Industrial Synthesis of 2-(7-Methoxy-1-Naphthyl)Ethylamine: A Breakthrough in Safe and Scalable Pharmaceutical Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates, particularly for high-value antidepressants like Agomelatine. Patent CN102234238B introduces a transformative methodology for preparing 2-(7-methoxy-1-naphthyl)ethylamine, a pivotal precursor in the synthesis of this novel melatonergic antidepressant. Unlike conventional pathways that rely on hazardous high-pressure hydrogenation, this innovation utilizes a Raney-Ni catalyzed reduction system combined with borohydride reagents. This technical advancement addresses long-standing challenges in process safety and impurity control, offering a pathway that operates under mild ambient conditions. For R&D directors and process chemists, this represents a significant opportunity to optimize manufacturing protocols, ensuring that the production of high-purity pharmaceutical intermediates aligns with modern safety standards and efficiency metrics without compromising yield or quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
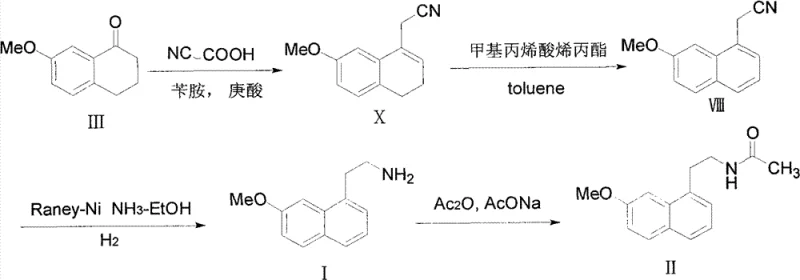
Historically, the synthesis of 2-(7-methoxy-1-naphthyl)ethylamine has been fraught with operational complexities and safety concerns. Prior art, such as the methods described in European patent EP0447285 and Chinese patent CN1680284, typically involves the reduction of nitrile precursors using catalytic hydrogenation under rigorous conditions. These legacy processes often necessitate hydrogen pressures as high as 30 bar and temperatures around 40°C, requiring specialized high-pressure reactors and stringent safety protocols that increase capital expenditure. Furthermore, these high-energy conditions frequently promote side reactions, leading to the formation of dimeric byproducts that are structurally similar to the target molecule. The presence of these impurities complicates the purification process, often requiring multiple recrystallization steps or chromatography, which drastically reduces overall throughput and increases solvent consumption, thereby inflating the cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
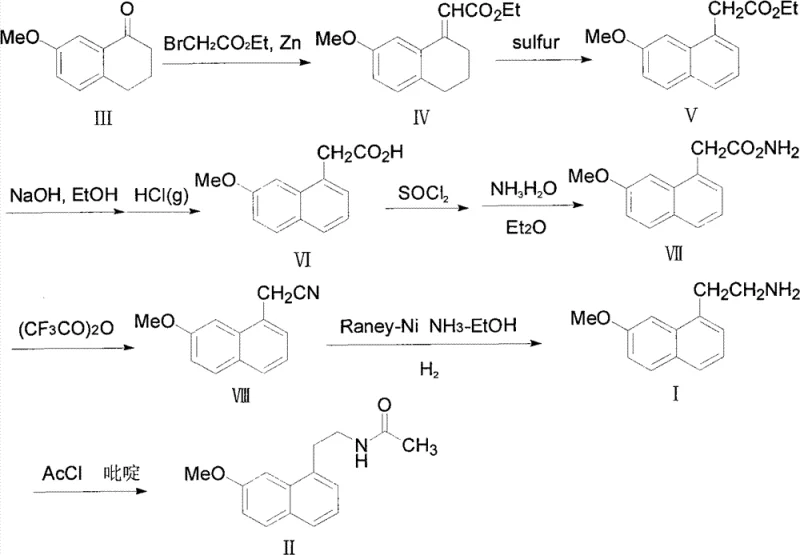
In stark contrast, the methodology disclosed in CN102234238B offers a streamlined and safer alternative that circumvents the need for high-pressure equipment. By employing Raney-Ni in conjunction with a borohydride reducing agent, such as potassium borohydride, the reaction proceeds efficiently at normal temperature and pressure. This shift from high-pressure hydrogen gas to a chemical reduction system significantly lowers the barrier to entry for manufacturing, allowing facilities without specialized high-pressure infrastructure to produce this critical intermediate. The mild reaction conditions not only enhance operational safety by eliminating explosion risks associated with high-pressure hydrogen but also improve the selectivity of the reduction. This results in a cleaner reaction profile where the target amine is generated with minimal side reactions, facilitating a much simpler work-up procedure that is highly advantageous for cost reduction in API manufacturing and supply chain optimization.
Mechanistic Insights into Raney-Ni Catalyzed Reduction
The core of this technological breakthrough lies in the synergistic interaction between the Raney-Ni catalyst and the borohydride species within an alcoholic solvent medium. Mechanistically, the Raney-Ni surface activates the borohydride anion, facilitating the transfer of hydride ions to the electrophilic carbon of the nitrile group in Compound VIII. This catalytic cycle is highly efficient under mild thermal conditions, typically between 0°C and 30°C, which prevents the thermal degradation of sensitive functional groups often seen in harsher reduction methods. The choice of solvent, preferably lower alcohols like ethanol or methanol, plays a crucial role in stabilizing the transition states and ensuring the solubility of both the organic substrate and the inorganic reagents. This precise control over the reaction environment allows for the complete conversion of the nitrile to the primary amine without over-reduction or hydrogenolysis of the methoxy group, ensuring the structural integrity of the naphthalene core is maintained throughout the synthesis.
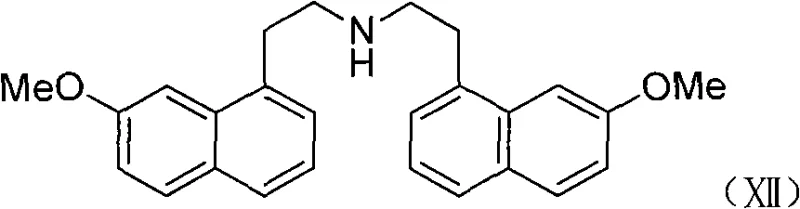
Furthermore, a critical advantage of this mechanism is the suppression of dimerization side reactions that plague traditional hydrogenation routes. In conventional high-pressure processes, the intermediate imine species can react with unreacted nitriles or amines to form secondary or tertiary amine byproducts, such as the dimeric structure shown in the patent data. The Raney-Ni borohydride system appears to favor a direct reduction pathway that rapidly converts the intermediate imine to the final amine, thereby minimizing the residence time of reactive intermediates that could lead to oligomerization. This inherent selectivity is paramount for R&D teams focused on impurity profiling, as it eliminates the need for complex chromatographic separations to remove these structurally similar byproducts. The result is a process that delivers high-purity 2-(7-methoxy-1-naphthyl)ethylamine with yields consistently exceeding 90%, providing a reliable foundation for downstream acetylation to Agomelatine.
How to Synthesize 2-(7-Methoxy-1-Naphthyl)Ethylamine Efficiently
Implementing this synthesis route requires careful attention to reagent addition and temperature control to maximize the benefits of the Raney-Ni catalytic system. The process begins by dissolving the nitrile precursor in a suitable alcohol solvent, followed by the addition of the catalyst and the controlled introduction of the borohydride reducing agent. Maintaining the reaction temperature within the optimal range is critical to balance reaction rate and selectivity, ensuring that the exothermic nature of the reduction is managed safely. While the general procedure is straightforward, specific parameters regarding catalyst loading and stoichiometry are essential for reproducibility on a commercial scale. For detailed operational parameters, stoichiometry ratios, and step-by-step work-up instructions, please refer to the standardized synthesis guide provided below which outlines the exact protocol for industrial implementation.
- Dissolve the nitrile precursor (Compound VIII) in a C1-C4 alcohol solvent such as ethanol or methanol within a standard reaction vessel.
- Add Raney-Ni catalyst and cool the mixture to between 0°C and 10°C before introducing potassium borohydride in batches.
- Allow the reaction to proceed at room temperature for 18 to 28 hours, followed by filtration, extraction, and concentration to isolate the high-purity amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented synthesis route offers substantial strategic advantages that extend beyond mere technical feasibility. The elimination of high-pressure hydrogenation equipment translates directly into reduced capital expenditure and lower maintenance costs for manufacturing facilities, as standard glass-lined or stainless steel reactors can be utilized instead of specialized autoclaves. This flexibility allows for a more agile supply chain, enabling production to be scaled up or shifted between different manufacturing sites without the constraint of high-pressure infrastructure. Additionally, the use of common, commercially available reagents like Raney-Ni and potassium borohydride ensures a stable supply of raw materials, mitigating the risk of supply chain disruptions that can occur with specialized catalysts or high-purity hydrogen gas. These factors collectively contribute to a more resilient and cost-effective supply chain for pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The simplified process design significantly lowers operational costs by removing the need for expensive high-pressure safety systems and reducing energy consumption associated with heating and pressurization. The high selectivity of the reaction minimizes waste generation and solvent usage during purification, leading to substantial cost savings in raw material consumption and waste disposal. Furthermore, the improved yield reduces the amount of starting material required per unit of final product, enhancing the overall economic efficiency of the manufacturing process and allowing for more competitive pricing strategies in the global market.
- Enhanced Supply Chain Reliability: By relying on ambient pressure and temperature conditions, the manufacturing process becomes less susceptible to equipment failures and safety shutdowns that can disrupt production schedules. The robustness of the Raney-Ni catalyst and the stability of the reaction conditions ensure consistent batch-to-batch quality, which is critical for maintaining long-term supply agreements with pharmaceutical clients. This reliability reduces the risk of stockouts and ensures a continuous flow of high-quality intermediates, supporting the uninterrupted production of downstream active pharmaceutical ingredients and finished dosage forms.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of standard organic solvents facilitate easy scale-up from laboratory to commercial production without significant process re-engineering. The reduction in byproduct formation simplifies waste treatment processes, aligning with increasingly stringent environmental regulations and sustainability goals. This environmentally friendly profile not only reduces regulatory compliance costs but also enhances the corporate social responsibility image of the manufacturer, making it a preferred partner for global pharmaceutical companies committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the patent data and practical manufacturing experience, providing clarity on safety, purity, and scalability. Understanding these aspects is crucial for decision-makers evaluating the feasibility of adopting this technology for their supply chains. The information provided here serves as a preliminary guide, and specific technical consultations are recommended for detailed process validation and customization.
Q: How does the Raney-Ni method improve safety compared to traditional hydrogenation?
A: Traditional methods often require high-pressure hydrogenation (up to 30 bar) and elevated temperatures, posing significant safety risks. The patented Raney-Ni method operates at ambient temperature and pressure, eliminating the need for specialized high-pressure reactors and reducing operational hazards.
Q: What is the impact of this method on product purity and impurity profiles?
A: Conventional high-pressure routes often generate difficult-to-remove dimeric byproducts. This novel approach selectively reduces the nitrile group without forming these side products, significantly simplifying downstream purification and ensuring higher final purity suitable for pharmaceutical applications.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the method uses commercially available reagents like Raney-Ni and potassium borohydride in common alcohol solvents. The mild conditions and simple work-up procedure make it highly suitable for large-scale industrial manufacturing without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(7-Methoxy-1-Naphthyl)Ethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the production of high-value pharmaceutical intermediates. Our technical team has extensively analyzed the methodology described in CN102234238B and possesses the expertise to implement this Raney-Ni catalyzed reduction on a commercial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-(7-methoxy-1-naphthyl)ethylamine meets the exacting standards required for API synthesis. We are committed to delivering not just a chemical product, but a reliable supply solution that enhances your production efficiency.
We invite procurement leaders and R&D directors to collaborate with us to optimize your supply chain for Agomelatine intermediates. By leveraging our technical capabilities and this advanced synthesis route, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us partner with you to secure a stable, high-quality, and cost-effective supply of this critical intermediate, driving value and innovation in your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
