Scalable Synthesis of JAK3 Inhibitor Intermediate via Classical Resolution for Commercial Production
Scalable Synthesis of JAK3 Inhibitor Intermediate via Classical Resolution for Commercial Production
The pharmaceutical industry is constantly seeking robust and scalable synthetic routes for key enzyme inhibitor intermediates, particularly for high-value targets like Janus kinase 3 (JAK3) inhibitors. A recent breakthrough detailed in patent CN113121413A introduces a highly efficient preparation method for the key intermediate (3R,6S)-1-benzyl-6-methylpiperidine-3-amine, which is crucial for the synthesis of PF-06651600. This novel approach fundamentally shifts the paradigm from reliance on low-yield chiral chromatography to a cost-effective classical resolution strategy. By leveraging standard chemical transformations such as Boc protection, catalytic hydrogenation, and stereospecific salt formation, this method addresses the critical bottlenecks of scalability and cost that have historically plagued the production of this specific piperidine derivative. For global procurement teams and R&D directors, this represents a significant opportunity to optimize the supply chain for immunomodulatory treatments targeting conditions like alopecia areata and rheumatoid arthritis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral piperidine intermediates for JAK3 inhibitors has relied heavily on chiral chromatography for enantiomeric separation, a technique that presents severe limitations for industrial application. As illustrated in the prior art pathways, obtaining the pure enantiomer often results in a mixture where the target substance is difficult to isolate in high yield without multiple separation cycles. This dependency on preparative chiral HPLC not only inflates the cost of goods sold due to the expense of chiral stationary phases but also creates a significant bottleneck in throughput. Furthermore, chromatographic methods are inherently difficult to scale beyond kilogram quantities without massive capital investment in specialized equipment, making them unsuitable for the multi-ton demands of commercial API manufacturing. The low overall yield associated with discarding the unwanted enantiomer without recycling strategies further exacerbates the economic inefficiency of these conventional routes.
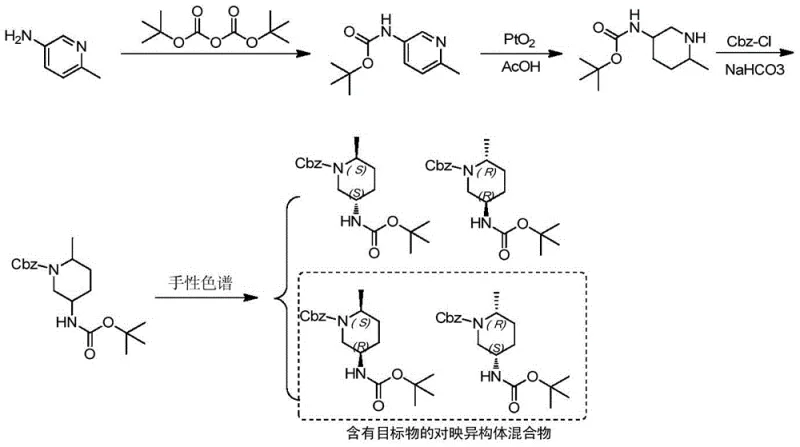
The Novel Approach
In stark contrast, the methodology disclosed in the patent employs a streamlined five-step synthetic sequence that culminates in a classical resolution process, effectively bypassing the need for chromatographic separation. The route begins with the Boc protection of 6-methylpyridin-3-amine, followed by a highly efficient catalytic hydrogenation to saturate the pyridine ring. Subsequent N-benzylation and Boc deprotection yield a racemic amine which is then subjected to resolution using a chiral acid resolving agent. This strategic pivot allows for the isolation of the desired (3R,6S) enantiomer through crystallization of a diastereomeric salt, a unit operation that is infinitely more scalable and economical than chromatography. The entire process utilizes cheap and easily obtained raw materials and operates under mild reaction conditions, ensuring high reaction controllability and a robust profile for industrial application.
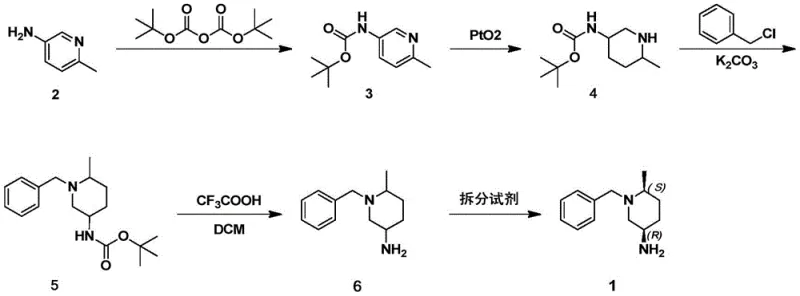
Mechanistic Insights into Catalytic Hydrogenation and Classical Resolution
The core of this synthetic success lies in the precise control of the catalytic hydrogenation step and the subsequent stereoselective crystallization. During the reduction of the pyridine ring, the use of platinum dioxide (PtO2) in a mixed solvent system of acetic acid and ethanol proves critical. The acidic environment facilitates the protonation of the nitrogen atom, enhancing the adsorption of the heterocycle onto the catalyst surface and driving the reaction to completion at moderate temperatures of 50-60°C and a hydrogen pressure of 50 psi. This specific catalytic system minimizes the formation of over-reduced byproducts or isomeric impurities, ensuring that the resulting piperidine scaffold retains the necessary structural integrity for downstream functionalization. The high conversion rate observed, often exceeding 99%, demonstrates the efficacy of this catalyst-solvent combination in handling the steric and electronic properties of the substituted pyridine substrate.
Following the generation of the racemic amine, the resolution mechanism relies on the formation of diastereomeric salts with di-p-toluoyl-L-tartaric acid. This resolving agent exhibits defined stereospecificity, selectively binding to one enantiomer of the racemic mixture to form an insoluble precipitate while leaving the other enantiomer in solution. The solubility difference between the two diastereomeric salts in an ethanol-water mixture is exploited to achieve high optical purity. By carefully controlling the cooling profile and solvent ratios, the target (3R,6S) enantiomer can be precipitated with high selectivity. This process not only achieves high enantiomeric excess but also allows for the potential recovery and recycling of the mother liquor containing the opposite enantiomer, further enhancing the atom economy of the overall process compared to destructive separation methods.
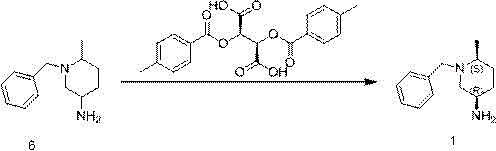
How to Synthesize (3R,6S)-1-benzyl-6-methylpiperidine-3-amine Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly during the hydrogenation and resolution stages, to maximize yield and purity. The process is designed to be operationally simple, utilizing standard reactor equipment found in most fine chemical facilities. The initial protection and reduction steps set the stage for the critical stereochemical differentiation that occurs in the final step. Operators must ensure strict temperature control during the addition of trifluoroacetic acid for deprotection to prevent side reactions, and precise solvent ratios during the resolution to optimize crystal growth. The following guide outlines the standardized workflow derived from the patent examples, providing a clear roadmap for technical teams aiming to implement this route for commercial scale-up of complex pharmaceutical intermediates.
- Perform Boc protection on 6-methylpyridin-3-amine using di-tert-butyl dicarbonate in ethanol to form the carbamate intermediate.
- Execute catalytic hydrogenation using platinum dioxide in acetic acid and ethanol at 50 psi to reduce the pyridine ring.
- Conduct benzyl protection with benzyl chloride and potassium carbonate, followed by Boc deprotection using trifluoroacetic acid.
- Perform classical resolution using di-p-toluoyl-L-tartaric acid in ethanol/water to isolate the target (3R,6S) enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from chromatographic separation to classical resolution offers profound economic and logistical benefits. The elimination of expensive chiral columns and the associated solvent consumption drastically simplifies the cost structure of the intermediate. Moreover, the reliance on commodity chemicals like benzyl chloride, potassium carbonate, and tartaric acid derivatives ensures that the supply chain is resilient to fluctuations in the availability of exotic reagents. This robustness translates directly into enhanced supply chain reliability, as the risk of production stoppages due to specialized material shortages is significantly mitigated. The ability to produce high-purity intermediates using standard crystallization techniques also means that lead times can be reduced, allowing for more responsive inventory management and faster time-to-market for the final drug product.
- Cost Reduction in Manufacturing: The replacement of chiral chromatography with classical resolution removes one of the most expensive unit operations in fine chemical synthesis. Chromatographic resins are costly to purchase and maintain, and the process typically suffers from low throughput and high solvent usage. By switching to a precipitation-based separation, the process leverages inexpensive resolving agents and standard filtration equipment. This fundamental change in the purification strategy leads to substantial cost savings in both direct material costs and operational overhead, making the final API more competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The synthetic route is built upon a foundation of readily available starting materials and reagents. 6-methylpyridin-3-amine is a common building block, and the protecting groups used (Boc, Benzyl) are industry standards with secure global supply lines. This accessibility ensures that production schedules are not held hostage by the lead times of niche catalysts or specialized chiral auxiliaries. Furthermore, the mild reaction conditions reduce the stress on equipment, lowering maintenance requirements and increasing the overall uptime of the manufacturing facility, thereby guaranteeing a continuous and reliable flow of materials to downstream API synthesis sites.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this route is superior to many alternative methods. The hydrogenation step operates at relatively low pressures, and the resolution step avoids the massive volumes of organic solvents typically required for preparative HPLC. The waste stream is easier to manage and treat, aligning with increasingly stringent environmental regulations. The process is inherently scalable from laboratory benchtop to multi-ton commercial production without the need for re-engineering the separation logic. This scalability ensures that as demand for the JAK3 inhibitor grows, the supply of the key intermediate can be ramped up seamlessly to meet market needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, based on the detailed experimental data and advantages outlined in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for their own supply chains. The answers provided reflect the specific conditions and outcomes reported in the intellectual property, offering a transparent view of the process capabilities and limitations.
Q: Why is classical resolution preferred over chiral chromatography for this intermediate?
A: Classical resolution using di-p-toluoyl-L-tartaric acid eliminates the need for expensive chiral columns and low-yield separation processes, significantly reducing production costs and enabling large-scale manufacturing capabilities that chromatography cannot support economically.
Q: What catalyst is used for the hydrogenation step in this novel route?
A: The process utilizes platinum dioxide (PtO2) as the preferred catalyst in a mixed solvent system of acetic acid and ethanol, operating under mild hydrogen pressure of approximately 50 psi to ensure high conversion rates.
Q: How does this method improve supply chain reliability for JAK3 inhibitors?
A: By relying on cheap and easily obtained starting materials like 6-methylpyridin-3-amine and avoiding complex purification technologies, the method ensures consistent batch-to-batch quality and reduces lead times for high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3R,6S)-1-benzyl-6-methylpiperidine-3-amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of key intermediates like (3R,6S)-1-benzyl-6-methylpiperidine-3-amine for the development of next-generation immunomodulators. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical partners. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required for clinical and commercial applications. Our expertise in classical resolution and catalytic hydrogenation allows us to deliver this complex intermediate with the consistency and reliability that your projects deserve.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing process can drive value and efficiency for your organization. Let us be your partner in bringing life-saving therapies to patients faster and more affordably.
