Advanced Fries Rearrangement Technology for Commercial Scale Halogenated P-Acetyl Phenol Production
Introduction to Advanced Intermediate Synthesis
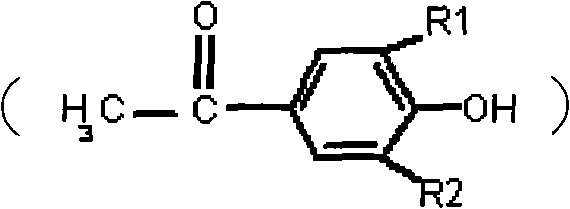
The pharmaceutical and fine chemical industries rely heavily on the availability of high-purity aromatic ketones, specifically halogenated p-acetyl phenol compounds, which serve as critical building blocks for a wide array of bioactive molecules. A pivotal advancement in the manufacturing of these essential intermediates is detailed in patent CN101619021A, which introduces a transformative approach to the classic Fries rearrangement reaction. This intellectual property addresses long-standing inefficiencies in synthetic organic chemistry, particularly focusing on the optimization of reaction conditions to maximize yield while minimizing environmental impact and operational complexity. By fundamentally altering the sequence in which reagents are introduced into the reaction vessel, this technology overcomes the thermodynamic and kinetic barriers that have historically plagued the production of compounds such as 2-chloro-4-acetyl phenol. For R&D directors and process chemists, understanding this shift is crucial, as it represents a move from empirical trial-and-error to a mechanism-driven process design that ensures reproducibility and scalability. The implications of this patent extend beyond mere academic interest, offering a tangible pathway for industrial manufacturers to secure a more reliable supply chain for these high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogenated p-acetyl phenol compounds via Fries rearrangement has been fraught with significant technical challenges that hinder industrial adoption. Traditional protocols typically involve the simultaneous introduction of the Lewis acid catalyst, such as anhydrous aluminum chloride, and the halogenophenyl acetate substrate into the reaction vessel, often followed by heating. This indiscriminate mixing triggers an immediate and vigorous exothermic reaction, releasing a substantial amount of heat that is difficult to control. Consequently, the reaction mixture frequently solidifies into a "deadlocked" mass, a phenomenon where the materials fuse together, rendering mechanical stirring ineffective. When stirring is interrupted, the contact between the catalyst and the substrate is severely compromised, leading to incomplete conversion and the formation of complex by-product mixtures. Furthermore, to勉强 ensure the reaction proceeds to any meaningful extent, conventional methods require a massive excess of catalyst, often utilizing three times the stoichiometric amount. This not only drives up raw material costs but also creates a heavy burden on downstream waste treatment facilities due to the generation of aluminum-containing sludge. Yields in these traditional processes are notoriously poor, with literature citing conversion rates as low as 23% for key derivatives like 2-chloro-4-acetyl phenol, making the process economically unviable for large-scale commercial production.
The Novel Approach
In stark contrast to the chaotic nature of conventional mixing, the methodology outlined in patent CN101619021A introduces a disciplined, sequential feeding strategy that fundamentally changes the reaction environment. The core innovation lies in pre-dissolving the anhydrous aluminum chloride catalyst in nitrobenzene solvent before any substrate is introduced. This creates a homogeneous catalytic solution that is thermally stable and ready for reaction. The halogenophenyl acetate compound is then added slowly and controllably into this prepared catalyst solution. This reverse feeding order prevents the localized thermal spikes that cause solidification, ensuring that the reaction mixture remains fluid and stirrable throughout the entire process. As the reaction progresses, the product formed is slightly soluble in the nitrobenzene solvent, effectively removing it from the immediate catalytic zone and preventing it from interfering with the ongoing rearrangement. This dynamic allows the catalyst to remain active and recyclable within the system, eliminating the need for the massive excesses required in older methods. The result is a dramatic improvement in operational safety and chemical efficiency, transforming a problematic batch process into a robust, high-yielding manufacturing operation suitable for modern chemical plants.
Mechanistic Insights into Modified Fries Rearrangement
To fully appreciate the technical superiority of this process, one must delve into the mechanistic nuances of the Lewis acid-catalyzed rearrangement. The Fries rearrangement involves the migration of an acyl group from the oxygen of a phenolic ester to the ortho or para position of the aromatic ring. In the presence of aluminum chloride, the carbonyl oxygen of the ester coordinates with the aluminum, generating a highly electrophilic acylium ion intermediate. In the traditional "all-in" method, the rapid generation of these ions in a concentrated, solvent-poor environment leads to polymerization and tar formation. However, by pre-dissolving the catalyst in nitrobenzene, the new process modulates the concentration of active catalytic species. Nitrobenzene, being a polar aprotic solvent with high boiling point characteristics, stabilizes the ionic intermediates without participating in the reaction itself. This stabilization is critical for controlling the regioselectivity of the migration, favoring the formation of the para-isomer (p-acetyl phenol) over the ortho-isomer, which is often the desired outcome for pharmaceutical applications. The slow addition of the substrate ensures that the rate of acylium ion generation matches the rate of the electrophilic aromatic substitution, preventing the accumulation of reactive intermediates that could lead to side reactions. This precise kinetic control is the key to achieving the high purity levels required by regulatory bodies for API intermediates.
Furthermore, the mechanism explains the significant reduction in impurity profiles observed in this novel process. In conventional setups, the "deadlocked" solid mass creates micro-environments where heat cannot dissipate, leading to thermal degradation of the product and the formation of chlorinated or brominated by-products depending on the substrate. By maintaining a homogeneous liquid phase through the use of nitrobenzene and controlled feeding, heat transfer is optimized, and the reaction temperature can be strictly maintained within the optimal range of 90°C to 100°C. This thermal uniformity ensures that the rearrangement proceeds cleanly, minimizing the formation of tars and polymeric residues. Additionally, because the catalyst is not consumed in side reactions or trapped in solid waste, the effective molar ratio of catalyst to substrate can be reduced to between 1.2:1 and 2:1, compared to the 3:1 ratio of the past. This efficiency not only lowers costs but also simplifies the work-up procedure, as there is less aluminum sludge to filter and dispose of, directly contributing to a greener and more sustainable manufacturing footprint.
How to Synthesize Halogenated P-Acetyl Phenol Efficiently
Implementing this advanced synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure consistent quality and yield. The process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to multi-ton industrial reactors without losing efficiency. The critical success factors involve the precise control of solvent-to-catalyst ratios, the rate of substrate addition, and the maintenance of specific temperature windows throughout the reaction duration. Operators must ensure that the anhydrous conditions are maintained to prevent catalyst deactivation, and the slow addition protocol must be automated or carefully monitored to avoid thermal runaway. The following guide outlines the standardized operational steps derived from the patent examples, providing a clear roadmap for process engineers to replicate the high yields reported in the intellectual property documentation.
- Dissolve the anhydrous aluminum chloride catalyst in nitrobenzene solvent within the reaction vessel to form a homogeneous catalytic solution.
- Slowly add the halogenophenyl acetate compound into the catalyst-solvent mixture over a controlled period, typically 30 minutes, while maintaining temperature between 60°C and 130°C.
- Stir the reaction mixture for 1 to 6 hours to ensure complete rearrangement, followed by hydrolysis with ice dilute hydrochloric acid to isolate the crude product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis process offers compelling economic and logistical benefits that directly impact the bottom line. The primary advantage lies in the drastic reduction of raw material costs, driven by the elimination of excessive catalyst usage. In traditional manufacturing, the requirement for three equivalents of aluminum chloride represented a significant direct cost, alongside the indirect costs associated with handling, storage, and disposal of hazardous chemical waste. By optimizing the catalyst loading to near-stoichiometric levels, the new process significantly reduces the bill of materials. Moreover, the substantial increase in yield—from roughly 23% in legacy methods to over 96% in the optimized process—means that less starting material is required to produce the same amount of final product. This efficiency gain translates directly into lower unit costs, allowing suppliers to offer more competitive pricing structures without compromising on margin. The reduction in waste generation also lowers the environmental compliance costs, which are becoming increasingly significant in the global chemical industry.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily due to the optimization of catalyst consumption and the maximization of product yield. By avoiding the need for a three-fold excess of aluminum chloride, manufacturers can achieve substantial savings on one of the most consumable reagents in the process. Additionally, the high yield ensures that the expensive halogenated phenyl acetate starting materials are converted into valuable product rather than lost as tar or by-products. This efficiency eliminates the need for complex and costly purification steps that are often required to salvage product from low-yield reactions. The cumulative effect is a streamlined cost structure that enhances profitability and provides a buffer against fluctuations in raw material market prices.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the operability of the new process is a game-changer. The elimination of the "deadlock" phenomenon means that batch failures due to stirring issues are virtually eradicated. In the past, a solidified reactor meant a lost batch, delayed shipments, and potential penalties for non-delivery. With the new liquid-phase protocol, reaction consistency is guaranteed, leading to predictable production schedules and reliable lead times. This reliability is crucial for pharmaceutical customers who operate on Just-In-Time inventory models and cannot afford interruptions in their API synthesis lines. The ability to consistently deliver high-purity intermediates on schedule strengthens the supplier-buyer relationship and reduces the risk profile of the supply chain.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, addressing one of the biggest hurdles in fine chemical manufacturing. The use of nitrobenzene as a solvent facilitates heat management in large reactors, preventing the hot spots that are common in bulkier traditional batches. This thermal control makes it safer to operate at the 100 MT scale, ensuring that the quality achieved in the lab is maintained in the plant. Furthermore, the reduction in aluminum waste aligns with increasingly stringent global environmental regulations. Lower waste volumes mean reduced fees for hazardous waste disposal and a smaller carbon footprint for the manufacturing site. This environmental stewardship is not just a regulatory necessity but also a marketing asset for companies aiming to partner with sustainability-focused multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific Fries rearrangement technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms historical precedents. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific supply chain needs.
Q: Why does the traditional Fries rearrangement method often result in low yields and operational difficulties?
A: Conventional methods often mix the catalyst and substrate simultaneously, causing an immediate, intense exothermic reaction that leads to the formation of a solidified, deadlocked mass. This physical state prevents effective stirring and contact between reactants, resulting in poor conversion rates (often as low as 23%) and significant catalyst wastage.
Q: How does the modified feeding sequence in Patent CN101619021A improve reaction efficiency?
A: By pre-dissolving the aluminum chloride catalyst in nitrobenzene before adding the substrate, the reaction system avoids localized overheating and solidification. This ensures the catalyst remains active and recyclable within the liquid phase, allowing for a smooth reaction progression and significantly higher yields, reaching up to 96% in optimized examples.
Q: What are the specific advantages of using nitrobenzene as the solvent in this process?
A: Nitrobenzene acts as an inert solvent that does not react with the catalyst or the acetyl phenol products. Its specific solubility properties allow the reaction product to remain free outside the immediate reaction zone while keeping the catalyst dissolved, facilitating better heat transfer and easier post-reaction processing compared to solvent-free or steam distillation methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated P-Acetyl Phenol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires deep technical expertise and robust manufacturing infrastructure. As a leading CDMO and supplier in the fine chemical sector, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this advanced synthesis route to life. Our facilities are equipped with state-of-the-art reactors capable of handling the specific solvent and temperature requirements of the modified Fries rearrangement, ensuring that every batch meets stringent purity specifications. We understand that for R&D directors, consistency is key; therefore, our rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every lot of halogenated p-acetyl phenol compounds we produce. We are committed to delivering not just a chemical product, but a guarantee of quality that supports your downstream drug development timelines.
We invite procurement leaders and supply chain heads to engage with us to explore how this optimized manufacturing process can benefit your organization. By leveraging our technical capabilities, you can secure a supply of high-purity intermediates that are both cost-effective and reliably delivered. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Whether you need specific COA data to validate our quality standards or route feasibility assessments to integrate this intermediate into your existing workflow, our team is ready to provide the detailed support you need to make informed sourcing decisions. Partner with us to transform the potential of patent CN101619021A into a tangible competitive advantage for your business.
