Scalable Synthesis of 3,3'-Diindole Derivatives via Iron-Catalyzed Oxidative Coupling for Commercial API Production
Scalable Synthesis of 3,3'-Diindole Derivatives via Iron-Catalyzed Oxidative Coupling for Commercial API Production
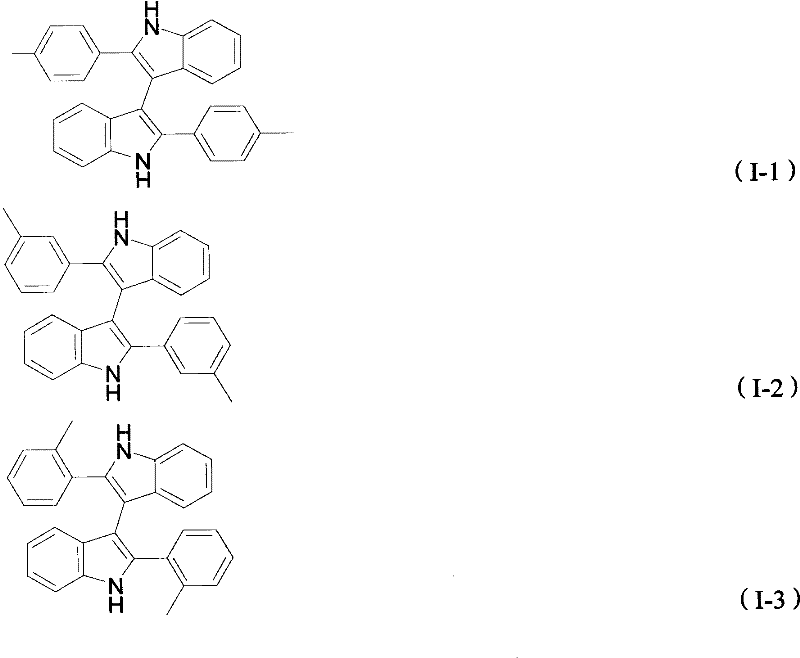
The pharmaceutical and fine chemical industries are constantly seeking robust, environmentally benign pathways to access complex heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in patent CN101935297A, which discloses a novel and efficient preparation method for 3,3'-diindole derivatives. These compounds are not merely academic curiosities; they represent a vital class of organic synthesis intermediates known for their potent biological anticancer activity and their utility as chiral catalysts in asymmetric synthesis. The patent highlights a paradigm shift from traditional, waste-intensive methods to a greener, iron-catalyzed oxidative coupling strategy. By leveraging the abundant and biocompatible nature of iron, this technology offers a sustainable alternative that aligns with modern green chemistry principles while delivering high-purity products essential for downstream drug development. The structural diversity achievable through this method is substantial, allowing for the introduction of various functional groups such as alkyl, alkoxy, halogen, and trifluoromethyl moieties, thereby expanding the chemical space available for medicinal chemists.
For procurement managers and supply chain directors, the implications of adopting this technology are profound. The reliance on readily available raw materials and the elimination of precious metal catalysts directly translate to reduced cost of goods sold (COGS) and enhanced supply chain resilience. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such proprietary synthetic routes is crucial for ensuring continuity of supply and maintaining competitive pricing structures in the global market. The ability to produce high-purity 3,3'-diindole derivatives at scale without the baggage of hazardous reagents positions manufacturers to meet stringent regulatory requirements more effectively.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,3'-diindole derivatives has been plagued by significant operational and environmental challenges that hindered their widespread commercial adoption. Traditional literature methods, such as those reported by Desarbre et al., typically involve a multi-step sequence starting with the nucleophilic reaction of indole with isatin to generate 3-(indolyl)-3-hydroxyindole, followed by a reduction step using sodium borohydride. This approach is inherently inefficient due to the sheer number of unit operations required, each introducing potential yield losses and increasing the overall processing time. Furthermore, the use of stoichiometric amounts of reducing agents generates substantial chemical waste, posing serious environmental pollution concerns that conflict with modern sustainability goals. Another prevalent method involves Suzuki-Miyaura coupling reactions, as described by Duong et al., which necessitates a preliminary bromination step using strong corrosive reagents like liquid bromine. The handling of liquid bromine requires specialized equipment and rigorous safety protocols due to its high toxicity and corrosivity, driving up capital expenditure and operational costs. Additionally, the reliance on palladium catalysts in Suzuki coupling introduces the risk of heavy metal contamination in the final product, necessitating expensive and time-consuming purification steps to meet pharmaceutical purity specifications.
The Novel Approach
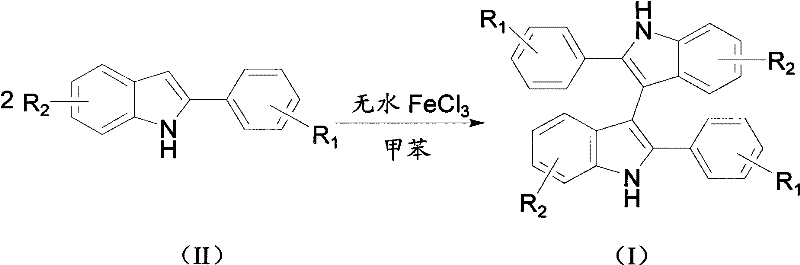
In stark contrast to these legacy processes, the method disclosed in CN101935297A presents a streamlined, direct oxidative coupling pathway that fundamentally reshapes the manufacturing landscape for these intermediates. This novel approach utilizes 2-arylindole compounds as the sole starting materials, which undergo direct dimerization in the presence of anhydrous ferric chloride (FeCl3) acting as a catalyst. The reaction is conducted in toluene under an oxygen atmosphere, eliminating the need for hazardous halogenation reagents or expensive transition metals like palladium. The simplicity of the reagent system—comprising essentially just the substrate, an iron salt, and a common organic solvent—drastically reduces the complexity of the supply chain and minimizes the inventory of dangerous chemicals required on-site. Moreover, the reaction conditions are remarkably mild, operating effectively within a temperature range of 90°C to 150°C, which reduces energy consumption compared to high-temperature pyrolytic methods. The use of oxygen as the terminal oxidant is particularly advantageous from an environmental perspective, as the only byproduct is water, rendering the entire synthesis route exceptionally clean and environmentally friendly. This direct coupling strategy not only shortens the production timeline but also enhances the overall atom economy, making it a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Coupling
The core of this technological advancement lies in the mechanistic efficiency of the iron-catalyzed oxidative coupling. Anhydrous ferric chloride serves as a Lewis acid and a single-electron oxidant, facilitating the generation of radical intermediates from the 2-arylindole substrate. Under the protection of an oxygen atmosphere, the iron catalyst promotes the selective formation of a carbon-carbon bond at the 3-position of the indole ring, leading to the dimeric 3,3'-diindole structure. The regioselectivity of this reaction is a critical feature, as it avoids the formation of unwanted regioisomers that often complicate purification in electrophilic aromatic substitution reactions. The presence of oxygen ensures the regeneration of the active catalytic species and drives the equilibrium towards the product, preventing the accumulation of partially oxidized byproducts. This mechanistic pathway is robust enough to tolerate a wide array of electronic environments on the aryl ring, whether electron-donating groups like methyl and methoxy or electron-withdrawing groups like halogens and trifluoromethyl are present. The stability of the iron catalyst under these thermal conditions ensures consistent performance throughout the reaction duration, which typically spans 15 to 25 hours depending on the specific substrate reactivity.
From an impurity control perspective, this mechanism offers distinct advantages over traditional methods. The absence of halogenation steps means there is no risk of poly-halogenated impurities or residual bromine/chlorine contaminants that are notoriously difficult to remove. Furthermore, the mild reaction conditions minimize thermal degradation of the sensitive indole scaffold, preserving the integrity of the molecular structure. The high selectivity observed in this process results in a cleaner crude reaction mixture, which simplifies the downstream purification process. Instead of requiring complex recrystallization sequences or preparative HPLC to remove heavy metal residues or halogenated side products, the crude product can often be purified effectively using standard column chromatography with common eluent systems like petroleum ether and ethyl acetate. This ease of purification is a key factor in achieving the high purity specifications required for pharmaceutical applications, ensuring that the final API intermediate meets rigorous quality standards without excessive processing costs.
How to Synthesize 3,3'-Diindole Derivatives Efficiently
Implementing this synthesis route in a production environment requires careful attention to reaction parameters to maximize yield and reproducibility. The process begins with the precise weighing of the 2-arylindole compound and anhydrous ferric chloride, which are then introduced into a reaction vessel containing toluene. The molar ratio of the substrate to the catalyst is a critical variable, typically optimized between 1:0.1 and 1:0.5 to balance catalytic activity with cost efficiency. The reaction mixture is stirred vigorously to ensure homogeneous suspension of the catalyst and efficient mass transfer of oxygen into the liquid phase. Heating is applied to reach the target temperature, usually between 90°C and 120°C for most substrates, although some less reactive variants may require temperatures up to 150°C. The reaction progress is monitored until completion, after which the solvent is removed under reduced pressure. The resulting crude solid is then subjected to purification, typically via silica gel column chromatography, to isolate the pure 3,3'-diindole derivative. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this process.
- Charge a reaction vessel with 2-arylindole compound and anhydrous ferric chloride in toluene solvent under stirring conditions.
- Maintain the reaction mixture under oxygen protection and heat to a temperature range of 90°C to 150°C until completion.
- Remove the solvent via rotary evaporation and purify the crude product using column chromatography to obtain the final 3,3'-diindole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed synthesis route offers compelling strategic benefits that extend beyond simple chemical transformation. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for liquid bromine, isatin, and palladium catalysts, companies can reduce their exposure to volatile commodity markets for precious metals and hazardous chemicals. This simplification leads to substantial cost savings in raw material procurement and reduces the administrative burden associated with managing hazardous substance inventories. Furthermore, the use of toluene, a widely available and easily recyclable solvent, enhances the sustainability profile of the manufacturing process, aligning with corporate social responsibility goals and reducing waste disposal costs. The robustness of the reaction conditions also implies a lower risk of batch failures due to sensitive reagent degradation, thereby improving overall supply reliability.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with inexpensive ferric chloride represents a direct and significant reduction in catalyst costs. Additionally, the elimination of the bromination step removes the need for corrosion-resistant reactors and specialized safety infrastructure, lowering capital expenditure. The simplified workup procedure reduces labor hours and solvent consumption during purification, contributing to a leaner manufacturing cost structure. These cumulative efficiencies allow for a more competitive pricing model for the final intermediate, providing a clear margin advantage in the marketplace.
- Enhanced Supply Chain Reliability: The starting materials, specifically 2-arylindole compounds, are readily available from multiple commercial sources, reducing the risk of single-source dependency. The stability of the reagents under ambient storage conditions further mitigates supply chain disruptions caused by shelf-life limitations. The straightforward nature of the reaction setup allows for flexible production scheduling, enabling manufacturers to respond quickly to fluctuations in demand without lengthy changeover times. This agility is crucial for maintaining continuous supply to downstream API manufacturers who operate on tight just-in-time delivery schedules.
- Scalability and Environmental Compliance: The reaction's compatibility with standard glass-lined or stainless steel reactors facilitates seamless scale-up from laboratory to commercial production volumes. The use of oxygen as a green oxidant minimizes the generation of toxic waste streams, simplifying compliance with increasingly stringent environmental regulations. The reduced E-factor (mass of waste per mass of product) associated with this method lowers waste treatment costs and enhances the overall environmental footprint of the facility. This alignment with green chemistry principles not only ensures regulatory compliance but also strengthens the brand reputation of the manufacturer as a sustainable partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3,3'-diindole derivatives using this novel methodology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their specific drug development pipelines or manufacturing processes. The answers reflect the practical realities of scaling this chemistry while maintaining high quality standards.
Q: What are the primary advantages of this FeCl3-catalyzed method over traditional Suzuki coupling?
A: Unlike traditional Suzuki-Miyaura coupling which often requires corrosive liquid bromine for halogenation and expensive palladium catalysts, this novel method utilizes inexpensive and biocompatible iron catalysts. It eliminates the need for hazardous halogenation steps, significantly reducing environmental pollution and raw material costs while maintaining high reaction selectivity.
Q: What is the typical reaction condition profile for this oxidative coupling?
A: The reaction operates under mild to moderate thermal conditions, typically between 90°C and 150°C, using toluene as the solvent. The process requires an oxygen atmosphere to facilitate the oxidative dimerization, avoiding the need for complex inert gas setups beyond standard oxygen protection, which simplifies the operational protocol for large-scale manufacturing.
Q: Can this method accommodate diverse substituents on the indole ring?
A: Yes, the method demonstrates excellent substrate tolerance. It successfully synthesizes derivatives with various substituents including alkyl groups (methyl, ethyl, butyl), alkoxy groups, halogens (fluorine, chlorine, bromine, iodine), and trifluoromethyl groups at different positions on the phenyl ring, making it highly versatile for generating diverse pharmaceutical intermediate libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Diindole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical innovation. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering 3,3'-diindole derivatives with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our expertise in iron-catalyzed transformations allows us to optimize this specific route for maximum yield and minimal environmental impact, providing our clients with a sustainable and cost-effective supply solution.
We invite you to collaborate with us to explore how this advanced synthesis technology can benefit your projects. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your quality and supply chain objectives. Let us be your partner in bringing high-purity pharmaceutical intermediates to market faster and more efficiently.
