Revolutionizing Chiral Synthesis: Microbial Production of High-Purity (S)-Phenylglycol
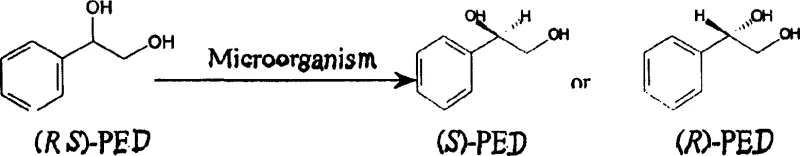
The pharmaceutical and fine chemical industries are constantly seeking more efficient, environmentally benign, and cost-effective methods for producing chiral intermediates, which are the building blocks of countless active pharmaceutical ingredients and advanced functional materials. Patent CN1212403C introduces a groundbreaking approach to this challenge by detailing a method for preparing optical pure phenylethanediol, specifically (S)-phenylglycol, utilizing microbial stereoselectivity transformation. This technology represents a significant leap forward from traditional chemical resolution methods, leveraging the inherent chirality of specific microbial strains to achieve high enantiomeric excess without the need for toxic heavy metal catalysts or complex synthetic protection groups. The patent identifies two specific strains, Candida parapsilosis (CCTCC M203011) and Bacillus polymyxa (CCTCC M203010), which have been screened and optimized to asymmetrically convert racemic phenylethanediol into the desired (S)-enantiomer with exceptional purity levels ranging from 90% to 100% e.e. This biological pathway not only simplifies the production workflow but also aligns with the growing global demand for green chemistry solutions that minimize environmental impact while maximizing yield and product quality for high-value applications in liquid crystal materials and optically active medicines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optical pure phenylglycol has relied heavily on chemical resolution methods, chromatographic separation, or enzymatic processes that often suffer from significant drawbacks in terms of cost, complexity, and environmental safety. Chemical resolution typically requires the use of stoichiometric amounts of chiral resolving agents, which are often expensive and difficult to recover, leading to a theoretical maximum yield of only 50% unless dynamic kinetic resolution is employed, which adds further complexity. Furthermore, traditional chemical synthesis often necessitates the selective protection of hydroxyl groups to prevent side reactions, introducing additional synthetic steps that consume time, energy, and raw materials while generating substantial chemical waste. The use of chiral catalysts in chemical methods can also introduce toxicity concerns, as many effective catalysts contain heavy metals that must be rigorously removed from the final product to meet stringent pharmaceutical safety standards, thereby increasing downstream processing costs. Chromatographic methods, while effective for small-scale purification, are often prohibitively expensive for large-scale industrial production due to the high cost of chiral stationary phases and the low throughput associated with column chromatography, making them unsuitable for the mass production required by the global supply chain.
The Novel Approach
In stark contrast to these conventional limitations, the novel approach described in the patent utilizes whole-cell biocatalysis to achieve asymmetric conversion with superior efficiency and selectivity. By employing specific microbial strains such as Candida parapsilosis, the process leverages the natural stereoselectivity of intracellular enzymes to distinguish between enantiomers in a racemic mixture, effectively converting the unwanted (R)-isomer into the desired (S)-isomer or selectively oxidizing it to an intermediate that is subsequently reduced. This biological system operates under mild reaction conditions, typically at temperatures between 20°C and 40°C and near-neutral pH levels, which significantly reduces energy consumption compared to the high temperatures and pressures often required in chemical synthesis. The use of whole cells eliminates the need for expensive enzyme purification steps, as the microbial cell wall provides a natural barrier and stabilizing environment for the catalytic enzymes, enhancing their operational stability and reusability. Moreover, this method avoids the use of toxic organic solvents and heavy metal catalysts, resulting in a cleaner reaction profile that simplifies product isolation and reduces the environmental burden associated with waste disposal, making it an ideal candidate for sustainable manufacturing practices in the fine chemical sector.
Mechanistic Insights into Microbial Stereoselective Transformation
The core of this technology lies in the intricate biochemical pathways employed by the selected microbial strains to achieve high enantiomeric excess. For Candida parapsilosis CCTCC M203011, the transformation mechanism involves a sophisticated two-step redox process that effectively recycles the unwanted enantiomer into the desired product. Initially, the microorganism selectively oxidizes the (R)-phenylglycol present in the racemic mixture to form an intermediate, identified as beta-hydroxyphenyl ethyl ketone, through the action of specific oxidoreductases that require cofactors such as NADP+. This oxidation step is highly stereoselective, leaving the (S)-phenylglycol untouched in the reaction mixture. Subsequently, the accumulated ketone intermediate undergoes an asymmetric reduction, again catalyzed by intracellular enzymes, to form (S)-phenylglycol, thereby converting the 'waste' (R)-isomer into valuable product and theoretically allowing yields to exceed the 50% limit imposed by simple resolution. This dual-enzyme system operates in a coordinated manner within the whole cell, utilizing the cell's metabolic machinery to regenerate necessary cofactors, which sustains the catalytic cycle over extended reaction periods without the need for external cofactor addition.
Understanding the impurity control mechanism is crucial for ensuring the high purity required in pharmaceutical applications, and this biological method offers inherent advantages in this regard. The high regioselectivity and chemoselectivity of the microbial enzymes ensure that side reactions, such as the over-oxidation of the product or the formation of unrelated by-products, are minimized. The patent data indicates that by optimizing the nutrient medium composition, specifically the ratios of carbon sources like glucose and nitrogen sources like yeast extract, the metabolic activity of the cells can be tuned to favor the desired transformation pathway while suppressing competing metabolic routes. Furthermore, the use of whole cells provides a natural filtration effect, where the cell membrane prevents large molecular impurities from interfering with the active site of the enzymes, and the mild aqueous reaction conditions prevent the degradation of the sensitive diol structure that might occur under harsh chemical conditions. The analytical data provided in the patent, utilizing chiral HPLC, confirms that the resulting product profile is clean, with the primary impurity being the unreacted starting material or the transient ketone intermediate, both of which can be easily removed through standard extraction and crystallization techniques, ensuring the final (S)-phenylglycol meets the stringent purity specifications demanded by R&D directors for downstream synthesis.
How to Synthesize (S)-Phenylglycol Efficiently
Implementing this biocatalytic route requires careful attention to the cultivation and reaction conditions to maximize the efficiency of the whole-cell catalyst. The patent outlines a robust protocol that begins with the activation of the Candida parapsilosis strain in a specifically formulated nutrient medium, followed by the harvesting of cells at the optimal growth phase to ensure high enzymatic activity. The reaction is then conducted in a buffered aqueous system where the concentration of the substrate and the cell density are balanced to prevent substrate inhibition while maintaining a high reaction rate. While the specific parameters such as pH, temperature, and incubation time are critical and detailed in the technical documentation, the general workflow emphasizes the simplicity of the operation, requiring standard fermentation and downstream processing equipment that is readily available in most chemical manufacturing facilities. For a comprehensive understanding of the exact operational parameters and step-by-step execution required to replicate these results in a GMP environment, please refer to the standardized synthesis guide provided below.
- Cultivate Candida parapsilosis CCTCC M203011 in optimized nutrient medium containing glucose and yeast extract at 30°C for 48 hours to generate sufficient biomass.
- Harvest the microbial cells via centrifugation, wash with physiological saline, and suspend in a phosphate buffer system to prepare the whole-cell biocatalyst.
- Introduce racemic phenylethanediol to the cell suspension and maintain at 33°C for 48 hours, allowing stereoselective oxidation and reduction to yield (S)-phenylglycol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this microbial transformation technology offers substantial strategic advantages for procurement managers and supply chain heads looking to optimize their sourcing of chiral intermediates. The shift from chemical to biological synthesis fundamentally alters the cost structure of production by eliminating the need for expensive chiral catalysts, protecting groups, and hazardous reagents, which translates directly into a more competitive pricing model for the final product. The simplified process flow, which combines cultivation and conversion in aqueous media, reduces the number of unit operations required, thereby lowering capital expenditure on specialized equipment and reducing the overall energy footprint of the manufacturing process. This efficiency gain is particularly significant for high-volume applications where even marginal improvements in yield or processing time can result in substantial cost savings over the lifecycle of a product, making it an attractive option for companies aiming to reduce their cost of goods sold without compromising on quality.
- Cost Reduction in Manufacturing: The elimination of toxic heavy metal catalysts and complex protection-deprotection sequences significantly reduces the raw material costs and waste disposal fees associated with traditional chemical synthesis. By utilizing renewable microbial resources and operating under mild conditions, the process minimizes energy consumption and solvent usage, leading to a leaner and more cost-effective production model that enhances profit margins for downstream users.
- Enhanced Supply Chain Reliability: The robustness of the microbial strains and the simplicity of the fermentation process ensure a stable and consistent supply of high-purity (S)-phenylglycol, mitigating the risks associated with supply chain disruptions common in complex chemical syntheses. The scalability of the biological system allows for rapid ramp-up of production capacity to meet fluctuating market demands, ensuring that procurement teams can secure reliable long-term contracts without the fear of supply shortages or quality variability.
- Scalability and Environmental Compliance: The aqueous nature of the reaction system and the biodegradability of the microbial biomass simplify waste treatment and ensure compliance with increasingly stringent environmental regulations. This green manufacturing profile not only reduces the regulatory burden on the supplier but also aligns with the sustainability goals of major pharmaceutical and chemical companies, facilitating smoother audits and fostering long-term partnerships based on shared values of environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the biocatalytic production of (S)-phenylglycol, based on the detailed findings of the patent data. These insights are designed to clarify the feasibility and advantages of this technology for potential partners and stakeholders. For more specific technical data or custom synthesis inquiries, our team is available to provide detailed route feasibility assessments and specific COA data to support your development efforts.
Q: What is the optical purity achievable with this microbial method?
A: According to patent CN1212403C, the use of Candida parapsilosis CCTCC M203011 can achieve an optical purity (e.e. value) ranging from 90% to 100%, with specific embodiments demonstrating values as high as 98.80% e.e.
Q: How does this biological process compare to chemical resolution?
A: Unlike chemical resolution which often requires toxic chiral catalysts and complex protection-deprotection steps, this biological process operates under mild conditions, eliminates heavy metal contamination, and offers higher stereoselectivity without the need for hazardous reagents.
Q: Can this process be scaled for industrial production?
A: Yes, the patent details optimized culture conditions and whole-cell catalytic systems that are designed for scalability. The use of robust microbial strains like Candida parapsilosis facilitates large-scale fermentation and downstream processing suitable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Phenylglycol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of next-generation pharmaceuticals and advanced materials. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the microbial transformation of phenylethanediol can be seamlessly transitioned from the laboratory to the manufacturing floor. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of (S)-phenylglycol we supply meets the exacting standards required by R&D directors and regulatory bodies worldwide. We are committed to delivering not just a product, but a comprehensive solution that enhances your supply chain resilience and accelerates your time to market.
We invite you to collaborate with us to explore how this advanced biocatalytic route can optimize your production costs and improve your product quality. Our technical procurement team is ready to assist you with a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By partnering with us, you gain access to our deep technical expertise and robust supply network, allowing you to focus on your core competencies while we handle the complexities of chemical manufacturing. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how we can become your trusted partner in chiral synthesis.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
