Advanced Synthesis of Four-Coordinate N,N-Chelated Monoaryl Monofluoroborate Compounds for Commercial Scale-Up
Advanced Synthesis of Four-Coordinate N,N-Chelated Monoaryl Monofluoroborate Compounds for Commercial Scale-Up
Introduction to Patent CN113150018A and Technical Breakthroughs
The chemical industry is constantly evolving, driven by the need for more efficient, stable, and versatile intermediates that can support the next generation of high-performance materials. Patent CN113150018A introduces a groundbreaking methodology for the preparation of four-coordinate N,N-chelated monoaryl monofluoroborate compounds, which are critical components in the fields of photoelectric materials, organic solar energy, and advanced dye synthesis. This patent addresses significant limitations in prior art by utilizing acylated 8-aminoquinoline derivatives as stable chelating ligands and potassium aryl trifluoroborate as a safe, robust aryl source. The innovation lies in the ability to conduct this substitution reaction at moderate temperatures of 90-120°C in the presence of cheap metallic accelerators and organic additives, thereby realizing rapid and efficient synthesis without the need for hazardous strong alkali metal organic reagents. This technical leap not only expands the diversity of available borate compounds but also provides a solid material basis for wider application in industries requiring high-purity electronic chemicals and pharmaceutical intermediates. For R&D directors and procurement managers alike, this represents a shift towards greener, more cost-effective manufacturing processes that do not compromise on the structural integrity or purity of the final product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of four-coordinate N,N-chelating borate derivatives has been constrained by the reliance on highly reactive and unstable reagents that pose significant safety and operational challenges in a commercial setting. Traditional methods often require the use of aryl metal reagents such as Grignard reagents, lithium reagents, or zinc reagents to introduce the aryl group onto the boron atom. These reagents are characterized by strong basicity and high nucleophilicity, which impose strict requirements on reaction conditions, such as the need for extremely low temperatures and strictly anhydrous environments. Furthermore, the aggressive nature of these reagents leads to poor compatibility with common functional groups; for instance, ester groups, aldehyde carbonyl groups, and amide groups often cannot survive the reaction conditions, leading to side reactions and reduced yields. This limitation severely restricts the structural diversity of the resulting borate compounds, making it difficult for manufacturers to tailor materials for specific optoelectronic or pharmaceutical applications without engaging in complex protection and deprotection strategies that drive up costs and extend lead times.
The Novel Approach
In stark contrast to the hazardous and limited conventional pathways, the novel approach detailed in patent CN113150018A leverages the stability of potassium aryl trifluoroborate to overcome the reactivity issues associated with traditional organometallics. By selecting an acylated 8-aminoquinoline derivative with unique stability and ready availability as the supporting chelating ligand, the method ensures that the reaction proceeds smoothly under much milder conditions. The use of stable potassium aryl trifluoroborate as the aryl source avoids the use of metal reagents entirely, thereby improving synthesis efficiency and expanding the diversity of the compound library. The reaction is facilitated by a cheap metallic accelerator and an organic additive, allowing for substitution at 90-120°C, which is significantly more energy-efficient and safer than cryogenic conditions. 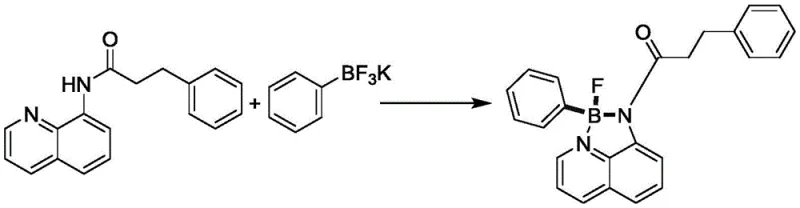 This shift in methodology not only reduces the experimental operation difficulty but also enhances the compatibility of functional groups under reaction conditions, allowing for the synthesis of complex molecules that were previously inaccessible. For supply chain heads, this means a more reliable process with fewer failure points and a broader substrate applicability that can adapt to varying raw material availabilities without re-engineering the entire process.
This shift in methodology not only reduces the experimental operation difficulty but also enhances the compatibility of functional groups under reaction conditions, allowing for the synthesis of complex molecules that were previously inaccessible. For supply chain heads, this means a more reliable process with fewer failure points and a broader substrate applicability that can adapt to varying raw material availabilities without re-engineering the entire process.
Mechanistic Insights into N,N-Chelating Boron Coordination
The core of this technological advancement lies in the precise mechanistic control over the boron atom's coordination environment, ensuring the formation of a stable four-coordinate structure with high selectivity. The acylated 8-aminoquinoline derivative acts as a bidentate ligand, coordinating to the boron center through its nitrogen atoms to form a rigid N,N-chelating framework. This chelation stabilizes the boron atom, preventing unwanted decomposition or side reactions that are common in less constrained borate systems. The reaction mechanism involves the activation of the potassium aryl trifluoroborate by the organic additive, which facilitates the transfer of the aryl group to the boron center in the presence of the metallic accelerator. This process is highly atom-economical, as it minimizes the generation of waste byproducts and maximizes the incorporation of starting materials into the final product. 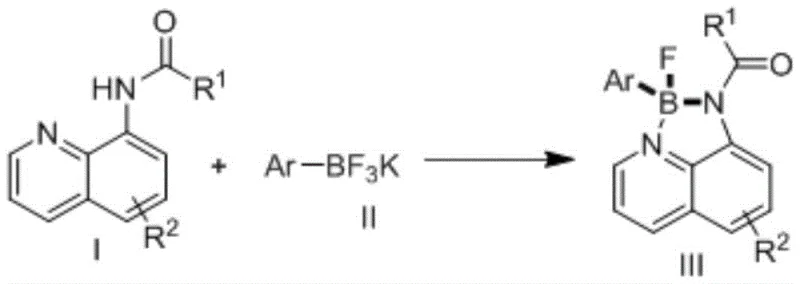 The use of inert gases such as nitrogen or argon as a protective atmosphere further ensures that the reaction proceeds without oxidation or moisture interference, which is critical for maintaining the integrity of the fluoroborate moiety. For R&D teams, understanding this mechanism is key to optimizing reaction parameters such as temperature, time, and molar ratios to achieve the highest possible purity and yield for their specific target molecules.
The use of inert gases such as nitrogen or argon as a protective atmosphere further ensures that the reaction proceeds without oxidation or moisture interference, which is critical for maintaining the integrity of the fluoroborate moiety. For R&D teams, understanding this mechanism is key to optimizing reaction parameters such as temperature, time, and molar ratios to achieve the highest possible purity and yield for their specific target molecules.
Impurity control is another critical aspect of this synthesis, as the presence of residual metals or unreacted starting materials can severely impact the performance of the final material in sensitive applications like organic solar cells. The patent outlines a robust purification protocol that involves filtering the reaction mixture to remove solid catalysts, followed by washing with dichloromethane to eliminate soluble impurities. The crude product is then subjected to rotary evaporation to remove solvents, and finally purified by silica gel column chromatography using a specific eluent system of petroleum ether and ethyl acetate. This multi-step purification ensures that the final tetra-coordinated N,N-chelated monoaryl monofluoroborate compound meets stringent purity specifications required for high-end electronic materials. The ability to consistently remove impurities such as dichloromethane residues and metal salts is a testament to the process's scalability and reliability, providing procurement managers with confidence in the quality consistency of the supply.
How to Synthesize Four-Coordinate N,N-Chelated Monoaryl Monofluoroborate Efficiently
Implementing this synthesis route in a production environment requires careful attention to the mixing ratios and reaction conditions to ensure optimal performance and safety. The process begins with the uniform mixing of the acylated 8-aminoquinoline derivative, potassium aryl trifluoroborate, additive, and accelerator in a suitable organic solvent such as toluene, acetonitrile, or tetrahydrofuran. The detailed standardized synthesis steps involve maintaining the reaction temperature between 90-120°C for a duration of 12 to 48 hours, depending on the specific substrate reactivity. It is crucial to maintain an inert atmosphere throughout the reaction to prevent degradation of the sensitive intermediates. The following guide outlines the critical operational parameters derived from the patent data to assist technical teams in replicating this high-efficiency process.
- Mix acylated 8-aminoquinoline derivative, potassium aryl trifluoroborate, organic additive, and accelerator in an organic solvent under protective gas.
- React the mixture at 90-120°C for 12 to 48 hours to ensure complete conversion and high selectivity.
- Filter the reaction solution, wash with dichloromethane, and purify via silica gel column chromatography to obtain the final high-purity compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis method offers substantial benefits for procurement and supply chain teams looking to optimize costs and ensure continuity of supply for critical chemical intermediates. The elimination of hazardous and expensive Grignard or lithium reagents significantly reduces the raw material costs associated with the synthesis, as potassium aryl trifluoroborate is not only more stable but also generally more affordable and easier to handle in bulk quantities. This shift in reagent strategy also simplifies the safety protocols required for storage and handling, reducing the overhead costs related to specialized containment and waste disposal. For procurement managers, this translates into a more predictable cost structure and reduced risk of supply disruptions caused by the volatility of highly reactive organometallic reagents. The process's ability to operate at moderate temperatures further contributes to energy savings, making the overall manufacturing process more sustainable and economically viable in the long term.
- Cost Reduction in Manufacturing: The replacement of strong alkali metal organic reagents with stable potassium aryl trifluoroborate eliminates the need for expensive cryogenic equipment and rigorous anhydrous conditions, leading to drastic simplification of the production infrastructure. By avoiding the use of costly transition metal catalysts that often require complex removal steps, the process inherently lowers the cost of goods sold through reduced material consumption and simplified downstream processing. The high atom economy of the reaction ensures that a greater proportion of raw materials are converted into the final product, minimizing waste and maximizing value extraction from every kilogram of input. This qualitative improvement in process efficiency allows manufacturers to offer more competitive pricing without compromising on the quality or purity of the final electronic chemical intermediates.
- Enhanced Supply Chain Reliability: The use of stable and readily available raw materials such as acylated 8-aminoquinoline derivatives and potassium aryl trifluoroborate significantly enhances the reliability of the supply chain by reducing dependence on specialized reagents with long lead times. The robustness of the reaction conditions means that production can be maintained consistently even with minor fluctuations in raw material quality, ensuring a steady flow of products to meet market demand. This stability is crucial for supply chain heads who need to guarantee delivery schedules to downstream clients in the fast-paced electronics and pharmaceutical sectors. The simplified purification process also reduces the bottleneck potential in the final stages of production, allowing for faster turnaround times and improved responsiveness to urgent orders.
- Scalability and Environmental Compliance: The method's adherence to green chemistry principles, such as high atom economy and the avoidance of toxic heavy metals, makes it highly scalable and compliant with increasingly stringent environmental regulations. The ability to scale from milligram to multi-ton production without significant changes to the core reaction chemistry ensures that the process can grow with market demand without requiring massive capital reinvestment in new technology. The reduced generation of hazardous waste and the use of common solvents like toluene and acetonitrile simplify waste treatment processes, lowering the environmental footprint of the manufacturing facility. This alignment with sustainability goals not only mitigates regulatory risk but also enhances the brand value of the supplier in the eyes of environmentally conscious global partners.
Frequently Asked Questions (FAQ)
The following questions and answers are derived directly from the technical details and beneficial effects outlined in patent CN113150018A, providing clarity on the practical implementation and advantages of this synthesis method. These insights are intended to address common concerns regarding reaction compatibility, yield consistency, and scalability for industrial applications. Understanding these technical nuances is essential for stakeholders evaluating the feasibility of integrating this new chemistry into their existing supply chains or R&D pipelines. The data presented here reflects the robust performance of the method across a variety of substrates, confirming its versatility and reliability for commercial production.
Q: What are the advantages of using potassium aryl trifluoroborate over Grignard reagents?
A: Potassium aryl trifluoroborate offers superior stability and functional group compatibility compared to Grignard reagents, which are highly basic and nucleophilic, often limiting substrate scope.
Q: What is the typical yield range for this synthesis method?
A: The patent data indicates yields ranging from 54% to 87% across various substrates, demonstrating robust efficiency for complex borate derivatives.
Q: Is this process scalable for industrial production?
A: Yes, the method uses stable raw materials and standard purification techniques like silica gel chromatography, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Four-Coordinate N,N-Chelated Monoaryl Monofluoroborate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in driving innovation within the electronic materials and pharmaceutical sectors. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN113150018A can be seamlessly translated into industrial reality. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of four-coordinate N,N-chelated monoaryl monofluoroborate compound meets the exacting standards required for optoelectronic applications. Our CDMO capabilities allow us to adapt this novel synthesis route to your specific needs, providing a reliable source of complex borate derivatives that can accelerate your product development cycles.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this advanced chemistry. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments that will demonstrate the tangible benefits of partnering with a leader in fine chemical synthesis. By leveraging our expertise and this patented technology, you can secure a competitive advantage in the market through access to superior materials and more efficient production processes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
