Scalable Synthesis of Salidroside Analogues for Advanced Neuroprotective Pharmaceutical Intermediates Manufacturing
Scalable Synthesis of Salidroside Analogues for Advanced Neuroprotective Pharmaceutical Intermediates Manufacturing
The global demand for neuroprotective agents has intensified the search for sustainable sources of bioactive compounds traditionally derived from rare botanical resources. Patent CN102241710A addresses this critical supply chain vulnerability by disclosing a robust chemical synthesis method for salidroside analogues, effectively bypassing the ecological limitations of wild Rhodiola harvesting. This technology enables the production of diverse glycoside structures where R1 represents various sugar donors such as glucose, galactose, or lactose, and R2 denotes substituents like hydroxyl, halogen, or alkoxy groups on the aromatic ring. By establishing a reliable synthetic pathway, manufacturers can secure a consistent supply of high-purity intermediates essential for developing treatments for central nervous system injuries and wasting diseases. The structural versatility offered by this patent allows for the fine-tuning of pharmacokinetic properties, potentially overcoming the blood-brain barrier limitations associated with natural salidroside. 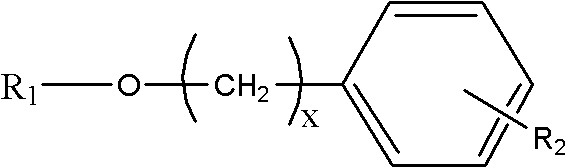
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the pharmaceutical industry has relied heavily on the extraction of salidroside from Rhodiola plants, a practice fraught with significant logistical and ecological challenges. The natural regeneration of Rhodiola in high-altitude alpine zones is exceptionally slow, typically requiring seven to eight years before the plants are suitable for harvest, leading to a precarious supply-demand imbalance. Furthermore, the extraction efficiency from dried plant material is notoriously poor, often yielding merely 0.4% to 0.8% of the dry weight, which necessitates the processing of massive quantities of biomass to obtain commercially viable amounts. This dependency on wild resources not only drives up costs due to scarcity but also raises serious sustainability concerns regarding the depletion of fragile ecosystems. Additionally, natural extracts often suffer from batch-to-batch variability in purity and potency, complicating the regulatory approval process for new drug applications that require stringent consistency.
The Novel Approach
The innovative methodology outlined in the patent data introduces a streamlined chemical synthesis route that fundamentally alters the economic and operational landscape of salidroside production. A key breakthrough involves the strategic use of acetyl groups to protect phenolic hydroxyls on the aromatic ring, which can be removed simultaneously with the acetyl groups on the sugar ring during the final deprotection step. This clever design eliminates the need for cumbersome allyl or benzyl protection groups, thereby shortening the overall reaction sequence and drastically reducing raw material consumption. The process operates under mild conditions, utilizing a low-residue aprotic mixed solvent system of dichloromethane and diethyl ether, which facilitates easier solvent recovery and minimizes environmental impact. 
Mechanistic Insights into Silver Carbonate Catalyzed Glycosylation
The core of this synthetic strategy relies on a highly efficient glycosylation reaction mediated by silver carbonate and trace iodine, which acts as a promoter for the activation of the glycosyl donor. In this mechanism, the acetyl alpha-bromide of the sugar donor reacts with the R2-substituted aromatic alcohol in the presence of anhydrous calcium sulfate, which serves as a crucial water scavenger to drive the equilibrium towards product formation. The reaction is conducted under an inert atmosphere at temperatures ranging from 15°C to 45°C, ensuring high stereoselectivity for the beta-glycosidic linkage which is critical for biological activity. The inclusion of powdered molecular sieves further enhances the reaction efficiency by adsorbing small molecules of hydrogen bromide and water, preventing side reactions that could lead to impurity formation. This precise control over reaction conditions allows for the synthesis of a wide library of analogues with varying sugar moieties and aromatic substitutions without compromising yield or purity.
Following the glycosylation step, the process employs a mild deacetylation protocol using common inorganic reagents such as potassium carbonate in methanol or ethanol. This step is pivotal as it cleaves the ester bonds on both the sugar ring and the aromatic substituent simultaneously, revealing the active hydroxyl groups necessary for the compound's pharmacological function. The purification is optimized using column chromatography with a developing solvent system of methanol and dichloromethane in a ratio of 1:6 to 1:9, which effectively separates the target analogue from unreacted starting materials and byproducts. By substituting chloroform with dichloromethane during this purification phase, the method significantly improves operator health safety while maintaining high resolution separation. The result is a high-purity crystalline product that meets the rigorous standards required for pharmaceutical intermediate applications, ready for subsequent formulation or biological testing.
How to Synthesize Salidroside Analogues Efficiently
The synthesis of these valuable neuroprotective intermediates follows a logical two-stage progression designed for scalability and reproducibility in a GMP environment. The initial stage focuses on the construction of the glycosidic bond using protected sugar donors, while the second stage involves the global deprotection to yield the final active molecule. Operators must strictly adhere to the specified temperature ranges and solvent ratios to ensure optimal stereochemical outcomes and minimize the formation of alpha-anomers. The detailed standardized synthesis steps below provide a comprehensive guide for replicating the high yields reported in the patent examples, ensuring that production teams can achieve consistent results across different batch sizes.
- Prepare the intermediate acetyl glucoside by reacting R2-substituted alcohol with acetyl alpha-bromide sugar donor using silver carbonate and iodine catalysts in aprotic solvent at 15-45°C.
- Perform deacetylation protection removal on the obtained intermediate using a strong alkaline reagent such as potassium carbonate in methanol or ethanol at 10-40°C for 2-6 hours.
- Purify the crude product using column chromatography with a methanol and dichloromethane developing solvent system to isolate the target salidroside analogue with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative benefits that extend far beyond simple cost savings, addressing fundamental vulnerabilities in the sourcing of botanical actives. By shifting from an agriculture-dependent model to a chemical manufacturing model, companies can decouple their supply from the unpredictability of weather patterns, crop diseases, and geopolitical instability in harvesting regions. This transition ensures a continuous, year-round production capability that aligns perfectly with the just-in-time inventory requirements of modern pharmaceutical manufacturing. Furthermore, the simplification of the synthetic route through the dual-function acetyl protection strategy reduces the number of unit operations, which directly correlates to lower capital expenditure on equipment and reduced labor hours per kilogram of output.
- Cost Reduction in Manufacturing: The elimination of expensive and difficult-to-remove protecting groups like benzyl or allyl ethers significantly lowers the cost of goods sold by reducing reagent expenses and purification complexity. The use of common inorganic bases like potassium carbonate instead of specialized organic bases further drives down raw material costs while simplifying waste treatment protocols. Additionally, the ability to perform reactions at room temperature or mild heating reduces energy consumption compared to processes requiring cryogenic conditions or high-pressure reactors. These cumulative efficiencies result in a substantially more competitive price point for the final intermediate, allowing for better margin management in downstream drug development.
- Enhanced Supply Chain Reliability: Synthetic production mitigates the risks associated with the slow natural regeneration of Rhodiola plants, which traditionally takes nearly a decade to replenish. By utilizing readily available chemical starting materials such as substituted benzyl alcohols and acetylated sugars, manufacturers can establish a robust supply chain that is immune to agricultural failures or seasonal fluctuations. This reliability is critical for long-term drug development projects where continuity of supply is a regulatory requirement for clinical trials and commercial launch. The flexibility to switch between different sugar donors and aromatic substituents also allows for rapid adaptation to changing market demands or specific client customization requests without retooling the entire production line.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing solvents like dichloromethane and diethyl ether that are easier to recover and recycle compared to more toxic alternatives. The replacement of chloroform with dichloromethane during chromatography not only protects worker health but also simplifies regulatory compliance regarding volatile organic compound emissions. The straightforward workup procedures, involving simple filtration and solvent evaporation, facilitate easy scale-up from laboratory grams to industrial tons without encountering complex engineering bottlenecks. This scalability ensures that the technology can support the growing global demand for neuroprotective agents as the population ages and the prevalence of neurological disorders increases.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating these intermediates into their existing pipelines. The answers provided reflect the specific advantages of the acetyl protection strategy and the operational parameters defined in the intellectual property.
Q: Why is chemical synthesis preferred over natural extraction for Salidroside?
A: Natural extraction from Rhodiola plants is limited by slow regeneration cycles of 7-8 years and low yields of only 0.4-0.8% dry weight. Chemical synthesis provides a sustainable, scalable alternative that ensures consistent supply without depleting ecological resources.
Q: What are the key advantages of the acetyl protection strategy in this patent?
A: The use of acetyl groups for protecting phenolic hydroxyls allows for simultaneous removal with sugar ring acetyl groups. This avoids complex allyl or benzyl protection steps, significantly shortening the reaction route and reducing overall production costs.
Q: How does this method improve operator safety and environmental compliance?
A: The process utilizes low-residue aprotic mixed solvents like dichloromethane and diethyl ether instead of highly toxic alternatives. Furthermore, replacing chloroform with dichloromethane during column chromatography enhances operational health safety and reduces hazardous waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Salidroside Analogues Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to translate the innovative synthesis methods described in patent CN102241710A into commercial reality. Our facilities are equipped to handle complex glycosylation chemistries with precision, ensuring that every batch of salidroside analogues meets the stringent purity specifications required for pharmaceutical applications. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our rigorous QC labs to guarantee consistency and quality at every stage of the manufacturing process. Our commitment to excellence means that we can deliver high-purity intermediates that accelerate your drug development timelines while maintaining full regulatory compliance.
We invite you to collaborate with our technical procurement team to explore how these advanced synthetic routes can optimize your supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to our synthetic salidroside analogues compared to traditional extraction methods. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique project requirements. Let us be your trusted partner in bringing next-generation neuroprotective therapies to the market with speed, efficiency, and unmatched quality assurance.
