Scalable Production of Advanced Photosensitive Dyes for Purple Laser Plate Making
Scalable Production of Advanced Photosensitive Dyes for Purple Laser Plate Making
The rapid evolution of the printing industry towards high-speed, high-precision purple laser computer plate making technology has created an urgent demand for advanced photosensitive materials with superior thermal stability and sensitivity. Patent CN102464626A introduces a groundbreaking preparation method for 5-(4-(N,N-diphenylamino)benzylidene)-3-(2-phenylethyl)-2,4-oxazolidinedione, a critical photosensitive dye component used in the photopolymerization layers of laser printing plates. This innovation addresses the severe safety hazards and operational complexities inherent in traditional synthesis routes, offering a robust pathway for industrial scale-up. By replacing hazardous reagents like phosgene and sodium azide with safer catalytic systems involving DBU and copper powder, this technology not only enhances operator safety but also streamlines the manufacturing workflow, making it an ideal candidate for reliable photosensitive dye supplier partnerships aiming to secure the electronic chemical manufacturing supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key isocyanate intermediates required for this class of oxazolidinedione dyes relied heavily on extremely dangerous and operationally difficult chemistries. One prevalent method involved the reaction of 2-phenylethylamine with phosgene, a process fraught with peril due to the hypertoxic nature of phosgene gas, necessitating massive equipment investments for containment and scrubbing systems that are often prohibitive for many manufacturers. 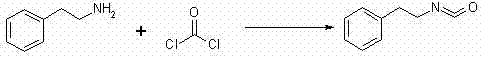 Alternative routes utilizing metal-catalyzed high-temperature pyrolysis or decomposition with phosphorus pentachloride and POCl3 suffered from significant side reactions, including the polymerization of the isocyanate intermediate under harsh Lewis acid conditions, leading to critically low yields and difficult post-reaction metal removal. Furthermore, other literature methods employing sodium azide introduced explosive risks and the generation of pungent ammonia gas, while routes utilizing n-butyl lithium required cryogenic conditions at -78°C, driving up energy costs and complicating reactor design with expensive low-temperature infrastructure.
Alternative routes utilizing metal-catalyzed high-temperature pyrolysis or decomposition with phosphorus pentachloride and POCl3 suffered from significant side reactions, including the polymerization of the isocyanate intermediate under harsh Lewis acid conditions, leading to critically low yields and difficult post-reaction metal removal. Furthermore, other literature methods employing sodium azide introduced explosive risks and the generation of pungent ammonia gas, while routes utilizing n-butyl lithium required cryogenic conditions at -78°C, driving up energy costs and complicating reactor design with expensive low-temperature infrastructure.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a mild, four-step sequence that begins with a DBU-catalyzed rearrangement of hydrocinnamamide, completely bypassing the need for phosgene or explosive azides. This modern synthetic strategy leverages the unique ability of DBU to facilitate rearrangement under ambient to moderate temperatures, followed by a clean cyclization with ethyl glycolate and a subsequent Knoevenagel condensation. 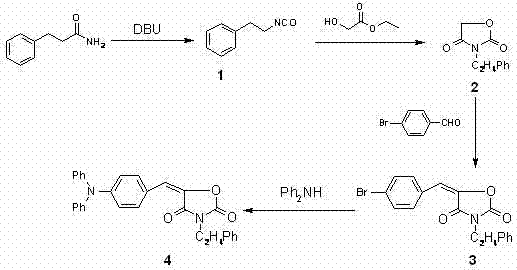 The final Ullmann coupling step employs copper powder and valeric acid in polar aprotic solvents, avoiding the use of expensive palladium catalysts often seen in similar cross-coupling reactions. This comprehensive route achieves a total product yield of approximately 65%, with individual step yields ranging from 80% to 95%, demonstrating a level of efficiency and operational simplicity that renders it highly suitable for industrialization and cost reduction in electronic chemical manufacturing.
The final Ullmann coupling step employs copper powder and valeric acid in polar aprotic solvents, avoiding the use of expensive palladium catalysts often seen in similar cross-coupling reactions. This comprehensive route achieves a total product yield of approximately 65%, with individual step yields ranging from 80% to 95%, demonstrating a level of efficiency and operational simplicity that renders it highly suitable for industrialization and cost reduction in electronic chemical manufacturing.
Mechanistic Insights into DBU-Catalyzed Rearrangement and Cyclization
The cornerstone of this improved synthesis lies in the initial transformation of hydrocinnamamide into 2-phenethyl isocyanate using 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) in the presence of Raney-Ni. Mechanistically, DBU acts as a potent non-nucleophilic base that facilitates the dehydration and rearrangement of the primary amide without the aggressive electrophilicity associated with phosgene. The presence of Raney-Ni likely assists in stabilizing the transition state or scavenging trace impurities that could lead to urea formation, ensuring a clean conversion with yields reaching up to 95%. This step is critical because it establishes the isocyanate functionality required for ring closure while avoiding the formation of polymeric byproducts that typically plague high-temperature isocyanate syntheses.
Following the formation of the isocyanate, the mechanism proceeds through a nucleophilic attack by the alkoxide generated from sodium methylate on the carbonyl of ethyl glycolate, followed by intramolecular cyclization to form the 2,4-oxazolidinedione core. The use of non-polar solvents like toluene or petroleum ether during this stage helps to drive the equilibrium towards the cyclic product by removing methanol via a water trap, a clever engineering solution that pushes the reaction to completion without requiring excessive reagent excess. The subsequent Knoevenagel condensation and Ullmann coupling are similarly optimized; the use of DMAP or DBU in pyridine for the condensation ensures high stereoselectivity for the benzylidene double bond, while the copper-mediated amination allows for the introduction of the bulky diphenylamino group under relatively mild thermal conditions (80-140°C), preserving the integrity of the sensitive oxazolidinedione ring.
How to Synthesize 5-(4-(N,N-diphenylamino)benzylidene)-3-(2-phenylethyl)-2,4-oxazolidinedione Efficiently
The synthesis of this high-value photosensitive dye is achieved through a streamlined four-step protocol that prioritizes safety and yield. The process begins with the rearrangement of hydrocinnamamide in DMF using DBU and Raney-Ni, followed by cyclization with ethyl glycolate under sodium alkoxide catalysis. The resulting intermediate is then condensed with 4-bromobenzaldehyde and finally coupled with diphenylamine via a modified Ullmann reaction. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below.
- Rearrange hydrocinnamamide using DBU and Raney-Ni to form 2-phenethyl isocyanate.
- Cyclize the isocyanate with ethyl glycolate using sodium alkoxide to form the oxazolidinedione core.
- Perform Knoevenagel condensation with 4-bromobenzaldehyde followed by Ullmann coupling with diphenylamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers transformative benefits that extend far beyond simple chemical yield improvements. By fundamentally altering the reagent profile from hazardous gases and cryogenic liquids to stable solids and liquids, the process drastically simplifies the regulatory compliance landscape and reduces the overhead costs associated with hazardous material storage and handling. The elimination of phosgene and sodium azide removes the need for specialized containment facilities and emergency response protocols, thereby lowering the barrier to entry for production and ensuring a more resilient supply chain that is less susceptible to shutdowns caused by safety inspections or regulatory changes.
- Cost Reduction in Manufacturing: The replacement of expensive and difficult-to-handle reagents like n-butyl lithium and phosgene with commodity chemicals such as DBU and copper powder results in substantial raw material cost savings. Furthermore, the ability to operate at near-ambient or moderate temperatures (60-100°C) rather than cryogenic conditions (-78°C) significantly reduces energy consumption for cooling, while the simplified purification via recrystallization instead of column chromatography lowers solvent usage and waste disposal costs, collectively driving down the cost of goods sold.
- Enhanced Supply Chain Reliability: The reliance on widely available, non-restricted starting materials like hydrocinnamamide and diphenylamine ensures a stable supply of inputs, mitigating the risk of shortages often associated with highly regulated precursors like phosgene. The robustness of the reaction conditions, which tolerate minor variations in temperature and mixing without significant yield loss, enhances batch-to-batch consistency, allowing for predictable production schedules and reliable delivery timelines for downstream laser plate manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard stainless steel reactors without the need for exotic alloys required for corrosive phosphorus halides. The reduction in toxic byproducts and the avoidance of heavy metal catalysts like palladium simplify wastewater treatment and solid waste management, aligning with increasingly stringent global environmental regulations and supporting the company's sustainability goals without compromising production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this photosensitive dye intermediate. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers evaluating this technology for integration into their manufacturing portfolios.
Q: Why is the DBU-catalyzed rearrangement preferred over phosgene methods?
A: The DBU-catalyzed method eliminates the need for highly toxic phosgene gas, significantly reducing equipment investment and safety risks associated with large-scale production, while maintaining high yields of 85-95%.
Q: How does this process improve supply chain reliability for laser plate manufacturers?
A: By avoiding explosive reagents like sodium azide and expensive cryogenic conditions (-78°C) required by n-butyl lithium, the process ensures consistent batch-to-batch availability and reduces dependency on specialized hazardous material logistics.
Q: What are the purity specifications achievable with this synthetic route?
A: The mild reaction conditions and simple recrystallization purification steps allow for the production of high-purity intermediates suitable for sensitive photopolymerization layers, minimizing impurities that could affect laser sensitivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(4-(N,N-diphenylamino)benzylidene)-3-(2-phenylethyl)-2,4-oxazolidinedione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance photosensitive dyes play in the next generation of purple laser plate making technologies. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 5-(4-(N,N-diphenylamino)benzylidene)-3-(2-phenylethyl)-2,4-oxazolidinedione performs consistently in your photopolymerization formulations.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific application needs. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your product quality while optimizing your overall supply chain costs.
