Revolutionizing 2-Bromo-1,4-Naphthoquinone Production: A Strategic Guide for Global Pharmaceutical Sourcing
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with operational safety and cost efficiency. A pivotal advancement in this domain is documented in patent CN113461506A, which details a novel preparation process for 2-bromo-1,4-naphthoquinone, a critical building block in the synthesis of various bioactive molecules and bulk drugs. Traditionally, the synthesis of this quinone derivative has been plagued by harsh reaction conditions and significant impurity profiles that complicate downstream processing. However, this new methodology shifts the synthetic paradigm by utilizing 1-naphthol as the starting material instead of the conventional 1,4-naphthoquinone. By employing N-bromosuccinimide (NBS) in a controlled aqueous acetic acid medium, the process achieves a remarkable enhancement in both yield and product quality. For global procurement teams and R&D directors, understanding this shift is essential, as it represents a move towards greener chemistry that eliminates the need for toxic heavy metal catalysts while ensuring the stringent purity specifications required for modern drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-bromo-1,4-naphthoquinone has relied heavily on the direct bromination of 1,4-naphthoquinone. As illustrated in the reaction scheme below, this conventional pathway typically necessitates the use of copper(II) bromide (CuBr2) alongside NBS, often in the presence of hazardous reagents or conditions that generate hydrogen cyanide (HCN) byproducts. 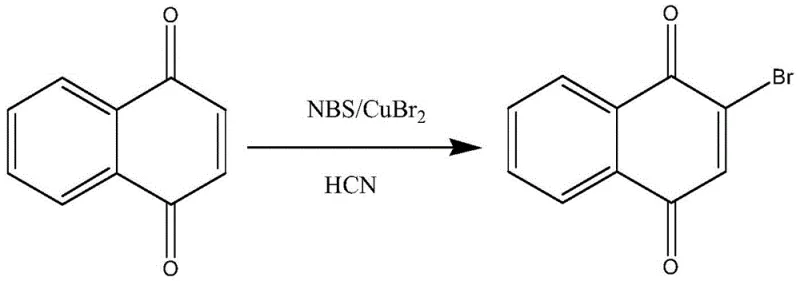 The reliance on copper salts introduces a significant burden on the purification workflow, as residual heavy metals must be rigorously removed to meet pharmaceutical standards, often requiring complex chelation or extensive washing procedures. Furthermore, data from prior art indicates that the purity of the product obtained via this traditional route frequently falls below 80%, necessitating labor-intensive purification steps such as silica gel column chromatography. This not only inflates the cost of goods sold (COGS) due to high solvent consumption but also creates bottlenecks in manufacturing throughput, making it less viable for large-scale commercial production where efficiency and environmental compliance are paramount.
The reliance on copper salts introduces a significant burden on the purification workflow, as residual heavy metals must be rigorously removed to meet pharmaceutical standards, often requiring complex chelation or extensive washing procedures. Furthermore, data from prior art indicates that the purity of the product obtained via this traditional route frequently falls below 80%, necessitating labor-intensive purification steps such as silica gel column chromatography. This not only inflates the cost of goods sold (COGS) due to high solvent consumption but also creates bottlenecks in manufacturing throughput, making it less viable for large-scale commercial production where efficiency and environmental compliance are paramount.
The Novel Approach
In stark contrast, the innovative process disclosed in the patent leverages 1-naphthol as a more reactive and selective precursor. The reaction proceeds smoothly in a mixture of glacial acetic acid and water, utilizing NBS as the sole brominating agent under mild thermal conditions ranging from 40°C to 50°C. This approach fundamentally alters the reaction landscape by avoiding the introduction of transition metal catalysts entirely. The absence of copper not only simplifies the workup procedure but also mitigates the risk of metal-catalyzed side reactions that often lead to poly-brominated impurities or structural degradation of the quinone core. The result is a white solid product that, after simple recrystallization from 95% ethanol, achieves high purity levels suitable for direct use in subsequent synthetic steps without further chromatographic purification. This streamlining of the workflow translates directly into operational excellence, offering a scalable solution that aligns with the principles of green chemistry and sustainable manufacturing practices demanded by modern regulatory bodies.
Mechanistic Insights into NBS-Mediated Oxidative Bromination
To fully appreciate the technical superiority of this method, one must examine the mechanistic underpinnings of the transformation from 1-naphthol to 2-bromo-1,4-naphthoquinone. The reaction likely proceeds through an initial electrophilic aromatic substitution where the electron-rich naphthol ring attacks the bromine species generated in situ from NBS. 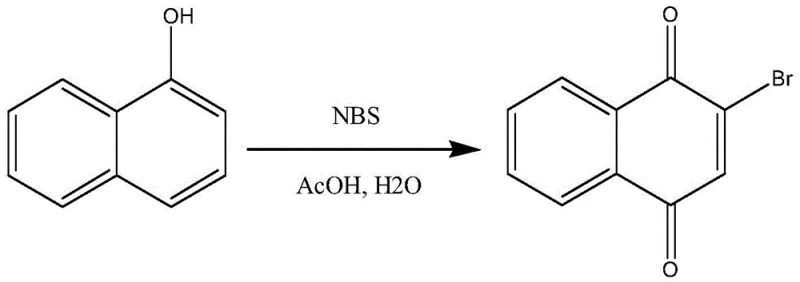 The presence of water in the acetic acid solvent system plays a dual role: it acts as a co-solvent to modulate the polarity of the reaction medium, facilitating the dissolution of polar intermediates, and potentially participates in the hydrolysis steps required to convert the initial brominated phenolic intermediate into the final quinone structure. The controlled temperature of 40°C to 50°C is critical; it provides sufficient activation energy for the oxidation and bromination to occur while preventing the thermal decomposition of the sensitive quinone product or the formation of dibromo byproducts. This precise control over reaction kinetics ensures that the bromine atom is installed selectively at the 2-position, driven by the electronic directing effects of the hydroxyl group and the subsequent oxidation potential of the system.
The presence of water in the acetic acid solvent system plays a dual role: it acts as a co-solvent to modulate the polarity of the reaction medium, facilitating the dissolution of polar intermediates, and potentially participates in the hydrolysis steps required to convert the initial brominated phenolic intermediate into the final quinone structure. The controlled temperature of 40°C to 50°C is critical; it provides sufficient activation energy for the oxidation and bromination to occur while preventing the thermal decomposition of the sensitive quinone product or the formation of dibromo byproducts. This precise control over reaction kinetics ensures that the bromine atom is installed selectively at the 2-position, driven by the electronic directing effects of the hydroxyl group and the subsequent oxidation potential of the system.
From an impurity control perspective, this mechanism offers distinct advantages over the copper-catalyzed route. In the absence of metal ions, the formation of metal-organic complexes that can trap product or catalyze polymerization is eliminated. The primary impurities are likely to be unreacted starting material or minor over-oxidation products, both of which are effectively removed during the recrystallization step in ethanol. The use of saturated sodium bicarbonate during the extraction phase further ensures the removal of acidic byproducts, such as succinimide and residual acetic acid, yielding a neutral and stable final product. This high level of chemical fidelity is crucial for R&D directors who require consistent batch-to-batch reproducibility when developing new drug candidates, as variable impurity profiles can significantly impact the safety and efficacy of the final pharmaceutical formulation.
How to Synthesize 2-Bromo-1,4-Naphthoquinone Efficiently
Implementing this synthesis on a pilot or commercial scale requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and safety. The process begins with the preparation of the brominating solution, followed by the controlled addition of the substrate, and concludes with a straightforward isolation protocol. The detailed standardized operating procedures, including specific stirring rates, addition timelines, and safety precautions for handling NBS and organic solvents, are outlined in the technical guide below.
- Prepare the brominating agent by dissolving N-bromosuccinimide (NBS) in a mixture of glacial acetic acid and water, maintaining a temperature between 40°C and 50°C.
- Slowly add a solution of 1-naphthol in glacial acetic acid to the reaction mixture over 75 minutes to control exothermicity and ensure selective mono-bromination.
- Isolate the crude product via dichloromethane extraction, wash with saturated sodium bicarbonate, and purify through recrystallization in 95% ethanol to achieve pharmaceutical grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling economic and logistical benefits that extend beyond mere chemical elegance. The elimination of expensive and toxic copper salts reduces the raw material costs significantly, while the simplified purification process drastically cuts down on solvent usage and waste disposal fees. This translates into a more competitive pricing structure for the final intermediate, allowing downstream manufacturers to optimize their own cost models without compromising on quality. Furthermore, the mild reaction conditions reduce the energy consumption associated with heating and cooling cycles, contributing to a lower carbon footprint for the manufacturing process. These factors combined create a resilient supply chain capable of meeting high-volume demands with greater flexibility and reduced operational risk.
- Cost Reduction in Manufacturing: The removal of copper bromide from the reagent list eliminates the need for specialized heavy metal scavenging resins or complex aqueous workups designed to lower metal content to ppm levels. This simplification of the downstream processing significantly reduces the consumption of auxiliary materials and labor hours. Additionally, the ability to bypass silica gel column chromatography in favor of simple recrystallization leads to substantial savings in solvent costs and processing time. The overall effect is a leaner manufacturing process that maximizes resource efficiency and minimizes waste generation, directly impacting the bottom line through reduced variable costs per kilogram of product produced.
- Enhanced Supply Chain Reliability: The reagents used in this novel process, specifically 1-naphthol and NBS, are commodity chemicals with robust global supply chains, reducing the risk of raw material shortages compared to specialized catalysts. The mild reaction conditions also place less stress on reactor equipment, potentially extending the lifespan of manufacturing assets and reducing maintenance downtime. By adopting a process that is less sensitive to extreme temperatures or pressures, manufacturers can ensure more consistent production schedules and faster turnaround times. This reliability is critical for maintaining continuous supply to pharmaceutical clients who operate on tight development and production timelines, ensuring that project milestones are met without delay.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to multi-ton production is facilitated by the homogeneous nature of the reaction mixture and the absence of heterogeneous metal catalysts that can cause fouling or heat transfer issues in large reactors. The aqueous acetic acid solvent system is easier to handle and recover compared to purely organic solvent systems, aligning with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. The reduction in hazardous waste, particularly heavy metal sludge, simplifies regulatory compliance and lowers the costs associated with waste treatment and disposal. This makes the process not only commercially viable but also environmentally sustainable, appealing to stakeholders who prioritize corporate social responsibility and green manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-bromo-1,4-naphthoquinone using this advanced methodology. These insights are derived directly from the patent data and practical manufacturing experience to provide clarity for potential partners.
Q: Why is the 1-naphthol route superior to the traditional 1,4-naphthoquinone bromination?
A: The traditional route utilizing 1,4-naphthoquinone often requires toxic copper salts and generates significant impurities, resulting in purity levels below 80%. The novel 1-naphthol pathway utilizes NBS in aqueous acetic acid, offering milder conditions, eliminating heavy metal contamination risks, and achieving significantly higher purity suitable for direct downstream usage.
Q: What are the critical process parameters for scaling this synthesis?
A: Critical parameters include maintaining the reaction temperature strictly between 40°C and 50°C to prevent over-bromination or decomposition. Additionally, the dropwise addition time of approximately 75 minutes is crucial for heat management and selectivity. The ratio of glacial acetic acid to water must be optimized between 1:2 and 1:5 to ensure proper solubility and reaction kinetics.
Q: Does this process require complex purification steps like column chromatography?
A: No, one of the primary commercial advantages of this patented process is the elimination of silica gel column purification. The crude product obtained after extraction and drying can be directly recrystallized from 95% ethanol to yield high-purity 2-bromo-1,4-naphthoquinone, drastically reducing processing time and solvent consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-1,4-Naphthoquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature are faithfully reproduced on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-bromo-1,4-naphthoquinone meets the highest international standards. Our commitment to quality assurance means that you can rely on us for consistent supply, whether you are in the early stages of drug discovery or ramping up for full-scale clinical manufacturing.
We invite you to collaborate with us to optimize your supply chain and reduce your overall production costs. By leveraging our expertise in this novel synthetic route, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing processes can support your strategic goals and drive success in your pharmaceutical development projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
