Optimized Industrial Synthesis of 2-Methoxyimino-2-Furylacetic Acid Ammonium Salt for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust, scalable, and environmentally compliant pathways for the production of critical cephalosporin intermediates. Patent CN101538255B, published in July 2011, introduces a significant technological advancement in the preparation of 2-methoxy imino group 2-furan ammonium acetate, commonly known as SMIA. This compound serves as the essential C-7 side chain for the synthesis of Cefuroxime Axetil, a widely prescribed second-generation cephalosporin antibiotic. The disclosed methodology addresses long-standing inefficiencies in traditional manufacturing routes by optimizing solvent systems and reaction thermodynamics. By shifting from hazardous reagents and high-loss solvents to a streamlined butyl acetate-based extraction protocol, this patent offers a blueprint for cost reduction in pharmaceutical intermediate manufacturing. For global procurement teams and R&D directors, understanding the nuances of this improved synthesis is vital for securing a reliable SMIA supplier capable of delivering high-purity materials with consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of SMIA has been plagued by significant safety hazards and economic inefficiencies. Traditional routes often relied on starting materials such as 2-furancarboxylic acid, which required conversion to acid chlorides using thionyl chloride, a highly corrosive reagent that generates substantial acidic waste. Alternative pathways utilized cyanation steps involving toxic cyanide salts, posing severe occupational health risks and complicating waste treatment protocols. Furthermore, even improved methods utilizing 2-acetylfuran as a starting material suffered from poor solvent economy. Previous iterations of the oximation-rearrangement route typically employed dichloromethane and ethyl acetate for extraction processes. These solvents, while effective, exhibit high solubility in aqueous phases, leading to massive solvent losses during workup. Additionally, conventional thermal controls were often too aggressive, with reaction temperatures hovering between 65°C and 70°C. This excessive heat promoted the hydrolysis of the intermediate, resulting in the formation of 2-furoic acid byproducts in quantities ranging from 5% to 15%, thereby drastically reducing the overall yield and complicating downstream purification efforts.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally re-engineers the extraction and thermal parameters to overcome these legacy deficits. The core of this novel approach lies in the substitution of ethyl acetate with butyl acetate as the primary extraction solvent. This seemingly simple switch yields profound technical benefits due to the physicochemical properties of butyl acetate, specifically its lower partition coefficient in water. As illustrated in the comprehensive reaction scheme below, the process flows from oximation through to salt formation with enhanced efficiency.
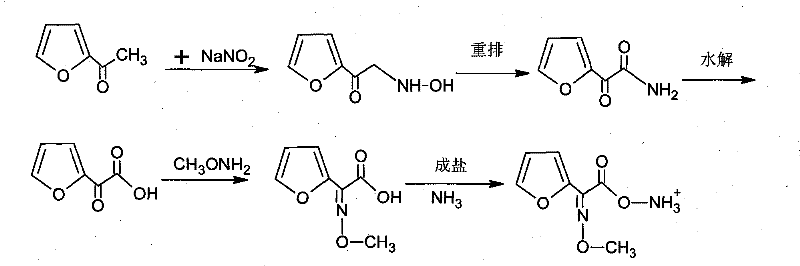
By implementing butyl acetate, the process achieves a dramatic reduction in solvent consumption, with technical metrics indicating a 50% decrease in unit usage compared to ethyl acetate systems. Moreover, the protocol introduces a precise temperature window of 58°C to 60°C for the critical oximation step. This tighter thermal control effectively suppresses the degradation pathway that leads to furoic acid, limiting its generation to less than 1%. The result is a cleaner reaction profile, higher crude yields, and a significantly more sustainable manufacturing footprint that aligns with modern green chemistry principles.
Mechanistic Insights into Oximation-Rearrangement and Solvent Dynamics
To fully appreciate the value of this synthesis for a reliable pharmaceutical intermediate supplier, one must delve into the mechanistic subtleties governing the transformation. The reaction initiates with the interaction of sodium nitrite and 2-acetylfuran under acidic conditions to form an oxime intermediate. This species undergoes a spontaneous rearrangement to generate an alpha-keto amide, which is subsequently hydrolyzed to yield the crucial furan keto acid. The integrity of this furan ring system is paramount, yet it is susceptible to oxidative degradation and hydrolytic cleavage if thermal energy is not meticulously managed. The patent specifies a reaction temperature of 59°C ± 1°C as the optimal setpoint. Operating within this narrow band ensures that the kinetic energy is sufficient to drive the rearrangement forward without providing the activation energy required for the competing hydrolysis reaction that produces unwanted furoic acid.
Following the formation of the keto acid, the process moves to condensation with methoxyamine hydrochloride. The efficiency of isolating the intermediate and final products relies heavily on the solvent choice. The detailed mechanism of extraction favors butyl acetate due to its lower polarity and reduced hydrogen bonding capability with water compared to ethyl acetate. This physical property minimizes the entrainment of solvent into the aqueous waste stream, allowing for higher recovery rates and easier solvent recycling. The final step involves the introduction of ammonia gas into a solution of the acid in a butyl acetate and methanol mixture. This salification step precipitates the final ammonium salt product. The use of a mixed solvent system here is critical for controlling the crystalline structure of the final API intermediate, ensuring good flow properties and stability for storage and transport.
How to Synthesize 2-Methoxyimino-2-Furylacetic Acid Ammonium Salt Efficiently
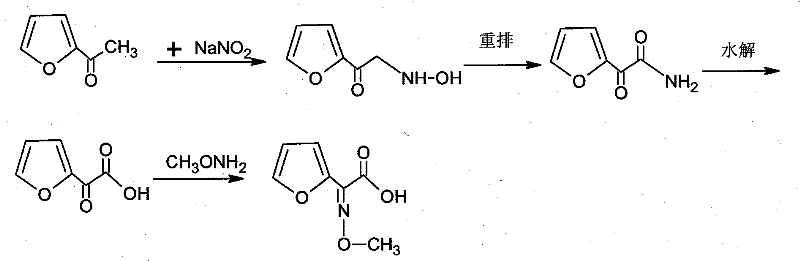
Implementing this optimized route requires strict adherence to the specified operational parameters to replicate the high yields and purity profiles reported in the intellectual property. The process is designed to be scalable, moving seamlessly from laboratory validation to commercial production tanks. Operators must prioritize the precise control of pH during the extraction phases, maintaining values around 0.2 for the initial keto acid extraction and 0.5 for the final product isolation to maximize partitioning into the organic phase. The detailed standardized synthesis steps, including specific reagent ratios and agitation speeds necessary for industrial replication, are outlined in the technical guide below.
- React sodium nitrite with 2-acetylfuran at 58-60°C to generate furan keto acid via oximation and rearrangement.
- Extract the furan keto acid using butyl acetate at pH 0.2 to minimize aqueous solubility losses.
- Condense the acid with methoxyamine hydrochloride, extract with butyl acetate, and form the ammonium salt using ammonia in a butyl acetate/methanol mixture.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented methodology represents a strategic opportunity to optimize the cost structure and reliability of the cephalosporin supply chain. The shift away from hazardous reagents like cyanides and thionyl chloride not only mitigates regulatory risk but also simplifies the logistics of raw material sourcing. By adopting a process that utilizes common, recoverable solvents and safer reagents, manufacturers can ensure continuous production uptime without the interruptions often caused by strict environmental inspections or hazardous material transport delays. The technical improvements directly translate into commercial resilience, offering a buffer against market volatility in raw material costs.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the drastic reduction in solvent consumption. Since the dissolution loss of ethyl acetate in water is technically documented to be four times larger than that of butyl acetate, switching solvents results in a verified 50% reduction in unit solvent consumption. This substantial decrease in variable costs directly improves the gross margin of the final intermediate. Furthermore, the suppression of the furoic acid byproduct from typical levels of 5-15% down to less than 1% means that less raw material is wasted on side reactions. This improvement in atom economy reduces the effective cost per kilogram of the active pharmaceutical ingredient, providing a competitive pricing advantage in the global market.
- Enhanced Supply Chain Reliability: The elimination of highly toxic cyanide salts and corrosive chlorinating agents removes significant bottlenecks associated with the procurement and handling of dangerous goods. Facilities utilizing this safer route face fewer regulatory hurdles and can maintain more consistent inventory levels. The robustness of the butyl acetate extraction system also ensures that production batches are less susceptible to variations in raw material quality, as the solvent system is more forgiving and efficient at separating the product from impurities. This consistency is crucial for downstream drug manufacturers who require uninterrupted supply to meet their own production schedules for finished antibiotics.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is inherently more scalable. The reduction in aqueous solvent waste lowers the burden on wastewater treatment facilities, reducing the operational expenditure associated with effluent management. The lower formation of byproducts simplifies the purification train, potentially reducing the number of recrystallization steps required to meet pharmacopeial standards. This streamlined workflow facilitates easier scale-up from pilot plants to multi-ton reactors, ensuring that the supply chain can rapidly respond to surges in demand for cephalosporin antibiotics without compromising on quality or environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the comparative data and experimental embodiments provided in the patent documentation, offering clarity on why this method represents a superior choice for modern chemical manufacturing. Understanding these details helps stakeholders make informed decisions about vendor qualification and process adoption.
Q: Why is butyl acetate preferred over ethyl acetate in this synthesis?
A: Butyl acetate exhibits significantly lower solubility in water compared to ethyl acetate. Technical data indicates that under identical conditions, the dissolution loss of ethyl acetate in the aqueous phase is approximately four times greater than that of butyl acetate, leading to a 50% reduction in unit solvent consumption when switching to butyl acetate.
Q: How does temperature control affect impurity profiles in SMIA production?
A: Maintaining the oximation reaction temperature strictly between 58°C and 60°C is critical. Conventional methods operating at 65-70°C often result in 5-15% formation of 2-furoic acid byproduct. The optimized low-temperature protocol reduces this byproduct to less than 1%, significantly improving overall yield and purity.
Q: Is this method suitable for large-scale commercial manufacturing?
A: Yes, the process eliminates the use of highly toxic cyanides and corrosive thionyl chloride found in older routes. Combined with the use of recoverable solvents like butyl acetate and simplified extraction steps, the method is designed for robust industrial scalability and environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable SMIA Supplier
At NINGBO INNO PHARMCHEM, we recognize that the synthesis of 2-methoxy imino group 2-furan ammonium acetate is a critical link in the global antibiotic value chain. Our technical team has extensively analyzed the pathway described in CN101538255B and integrated its core principles into our own manufacturing protocols. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of multinational pharmaceutical corporations. Our facilities are equipped with rigorous QC labs and advanced process control systems to maintain stringent purity specifications, guaranteeing that every batch of SMIA meets the exacting standards required for Cefuroxime Axetil synthesis.
We invite procurement leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume needs. By leveraging our optimized production capabilities, we can help you reduce lead time for high-purity pharmaceutical intermediates while securing a more sustainable supply source. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to technical excellence translates into tangible value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
