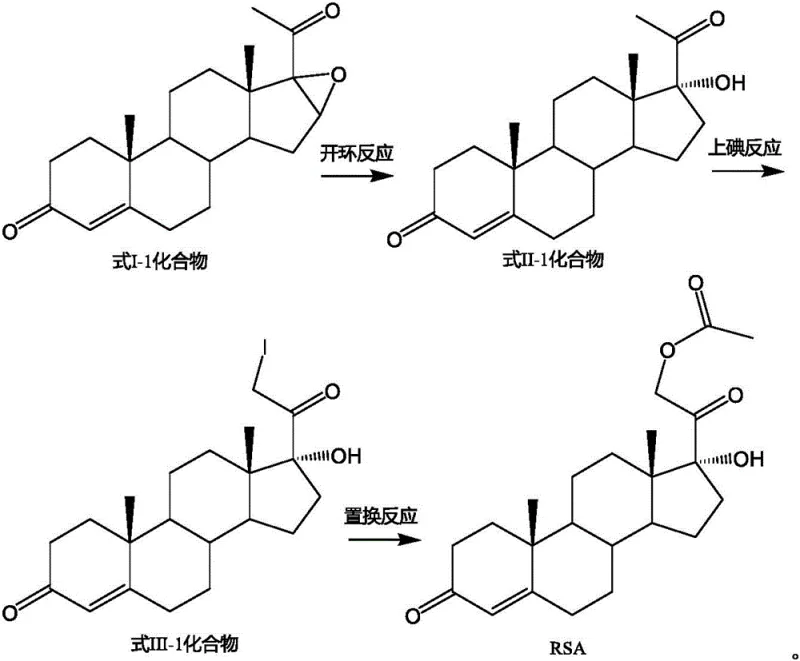
Advanced Synthesis of 21-Position-Iodo-Steroid Compounds for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical corticosteroid intermediates, particularly those leading to hydrocortisone and its derivatives. Patent CN116143857A introduces a transformative preparation method for 21-position-iodo-steroid compounds, addressing long-standing inefficiencies in steroid functionalization. This innovation centers on a novel iodination strategy that employs specific metal oxides to modulate reaction kinetics, thereby enhancing regioselectivity and minimizing byproduct formation. For R&D directors and procurement specialists, this technology represents a significant leap forward in the reliable supply of high-purity pharmaceutical intermediates. The method not only streamlines the synthesis of Pregna-4-ene-17-hydroxy-3, 20-dione-21-acetate (RSA) but also aligns with modern green chemistry principles by reducing hazardous waste. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic advancements is crucial for evaluating long-term process viability and cost structures in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key steroid intermediates like RSA has been plagued by cumbersome multi-step sequences involving harsh reagents and expensive catalysts. Traditional pathways often rely on the ring-opening of 16,17-epoxy steroids using large excesses of acetic acid and hydrogen bromide, which imposes severe corrosion requirements on production equipment and generates substantial acidic wastewater. Furthermore, subsequent debromination steps typically necessitate the use of precious metal catalysts such as palladium on carbon, which introduces significant cost volatility and supply chain risks associated with precious metal recovery. In the critical iodination stage, conventional methods utilizing calcium oxide and elemental iodine frequently suffer from poor atom economy, leading to excessive consumption of iodine and the formation of difficult-to-remove 21,21-diiodo impurities. These inefficiencies collectively inflate the cost reduction in API manufacturing and complicate the purification processes required to meet stringent pharmacopeial standards.
The Novel Approach
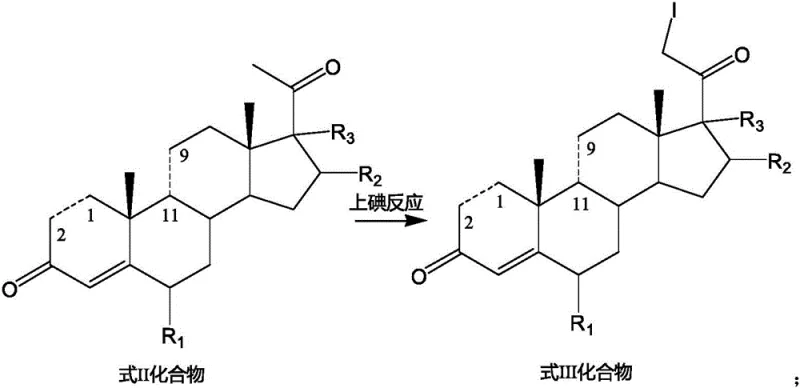
The disclosed invention fundamentally reengineers this synthetic landscape by introducing a metal oxide-mediated iodination protocol that bypasses the need for expensive transition metal catalysts and corrosive acid conditions. By selecting from a group of metal oxides including copper oxide, ferric oxide, ferroferric oxide, zinc oxide, or aluminum oxide, the process achieves a highly controlled electrophilic substitution at the C21 position. This approach effectively improves the utilization rate of elemental iodine, ensuring that the stoichiometric ratio can be tightly controlled between 0.75:1 and 0.9:1 relative to the substrate. The result is a drastic simplification of the post-treatment workflow, as the heterogeneous nature of the metal oxide catalyst facilitates easy filtration and separation. This novel route not only enhances the chemical purity of the intermediate but also significantly lowers the environmental footprint, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Metal Oxide-Catalyzed Iodination
The core innovation lies in the synergistic interaction between the elemental iodine and the selected metal oxide surface, which acts as a Lewis acid promoter and a scavenger for acidic byproducts. During the reaction, the metal oxide likely facilitates the generation of a more reactive iodine species while simultaneously neutralizing hydrogen iodide formed during the substitution, thus preventing acid-catalyzed degradation of the sensitive steroid backbone. This mechanistic pathway ensures that the reaction proceeds smoothly at mild temperatures ranging from 20°C to 80°C, avoiding the thermal stress that can lead to epimerization or double bond migration. The suppression of the 21-position diiodide impurity is particularly critical, as this byproduct is structurally similar and difficult to separate via standard crystallization; the metal oxide system inherently suppresses over-iodination, yielding a crude product with significantly higher purity profiles.
From an impurity control perspective, this method offers a distinct advantage by minimizing the introduction of metallic contaminants that are common with homogeneous catalysts. The use of earth-abundant metal oxides ensures that residual metal levels in the final active pharmaceutical ingredient remain well within regulatory limits without requiring extensive chelation or purification steps. The reaction solvent system, which can include methanol, ethanol, isopropanol, or acetonitrile, is chosen to optimize the solubility of the steroid substrate while maintaining the stability of the iodine complex. This level of control over the reaction environment allows for consistent batch-to-batch reproducibility, a key metric for supply chain heads who prioritize reducing lead time for high-purity steroid intermediates. The robustness of this chemistry underpins the feasibility of transferring the process from laboratory scale to multi-ton production without losing yield or selectivity.
How to Synthesize 21-Position-Iodo-Steroid Compound Efficiently
The implementation of this synthesis route requires careful attention to the preparation of the starting material and the precise control of reaction parameters during the iodination step. The process begins with the ring-opening of the epoxy-steroid precursor using a strong non-nucleophilic base, followed by the critical metal oxide-catalyzed iodination which defines the success of the entire sequence. Operators must ensure that the molar ratio of the metal oxide to the substrate is maintained between 1.0:1 and 3.0:1 to achieve optimal catalytic activity without excessive solid waste. The detailed standardized synthesis steps, including specific workup procedures and crystallization conditions to maximize recovery, are outlined in the technical guide below for process engineers.
- Prepare the substrate compound of Formula II-1 via ring-opening of the epoxy-steroid precursor using a strong alkali reagent like NaHMDS in THF.
- Conduct the iodination reaction by treating the Formula II-1 substrate with elemental iodine and a selected metal oxide (e.g., CuO, Fe2O3) in an alcohol solvent at 20-80°C.
- Perform the final substitution reaction by reacting the resulting 21-iodo intermediate with potassium acetate to yield the target RSA intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal oxide-catalyzed process translates into tangible strategic advantages regarding cost stability and operational continuity. By eliminating the dependency on volatile precious metal catalysts and reducing the consumption of high-cost elemental iodine through improved utilization rates, the overall cost of goods sold (COGS) for the intermediate is substantially optimized. The simplification of the workflow, which merges multiple functional transformations into a more direct sequence, reduces the number of unit operations required, thereby lowering labor costs and energy consumption per kilogram of product. Furthermore, the reduction in wastewater volume and the elimination of corrosive acid streams decrease the overhead associated with environmental compliance and waste disposal, contributing to significant cost savings in pharmaceutical intermediates manufacturing.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with inexpensive metal oxides like copper or iron oxide removes a major variable cost driver from the production budget. Additionally, the high selectivity of the reaction minimizes the loss of valuable steroid starting materials to diiodo-byproducts, effectively increasing the overall mass yield of the process. This efficiency gain means that less raw material is required to produce the same amount of finished intermediate, directly impacting the bottom line. The ability to recycle solvents and the ease of catalyst separation further contribute to a leaner manufacturing model that is resilient against raw material price fluctuations.
- Enhanced Supply Chain Reliability: Sourcing precious metal catalysts can often be a bottleneck due to geopolitical factors and limited mining outputs; shifting to abundant metal oxides secures the supply chain against such disruptions. The robustness of the reaction conditions allows for manufacturing in a wider range of facilities without requiring specialized corrosion-resistant equipment, increasing the pool of potential contract manufacturing organizations. This flexibility ensures that production schedules can be maintained even if one facility faces downtime, thereby guaranteeing a continuous flow of high-purity steroid intermediates to downstream API synthesis sites.
- Scalability and Environmental Compliance: The mild reaction temperatures and ambient pressure conditions make this process inherently safer and easier to scale from pilot plants to full commercial production. The significant reduction in hazardous waste generation aligns with increasingly strict global environmental regulations, reducing the risk of production stoppages due to compliance issues. This environmental compatibility not only protects the company's reputation but also future-proofs the manufacturing asset against tightening regulatory frameworks, ensuring long-term viability for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing production lines.
Q: How does the metal oxide catalyst improve iodine utilization compared to traditional methods?
A: The use of metal oxides such as copper oxide or ferric oxide effectively regulates the release of active iodine species, significantly reducing the formation of 21-position diiodide byproducts and minimizing iodine waste in the mother liquor.
Q: What are the environmental benefits of this new preparation method?
A: This process eliminates the need for large volumes of acetic acid and hydrogen bromide used in conventional ring-opening steps, drastically reducing wastewater generation and the burden on downstream waste treatment facilities.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the reaction conditions are mild (20-80°C) and utilize inexpensive, readily available metal oxides, making the process highly scalable and economically viable for commercial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 21-Position-Iodo-Steroid Compound Supplier
The technological advancements detailed in patent CN116143857A underscore the potential for more efficient and sustainable production of critical steroid intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards. We understand the critical nature of these intermediates in the global supply chain for corticosteroids and are dedicated to providing a stable, high-quality supply.
We invite procurement leaders and technical directors to collaborate with us to optimize their supply chains using this advanced chemistry. By requesting a Customized Cost-Saving Analysis, you can evaluate the specific economic benefits of switching to this metal oxide-catalyzed route for your projects. Please contact our technical procurement team to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a competitive advantage through superior chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
