Advanced Synthesis of Finasteride Intermediate: Scalable, Green, and Cost-Efficient Manufacturing Solutions
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates that balance high purity with environmental sustainability. Patent CN102241589B introduces a groundbreaking method for synthesizing A-nor-3,5-cracking-androstane-5-ketone-3,17 beta-diacid, a pivotal precursor in the manufacturing of Finasteride. This advanced protocol replaces hazardous traditional oxidants with a green catalytic system based on hydrogen peroxide and tungstate complexes. By shifting away from stoichiometric heavy metal oxidants, this technology offers a safer, more cost-effective pathway for producing high-purity pharmaceutical intermediates. The structural integrity of the final molecule is crucial for the efficacy of the downstream API, and this method ensures minimal impurity formation.
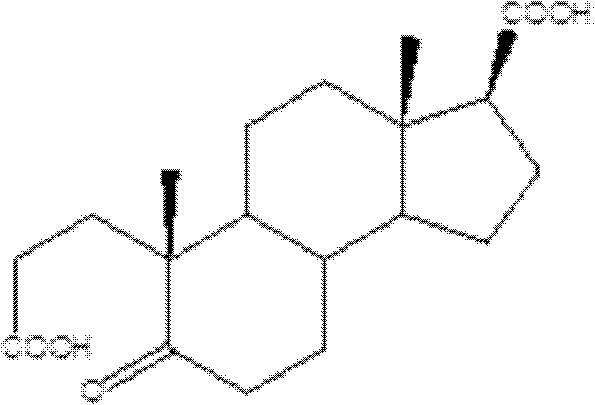
For R&D Directors and Procurement Managers, the implications of this patent extend beyond simple chemistry; it represents a strategic opportunity to optimize supply chains. The ability to source intermediates produced via this method means accessing materials with superior purity profiles (>99%) without the burden of complex heavy metal removal steps. This directly translates to reduced processing time and lower operational expenditures. As the demand for Finasteride remains steady for treating benign prostatic hyperplasia, securing a reliable supply of its key intermediates through such efficient synthetic routes is essential for maintaining market competitiveness and regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of A-nor-3,5-cracking-androstane-5-ketone-3,17 beta-diacid has relied on oxidative cleavage using reagents such as sodium periodate and potassium permanganate. These conventional methods present significant drawbacks for large-scale manufacturing. Firstly, the use of potassium permanganate generates substantial amounts of manganese dioxide sludge, a heavy metal waste that requires expensive and rigorous disposal procedures to meet environmental regulations. Secondly, sodium periodate is a costly reagent, and its stoichiometric usage drives up the raw material costs significantly. Furthermore, the purification processes associated with these methods are often complex, requiring multiple extraction and chromatography steps to remove metal residues, which inevitably lowers the overall yield and increases production lead times.
The Novel Approach
In stark contrast, the method disclosed in CN102241589B utilizes a catalytic system involving hydrogen peroxide and tungstate or molybdate salts. This approach fundamentally alters the economic and environmental landscape of the synthesis. Hydrogen peroxide serves as a clean oxidant, producing water as the only byproduct, thereby eliminating the generation of toxic heavy metal sludge. The catalytic nature of the tungstate species means that only minute quantities are required compared to the stoichiometric amounts of permanganate needed previously. This shift not only drastically reduces raw material costs but also simplifies the work-up procedure. The reaction conditions are mild, typically ranging from 20°C to 100°C, which reduces energy consumption and enhances operational safety within the plant.
Mechanistic Insights into Tungstate-Catalyzed Oxidative Cleavage
The core of this technological advancement lies in the formation of active peroxo-metal species in situ. When hydrogen peroxide interacts with tungstate or molybdate ions in the presence of an acid promoter, it generates highly reactive peroxo-tungstate complexes. These complexes act as electrophilic oxidants that selectively attack the electron-rich double bond in the 3-carbonyl-4-androstene-17-carboxylic acid substrate. The mechanism proceeds through the formation of a cyclic intermediate, which subsequently undergoes cleavage to yield the desired diacid product. This catalytic cycle is highly efficient, allowing the metal species to turnover multiple times, which is a key factor in achieving the reported high yields of 95% to 99.5%.
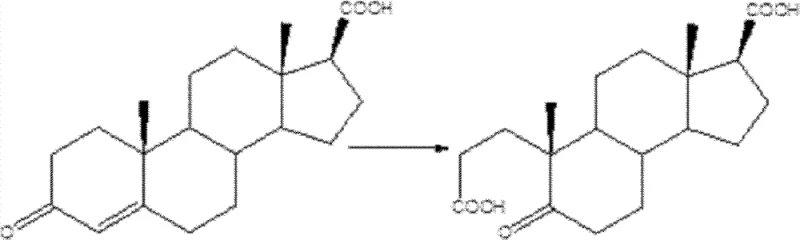
From an impurity control perspective, this mechanism offers superior selectivity compared to radical-based oxidations. The mild nature of the peroxo-metal complex minimizes over-oxidation or degradation of sensitive functional groups elsewhere in the steroid skeleton. This high chemoselectivity ensures that the impurity profile of the crude product is clean, often allowing for purification via simple recrystallization rather than complex chromatographic separation. For quality control teams, this means more consistent batch-to-batch purity, often exceeding 99% as confirmed by HPLC analysis. The ability to control the reaction precisely through temperature and acid concentration further enhances the robustness of the process, making it ideal for GMP manufacturing environments.
How to Synthesize A-nor-3,5-cracking-androstane-5-ketone-3,17 beta-diacid Efficiently
Implementing this synthesis requires careful attention to the molar ratios of the catalyst and acid promoter to ensure optimal turnover of the hydrogen peroxide. The process begins with dissolving the starting steroid material in a solvent such as acetone or 1,4-dioxane, followed by the sequential addition of the oxidant and catalyst system. Detailed standardized synthetic steps see the guide below.
- Dissolve 3-carbonyl-4-androstene-17-carboxylic acid in a suitable organic solvent such as acetone or dioxane under inert atmosphere.
- Add hydrogen peroxide and a metal catalyst like sodium tungstate along with an acid promoter such as phosphoric acid.
- Maintain reaction temperature between 20°C to 100°C, then quench with a reducing agent and purify via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this synthetic route offers tangible strategic benefits that go beyond mere chemical efficiency. The elimination of expensive and regulated heavy metal oxidants directly impacts the cost of goods sold (COGS). By replacing sodium periodate and potassium permanganate with commodity chemicals like hydrogen peroxide and sodium tungstate, the raw material cost structure is significantly optimized. Additionally, the reduction in hazardous waste generation lowers the environmental compliance costs associated with waste disposal and treatment, contributing to substantial overall cost savings in manufacturing operations.
- Cost Reduction in Manufacturing: The shift from stoichiometric heavy metal oxidants to a catalytic hydrogen peroxide system removes the need for purchasing high-cost reagents like sodium periodate. Furthermore, the simplified purification process reduces solvent consumption and labor hours required for work-up. The ability to recycle solvents such as acetone or dichloromethane further enhances the economic viability of the process. These factors combine to create a leaner production model that maximizes margin potential without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: The reagents required for this method, including hydrogen peroxide, tungstate salts, and common organic solvents, are widely available commodity chemicals. This reduces the risk of supply chain disruptions often associated with specialized or regulated reagents. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent production output. This reliability is crucial for maintaining continuous supply to downstream API manufacturers and meeting strict delivery schedules.
- Scalability and Environmental Compliance: The absence of heavy metal sludge simplifies the environmental permitting process for manufacturing facilities. Waste streams are easier to treat, reducing the burden on effluent treatment plants. The mild reaction temperatures and pressures make the process inherently safer to scale up from pilot plant to commercial production volumes. This scalability ensures that supply can be ramped up quickly to meet market demand surges without requiring significant capital investment in specialized safety infrastructure or waste management systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic method. These answers are derived directly from the patent data and practical manufacturing considerations to provide clarity for potential partners.
Q: How does this method improve upon traditional oxidative cleavage techniques?
A: Traditional methods rely on expensive and toxic oxidants like sodium periodate and potassium permanganate, which generate heavy metal waste. This novel approach utilizes hydrogen peroxide and recyclable tungstate catalysts, significantly reducing environmental impact and purification complexity.
Q: What is the expected purity and yield of the final intermediate?
A: The patented process consistently achieves yields between 95% and 99.5%, with final product purity exceeding 99% after simple recrystallization, ensuring high quality for downstream API synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (20-100°C) and utilize readily available commodity chemicals. The absence of hazardous heavy metal sludge simplifies waste treatment, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable A-nor-3,5-cracking-androstane-5-ketone-3,17 beta-diacid Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of securing high-quality intermediates for your pharmaceutical pipeline. Our expertise lies in translating complex patent technologies like CN102241589B into robust, commercial-scale processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of A-nor-3,5-cracking-androstane-5-ketone-3,17 beta-diacid meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for Finasteride production. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with us, you gain access to not just a product, but a comprehensive solution that includes specific COA data and route feasibility assessments. Contact us today to discuss how we can support your long-term manufacturing goals with reliable, cost-effective, and high-purity intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
