center forms a stable complex that is highly soluble in the organic reaction medium, ensuring uniform catalytic activity throughout the system. This complex facilitates the homolytic cleavage of the carbon-chlorine bond in carbon tetrachloride, generating trichloromethyl radicals that initiate the telomerization chain. The imidazole ligand plays a critical role in modulating the redox potential of the copper center, allowing it to effectively capture and release chlorine radicals in a controlled manner. This precise control over radical concentration is essential for preventing uncontrolled polymerization of acrylonitrile, which is a common side reaction in free radical processes. The result is a highly selective addition reaction that favors the formation of the desired 2,4,4,4-tetrachlorobutyronitrile over oligomeric by-products.
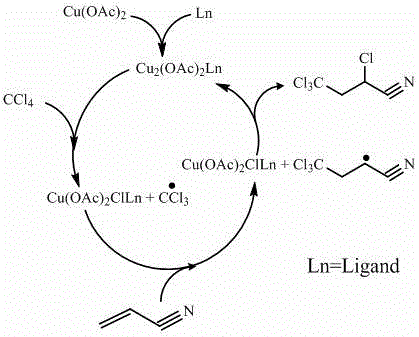
Furthermore, the specific structure of the imidazole ligand significantly influences the efficiency of the catalytic cycle. The nitrogen atoms in the imidazole ring coordinate with the copper ions, forming a bridge-shaped cyclic complex that enhances the stability of the catalyst in the organic phase. Substituents on the imidazole ring, such as alkyl chains, can further improve solubility and steric properties, optimizing the interaction between the catalyst and the reactants. This structural tuning ensures that the copper complex remains active throughout the reaction duration, even under mild thermal conditions. The ability of the ligand to form a stable yet reactive complex prevents the precipitation of copper salts, which is a frequent cause of catalyst deactivation in conventional methods. By maintaining the catalyst in a homogeneous state, the process ensures consistent reaction rates and high conversion levels. This deep understanding of the coordination chemistry involved allows for the precise optimization of reaction parameters, leading to the exceptional purity and yield reported in the patent data.

How to Synthesize 2,4,4,4-Tetrachlorobutyronitrile Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalytic system and the control of reaction parameters to maximize efficiency. The process begins with the formation of the active copper-imidazole complex in the presence of carbon tetrachloride, which serves as both a reactant and a solvent. Detailed standardized synthesis steps see the guide below to ensure reproducibility and safety during scale-up. The injection rate of acrylonitrile is a critical variable, as it must be balanced against the rate of radical generation to prevent local accumulation of monomers that could lead to polymerization. Temperature control is equally vital, with a two-stage heating profile often preferred to first activate the catalyst and then drive the addition reaction to completion. Post-reaction processing involves simple filtration to remove any insoluble residues, followed by distillation to recover the excess carbon tetrachloride for reuse. This streamlined workflow minimizes waste and operational complexity, making it an ideal candidate for continuous or batch production environments focused on high-purity pharmaceutical intermediates.
- Prepare the catalytic system by mixing copper carboxylate and imidazole ligand in carbon tetrachloride, heating to 70-90°C to ensure full dispersion and complex formation.
- Inject acrylonitrile slowly into the reaction kettle at a controlled rate while maintaining the temperature between 100-120°C to facilitate radical addition.
- Upon completion, filter the mixture, recover carbon tetrachloride via atmospheric distillation, and purify the product through reduced pressure rectification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis method offers substantial benefits that directly address the pain points of procurement and supply chain management in the fine chemical sector. The elimination of expensive and hazardous radical initiators like AIBN significantly reduces raw material costs and simplifies the handling requirements, thereby lowering the overall cost reduction in fine chemical manufacturing. The high solubility and stability of the copper carboxylate catalyst allow for efficient recovery and recycling, which not only reduces the consumption of precious metal resources but also minimizes the generation of heavy metal waste. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, enhancing the long-term viability of the supply chain. Additionally, the mild reaction conditions reduce energy consumption and equipment stress, leading to lower utility costs and extended equipment lifespan. For Supply Chain Heads, the robustness of this process ensures reducing lead time for high-purity intermediates by minimizing batch failures and reprocessing needs.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by eliminating the need for external initiators and utilizing cheap, readily available raw materials like carbon tetrachloride and acrylonitrile. The high single-pass yield of over 90% means that less raw material is wasted, and the downstream purification burden is drastically reduced. Furthermore, the ability to recycle the catalyst and solvent multiple times without significant loss of activity translates into substantial long-term savings on consumables. This economic efficiency makes the process highly competitive in the global market for agrochemical intermediates, where margin pressure is often intense.
- Enhanced Supply Chain Reliability: The use of stable, commercially available catalysts and ligands ensures a consistent supply of critical reagents, reducing the risk of production delays due to material shortages. The simplicity of the operation and the mild conditions enhance process safety, minimizing the likelihood of unplanned shutdowns or accidents that could disrupt supply continuity. High product purity of 99.64% reduces the need for extensive quality control re-testing and customer rejections, fostering stronger relationships with downstream partners. This reliability is crucial for maintaining the trust of multinational corporations that depend on just-in-time delivery of high-purity intermediates for their own manufacturing schedules.
- Scalability and Environmental Compliance: The homogeneous nature of the catalytic system and the absence of hazardous initiators make this process highly scalable from pilot plant to commercial production volumes. The reduced formation of polymeric tars and by-products simplifies waste treatment and lowers the environmental footprint of the manufacturing facility. Compliance with environmental standards is easier to achieve, as the process generates less hazardous waste and consumes less energy compared to traditional high-temperature methods. This scalability and compliance ensure that the production can grow in tandem with market demand without encountering regulatory bottlenecks, securing the long-term supply of 2,4,4,4-tetrachlorobutyronitrile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 2,4,4,4-tetrachlorobutyronitrile using this advanced catalytic method. These answers are derived directly from the technical specifications and beneficial effects outlined in patent CN109535029B, providing clarity for stakeholders evaluating this technology. Understanding the mechanistic advantages and operational parameters is essential for making informed decisions about process adoption and supply partnerships. The data presented here reflects the rigorous testing and optimization performed during the patent development phase, ensuring accuracy and reliability for industrial application.
Q: What are the advantages of using copper carboxylate over inorganic copper salts?
A: Copper carboxylates possess a 'Chinese lantern' dimeric structure that offers superior solubility in organic solvents compared to inorganic salts, leading to better catalyst dispersion and higher reaction efficiency without requiring harsh conditions.
Q: How does the imidazole ligand improve the reaction yield?
A: The imidazole ligand forms a stable complex with copper ions, enhancing their ability to generate chlorine radicals from carbon tetrachloride and effectively controlling the radical concentration to minimize polymerization by-products.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates under mild temperatures without initiators like AIBN, uses recyclable catalysts, and achieves high single-pass yields, making it economically viable and safe for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,4,4-Tetrachlorobutyronitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthetic routes for key intermediates like 2,4,4,4-tetrachlorobutyronitrile. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch meets the exacting standards required by the global pharmaceutical and agrochemical industries. We understand the complexities involved in managing copper-catalyzed reactions and have the infrastructure in place to handle the specific safety and environmental requirements of this technology. By partnering with us, you gain access to a supply chain that is not only reliable but also optimized for cost and performance.
We invite you to collaborate with us to explore how this advanced synthesis method can enhance your product portfolio and operational efficiency. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to reach out to request specific COA data and route feasibility assessments to verify the compatibility of this material with your downstream processes. Together, we can drive innovation and efficiency in the production of high-value fine chemicals, ensuring a competitive edge in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
