Advanced Purification Technology for High-Purity Pregnenolone Acetate Intermediates
Advanced Purification Technology for High-Purity Pregnenolone Acetate Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, environmentally sustainable pathways for the production of critical steroid intermediates. A significant breakthrough in this domain is documented in patent CN101974058A, which details a novel purification technology for pregnenolone acetate. This innovation addresses a longstanding bottleneck in the synthesis of steroid hormones, specifically enabling the efficient isolation of high-purity products from crude materials generated via hydrogen peroxide oxidative degradation. As a leading entity in the chemical sector, understanding the nuances of this technology is vital for stakeholders aiming to optimize their supply chains for pharmaceutical intermediates. The transition from traditional, hazardous oxidation methods to greener alternatives has created a demand for downstream purification processes that can handle the specific impurity profiles of these new routes. This report provides a deep technical and commercial analysis of how this purification methodology resolves critical manufacturing challenges, offering a viable path toward cost-effective and eco-friendly production of pregnenolone acetate derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of pregnenolone acetate has relied heavily on the Marker degradation method, which utilizes chromic anhydride as a potent oxidizing agent. While chemically effective, this traditional approach generates substantial quantities of toxic chromium waste, posing severe environmental hazards and escalating disposal costs for manufacturers. Furthermore, when attempting to replace chromic anhydride with greener oxidants like hydrogen peroxide, the resulting crude pregnenolone products typically exhibit purity levels between 80% and 90%. Direct purification of this crude material via standard recrystallization techniques has proven notoriously difficult due to the similar polarity of the target molecule and its byproducts. Attempts to recrystallize using alcohols like methanol or ethanol often lead to the formation of unwanted ether derivatives, such as 16-methoxy or 16-ethoxy steroids, thereby compromising the chemical integrity of the batch. Additionally, using acetic acid for recrystallization introduces high boiling point complications that hinder efficient drying, while the minimal polarity difference between the product and impurities renders simple precipitation ineffective. These technical barriers have historically prevented the widespread industrial adoption of the more environmentally friendly hydrogen peroxide oxidation route.
The Novel Approach
The technology disclosed in patent CN101974058A introduces a transformative strategy that bypasses these purification hurdles through a strategic acetylation step followed by selective solvent extraction. Instead of struggling to purify the free alcohol form of pregnenolone directly, the process converts the crude mixture into pregnenolone acetate. This chemical modification fundamentally alters the physicochemical properties of the molecule, creating a significant divergence in solubility between the desired acetate product and the remaining impurities. By leveraging specific organic solvents such as cyclohexane, petroleum ether, or toluene, the process effectively extracts the acetylated product while leaving polar impurities behind in the aqueous or mother liquor phases. 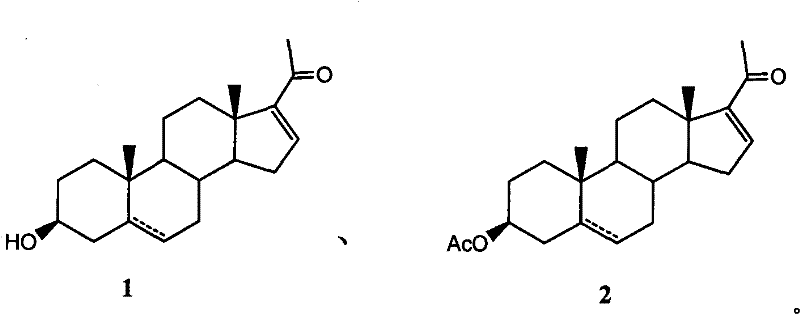
This approach not only facilitates high-yield recovery but also ensures that the final product achieves purity levels exceeding 96%, making it suitable for subsequent pharmaceutical synthesis. The method is versatile, accommodating various reaction conditions including direct acetylation in acetic anhydride or azeotropic dehydration in toluene, providing manufacturers with flexible operational parameters to suit their existing infrastructure.
Mechanistic Insights into Acetylation-Based Purification
The core mechanism driving the success of this purification technology lies in the differential solubility and partition coefficients established upon acetylation. When crude pregnenolone, containing approximately 80-90% active ingredient, reacts with acetic anhydride, the hydroxyl group at the C-3 position is esterified. This structural change reduces the overall polarity of the steroid nucleus compared to the unreacted impurities and side products which may retain more polar functional groups or possess different steric configurations. 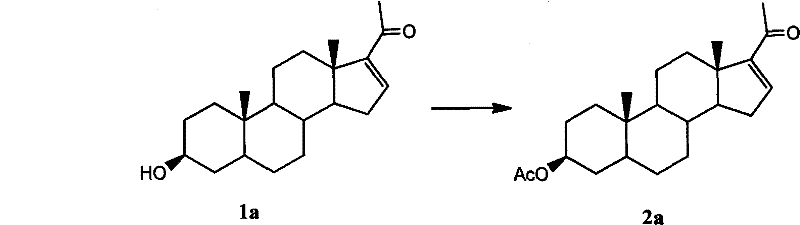
Following the reaction, the addition of water quenches excess acetic anhydride, creating a biphasic system where the lipophilic pregnenolone acetate preferentially partitions into non-polar organic solvents like warm cyclohexane or n-hexane. The process exploits the fact that the acetylated product has a much higher affinity for these aliphatic hydrocarbons than the polar degradation byproducts generated during the upstream hydrogen peroxide oxidation. Subsequent recrystallization from lower alcohols like ethanol or methanol further refines the crystal lattice, excluding any co-extracted impurities that do not fit the specific crystalline structure of the acetate. This multi-stage purification—acetylation, liquid-liquid extraction, and final recrystallization—creates a robust barrier against contamination, ensuring that the final high-purity pharmaceutical intermediate meets stringent quality specifications required for hormone synthesis.
Furthermore, the reaction kinetics are carefully controlled, typically proceeding at temperatures between 100°C and reflux for 0.5 to 1.5 hours, ensuring complete conversion without inducing thermal degradation of the sensitive delta-16 double bond. This precise control over reaction conditions is critical for maintaining the stereochemical integrity of the steroid backbone, which is essential for the biological activity of downstream drug products.
How to Synthesize Pregnenolone Acetate Efficiently
The synthesis of pregnenolone acetate via this patented method involves a streamlined sequence of acetylation, extraction, and crystallization steps designed for maximum efficiency and yield. The process begins with the preparation of the crude pregnenolone feedstock, which is then subjected to acetylation using acetic anhydride under controlled thermal conditions. Detailed operational parameters, including specific molar ratios of reagents and precise temperature ramps, are critical for optimizing the conversion rate and minimizing byproduct formation. Following the reaction, the workup procedure utilizes selective solvent extraction to isolate the crude acetate, which is then polished through recrystallization to achieve the final purity specification.
- React crude pregnenolone (80-90% purity) with acetic anhydride at 100°C to reflux temperature for 0.5 to 1.5 hours to ensure complete acetylation.
- Quench the reaction with water to decompose excess acetic anhydride, then perform multiple extractions using warm cyclohexane or petroleum ether.
- Recover the solvent under reduced pressure and recrystallize the residue from ethanol or methanol to obtain pure pregnenolone acetate with >96% content.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology offers substantial strategic advantages beyond mere technical feasibility. The primary value proposition lies in the significant reduction of environmental compliance costs associated with traditional chromium-based oxidation methods. By enabling the use of hydrogen peroxide upstream, manufacturers eliminate the need for expensive heavy metal waste treatment facilities and avoid the regulatory burdens linked to toxic effluent discharge. This shift translates directly into improved margin protection and long-term operational sustainability for the production of steroid hormone intermediates. Moreover, the simplification of the purification workflow removes the dependency on costly and time-consuming column chromatography, which is often a bottleneck in small-scale laboratory preparations but impractical for multi-ton commercial manufacturing.
- Cost Reduction in Manufacturing: The elimination of toxic chromium reagents and the associated waste disposal protocols results in a drastically simplified cost structure for steroid manufacturing. By utilizing common, inexpensive reagents like acetic anhydride and cyclohexane, the process avoids the volatility and high costs associated with specialized oxidizing agents. Furthermore, the high recovery yield from crude starting materials means that less raw material is wasted, enhancing the overall atom economy of the production line. The ability to process lower purity crude inputs (80-90%) directly into high-specification final products reduces the pressure on upstream synthesis units to achieve perfect conversion, thereby lowering the overall cost of goods sold (COGS) for the facility.
- Enhanced Supply Chain Reliability: The robustness of this purification method ensures a more stable and predictable supply of critical intermediates. Since the process relies on standard unit operations such as reflux, extraction, and filtration, it is less susceptible to the equipment failures or specialized catalyst shortages that can plague more complex synthetic routes. The use of widely available solvents and reagents mitigates the risk of supply chain disruptions caused by raw material scarcity. Additionally, the scalability of the method means that production volumes can be ramped up quickly to meet market demand without requiring extensive re-engineering of the plant infrastructure, providing a reliable buffer against market fluctuations.
- Scalability and Environmental Compliance: From a sustainability perspective, this technology aligns perfectly with modern green chemistry principles, facilitating easier permitting and community acceptance for manufacturing sites. The absence of heavy metal contaminants in the waste stream simplifies wastewater treatment processes and reduces the environmental footprint of the facility. The process is inherently scalable, having been demonstrated to work effectively across various batch sizes without loss of efficiency or purity. This scalability ensures that the technology can support the commercial scale-up of complex pharmaceutical intermediates from pilot plant quantities to full industrial production, securing the long-term viability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to legacy processes. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is acetylation necessary for purifying crude pregnenolone from hydrogen peroxide oxidation?
A: Direct recrystallization of crude pregnenolone is inefficient because impurities have similar polarity. Acetylation alters the solubility profile, allowing effective separation via solvent extraction and recrystallization.
Q: What are the environmental advantages of this purification method over traditional Marker degradation?
A: This method supports the upstream use of hydrogen peroxide instead of toxic chromic anhydride, eliminating heavy metal waste and significantly reducing environmental compliance costs.
Q: Can this process be scaled for industrial production of steroid intermediates?
A: Yes, the process utilizes standard unit operations like reflux, extraction, and recrystallization without requiring complex chromatography, making it highly suitable for multi-ton commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pregnenolone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced purification technologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101974058A are fully realized in practical, large-scale operations. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of pregnenolone acetate meets the exacting standards required for hormone synthesis. Our capability to handle complex steroid intermediates allows us to offer a secure and consistent supply source for multinational corporations seeking to diversify their vendor base.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our purified intermediates. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate our quality and capacity before making any commitment. Let us collaborate to build a more efficient and sustainable supply chain for your next generation of steroid therapeutics.
