Scalable Synthesis of Alpha Gamma-Tetrachlorobutyrate via Advanced Copper-Catalyzed ATRA Technology
The chemical industry is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthesis routes for critical intermediates. A recent technological breakthrough documented in patent CN112574037A introduces a highly optimized synthetic method for alpha, gamma-tetrachlorobutyrate, a compound of significant importance in the fields of agrochemical and polymer synthesis. This patent details a novel Atom Transfer Radical Addition (ATRA) process that utilizes a sophisticated copper-catalyst system to achieve exceptional purity and yield metrics. For R&D directors and procurement specialists, understanding the nuances of this technology is vital, as it represents a shift from traditional, cumbersome methods to a streamlined, industrial-grade protocol. The innovation lies not just in the final product quality, which reportedly reaches purity levels as high as 99.5%, but in the fundamental re-engineering of the catalytic cycle to ensure robustness and scalability. By leveraging specific copper salts combined with organic amine or imidazole ligands, the process overcomes historical solubility and activity limitations, paving the way for more reliable supply chains and reduced manufacturing overheads in the production of chlorine-containing fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha, gamma-tetrachlorobutyrate via Kharasch addition reactions has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Traditional catalytic systems often rely on inorganic copper salts which suffer from poor solubility in organic reaction media, leading to heterogeneous reaction conditions that are difficult to control and optimize. Furthermore, prior art methods frequently necessitate the use of expensive and complex ligands, such as scorpion-like copper complexes or multidentate amines, which are costly to prepare and difficult to source in bulk quantities. These legacy processes often require the addition of free radical initiators like AIBN, which introduces safety risks due to potential thermal instability and complicates the purification process by generating nitrogen-containing byproducts. The result is often a reaction mixture contaminated with high-boiling polymeric tars, low single-pass yields, and a product profile that requires extensive and energy-intensive downstream processing to meet pharmaceutical or agrochemical grade specifications. These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks for supply chain managers seeking consistent quality.
The Novel Approach
In stark contrast, the methodology outlined in CN112574037A presents a paradigm shift by employing a readily available copper salt or cuprous salt main catalyst compounded with simple organic amine or imidazole cocatalysts. This strategic combination forms a soluble Cu-N complex structure in situ, which dramatically enhances the catalytic activity and selectivity of the system without the need for exotic ligands. The process operates under mild conditions, typically between 40°C and 140°C, and eliminates the requirement for hazardous initiators or UV irradiation, thereby simplifying the operational safety profile. By optimizing the molar ratios of carbon tetrachloride to acrylate and carefully selecting the ligand environment, the reaction achieves a single-pass yield of up to 96.1% with minimal formation of polymeric byproducts. This streamlined approach not only reduces the complexity of the reaction setup but also facilitates the recovery and recycling of both the solvent and the catalyst system, offering a sustainable advantage. The ability to produce high-purity alpha, gamma-tetrachlorobutyrate with such efficiency makes this novel approach a compelling option for manufacturers looking to optimize their production lines.

Mechanistic Insights into Copper-Catalyzed Atom Transfer Radical Addition
The core of this technological advancement lies in the precise manipulation of the catalytic cycle, which governs the generation and termination of radical species during the addition reaction. The mechanism involves the formation of a stable copper-ligand complex that effectively induces the homolytic cleavage of the carbon-chlorine bond in carbon tetrachloride. This activation step generates the trichloromethyl radical, which then adds across the double bond of the acrylate substrate to form a new carbon-carbon bond. The resulting carbon-centered radical is subsequently capped by a chlorine atom transferred from the oxidized copper species, regenerating the active catalyst and completing the cycle. This controlled radical process ensures that the concentration of free radicals remains low enough to prevent uncontrolled polymerization but high enough to drive the addition reaction to completion efficiently. The use of imidazole or amine ligands is critical here, as they modulate the redox potential of the copper center, ensuring that the equilibrium between the active and dormant species is maintained throughout the reaction duration. This mechanistic control is what allows the process to achieve such high selectivity and purity, avoiding the formation of the tarry byproducts that plague less optimized systems.
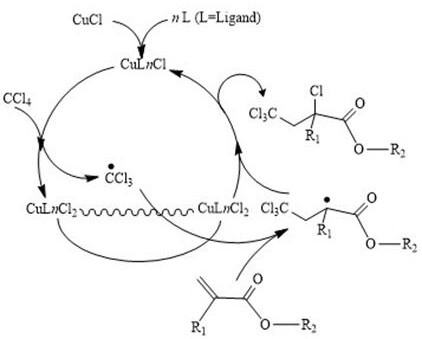
Impurity control is another critical aspect where this mechanism excels, particularly regarding the suppression of dimerization and polymerization side reactions. In conventional free radical additions, the lack of control over radical concentration often leads to the coupling of radical intermediates, resulting in dimers or high molecular weight polymers that are difficult to separate from the desired mono-addition product. The copper-catalyzed ATRA mechanism mitigates this by maintaining a dynamic equilibrium where the radical species are rapidly capped, limiting their lifetime and opportunity for side reactions. Furthermore, the specific choice of ligand and solvent system ensures that the catalyst remains homogeneous and active, preventing the localized hot spots of radical generation that can occur with heterogeneous catalysts. The result is a reaction profile that is clean and predictable, with the primary impurity being unreacted starting materials which are easily removed via distillation. This level of impurity control is essential for downstream applications in sensitive fields like agrochemical synthesis, where trace impurities can affect the efficacy and safety of the final pesticide products.
How to Synthesize Alpha, Gamma-Tetrachlorobutyrate Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalytic system and the control of reaction parameters to maximize yield and purity. The process begins with the mixing of carbon tetrachloride and the chosen acrylate substrate in a polar inert organic solvent, followed by the addition of the copper salt and the organic amine or imidazole ligand. It is crucial to ensure thorough mixing at room temperature before heating to allow for the proper formation of the active copper-ligand complex. The reaction is then conducted in a sealed reactor at elevated temperatures, typically ranging from 80°C to 120°C, for a duration of 10 to 20 hours depending on the specific substrates used. Post-reaction processing involves cooling the mixture, filtering off any insoluble materials, and then subjecting the filtrate to a two-stage distillation process. The first stage recovers excess carbon tetrachloride at atmospheric pressure, while the second stage utilizes vacuum rectification to isolate the high-purity alpha, gamma-tetrachlorobutyrate product. Detailed standardized synthesis steps see the guide below.
- Mix carbon tetrachloride and acrylate with a copper salt catalyst and organic amine ligand in a polar solvent.
- Heat the mixture to 40-140°C in a sealed reactor to facilitate the addition reaction.
- Cool, filter, and purify the product via atmospheric and vacuum distillation to recover solvents and isolate the ester.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers substantial benefits for procurement managers and supply chain heads who are tasked with reducing costs and ensuring material availability. The primary advantage stems from the use of cheap and readily available raw materials, including common copper salts and simple organic amines, which eliminates the dependency on expensive, specialized catalysts that can strain budgets and lead times. The high single-pass yield and the ability to recover and recycle the solvent and catalyst significantly reduce the raw material consumption per unit of product, leading to a drastic simplification of the cost structure. Furthermore, the mild reaction conditions and the absence of hazardous initiators lower the operational risks and energy requirements associated with the manufacturing process, contributing to overall cost reduction in agrochemical intermediate manufacturing. These factors combine to create a more resilient supply chain that is less susceptible to fluctuations in raw material prices and regulatory changes regarding hazardous chemicals.
- Cost Reduction in Manufacturing: The elimination of expensive ligands and initiators, coupled with the high efficiency of the copper-amine catalyst system, directly translates to lower production costs. The ability to recycle the catalyst and solvent further enhances economic viability by minimizing waste disposal fees and raw material procurement expenses. This process optimization allows manufacturers to offer competitive pricing without compromising on the quality or purity of the final intermediate, providing a significant margin advantage in the market.
- Enhanced Supply Chain Reliability: By utilizing commodity chemicals such as copper chloride and acetonitrile, the process reduces the risk of supply disruptions associated with specialty reagents. The robustness of the reaction conditions ensures consistent output quality, which is critical for maintaining long-term contracts with downstream pharmaceutical and agrochemical clients. This reliability simplifies inventory management and reduces the need for safety stock, allowing for a more lean and responsive supply chain operation that can adapt quickly to market demands.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, featuring simple unit operations like distillation and filtration that are easily replicated in large-scale reactors. The efficient recovery of carbon tetrachloride addresses environmental concerns regarding ozone-depleting substances by preventing release into the atmosphere, ensuring compliance with international environmental protocols. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer, appealing to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for making informed decisions about adopting this method for commercial production or integrating it into existing R&D pipelines. The insights provided here cover catalyst performance, environmental impact, and scalability, offering a comprehensive overview of the technology's value proposition.
Q: What are the advantages of the copper-amine catalyst system over traditional methods?
A: The copper-amine catalyst system offers superior solubility in organic solvents compared to inorganic salts alone, leading to higher catalytic efficiency, fewer byproducts like tar, and easier catalyst recovery, which significantly lowers production costs.
Q: How does this process handle environmental concerns regarding carbon tetrachloride?
A: The process utilizes excess carbon tetrachloride which is recovered via atmospheric distillation after the reaction, allowing it to be recycled for subsequent batches, thereby minimizing waste discharge and aligning with environmental regulations.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method operates under mild conditions without requiring expensive initiators like AIBN or UV light, uses cheap and readily available raw materials, and achieves high single-pass yields, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha, Gamma-Tetrachlorobutyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation agrochemicals and polymers. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative synthesis routes like the one described in CN112574037A can be successfully translated into industrial reality. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of alpha, gamma-tetrachlorobutyrate meets the exacting standards required by global pharmaceutical and chemical companies. We are committed to providing a seamless partnership that combines technical expertise with reliable manufacturing capacity to support your product development goals.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through advanced chemical synthesis. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your production efficiency and product quality. Together, we can drive innovation and efficiency in the fine chemical industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
