Advanced Rhodium-Catalyzed Synthesis of High-Optical-Purity Gem-Difluoro Cyclopropane Intermediates
The pharmaceutical and agrochemical industries are increasingly relying on fluorinated building blocks to enhance the metabolic stability and bioavailability of bioactive molecules. Patent CN115043704A introduces a groundbreaking methodology for the preparation of high-optical-purity gem-difluoro cyclopropanes, addressing a critical bottleneck in the synthesis of complex fluorinated intermediates. This technology leverages a sophisticated rhodium-catalyzed kinetic resolution strategy to transform readily available racemic starting materials into enantiomerically enriched products with exceptional efficiency. The ability to introduce a gem-difluoro cyclopropyl group into protein inhibitors has been shown to significantly improve oral bioavailability while reducing cytotoxicity, making this synthetic route highly valuable for modern drug discovery pipelines. By overcoming the limitations of previous asymmetric synthesis methods, this invention provides a robust platform for generating chiral quaternary and tertiary carbon centers essential for next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of gem-difluoro cyclopropanes has been plagued by significant stereochemical challenges that hinder their widespread application in commercial manufacturing. Most conventional methods rely on the in situ generation of gem-difluorocarbene precursors which subsequently add to alkenes, a process that inherently produces racemic mixtures devoid of optical purity. While chiral column chromatography can theoretically separate these enantiomers, it is an inefficient, low-throughput technique that is economically unviable for large-scale production. Other existing asymmetric approaches, such as lipase-catalyzed kinetic resolution or copper-catalyzed hydrogenation, suffer from narrow substrate scope and often fail to achieve high enantiomeric excess when applied to complex molecular architectures. Furthermore, many prior art methods require harsh reaction conditions or specialized substrates containing specific functional groups like esters or amides, limiting their utility in diverse medicinal chemistry campaigns.
The Novel Approach
In stark contrast to these restrictive legacy technologies, the method disclosed in CN115043704A utilizes a versatile rhodium-catalyzed system capable of resolving a broad spectrum of racemic gem-difluoro cyclopropane substrates. This novel approach employs a carefully tuned combination of a monovalent rhodium metal compound and a chiral bisphosphine ligand to induce high levels of stereoselectivity under relatively mild thermal conditions. The process is not limited to specific functional groups, allowing for the successful kinetic resolution of substrates containing various substituents on the aromatic rings and cyclopropane moiety. As illustrated in the general reaction scheme below, this methodology accommodates diverse R-groups, enabling the synthesis of both quaternary and tertiary carbon centers with high fidelity. This flexibility makes it an ideal candidate for the late-stage functionalization of complex drug candidates where structural diversity is paramount.
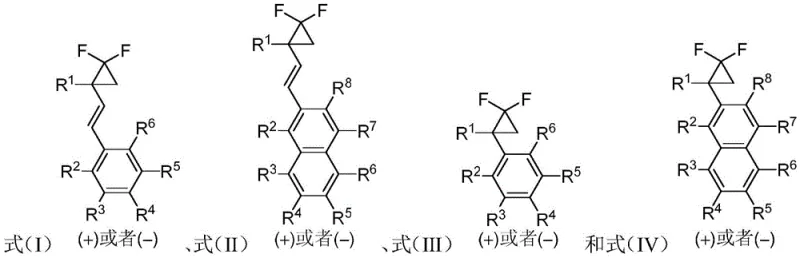
Mechanistic Insights into Rhodium-Catalyzed Kinetic Resolution
The core of this technological breakthrough lies in the precise interaction between the rhodium catalyst and the chiral bisphosphine ligand, which creates a highly asymmetric environment for the reaction. The patent specifies the use of monovalent rhodium sources such as dirhodium tetracarbonyl dichloride or (1,5-cyclooctadiene)chlororhodium(I) dimer, paired with bulky chiral ligands like BINAP derivatives or specialized phosphines containing methoxy and tert-butyl groups. These ligands are critical for discriminating between the enantiomers of the racemic starting material, facilitating the selective transformation of one enantiomer while leaving the other intact or converting it at a vastly different rate. The inclusion of aromatic additives, such as anisole or styrene, further modulates the electronic properties of the catalytic cycle, enhancing both the reaction rate and the stereochemical outcome. This synergistic effect allows the reaction to proceed efficiently at temperatures ranging from 70°C to 120°C, avoiding the decomposition issues often associated with more sensitive fluorinated intermediates.
From an impurity control perspective, this catalytic system offers distinct advantages by minimizing the formation of side products that typically complicate downstream purification. The kinetic resolution mechanism ensures that the desired enantiomer is produced with high optical purity, often exceeding 98% ee as demonstrated in the patent examples, thereby reducing the burden on final crystallization or chromatographic steps. The robustness of the rhodium catalyst against various functional groups means that protecting group strategies can often be simplified, leading to shorter synthetic routes and higher overall yields. Moreover, the reaction conditions are sufficiently mild to preserve sensitive moieties within the molecule, ensuring that the structural integrity of complex pharmaceutical intermediates is maintained throughout the synthesis. This level of control is essential for meeting the stringent purity specifications required by regulatory bodies for active pharmaceutical ingredients.
How to Synthesize High-Optical-Purity Gem-Difluoro Cyclopropane Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and maximum yield. The process begins with the precise weighing and mixing of the racemic substrate, the rhodium catalyst, the chiral ligand, and the aromatic additive under an inert atmosphere to prevent catalyst deactivation. Solvent selection is also critical, with tetrahydrofuran or trifluorotoluene being preferred for their ability to dissolve the reactants while maintaining stability at elevated temperatures. The reaction mixture is then heated and stirred for a duration determined by the specific substrate structure, typically ranging from 0.5 hours for simpler olefinic substrates to 7 hours for more sterically hindered aryl systems. Detailed standardized synthesis steps see the guide below.
- Mix racemic gem-difluoro cyclopropane with a monovalent rhodium metal compound, a chiral bisphosphine ligand, and an aromatic additive in a specific molar ratio.
- Add an appropriate solvent to the mixture and heat the reaction system to a temperature range of 70°C to 120°C while stirring.
- Maintain the reaction for 0.5 to 12 hours depending on the substrate structure, followed by purification to isolate the high-optical-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed technology translates into tangible improvements in cost structure and supply reliability for fluorinated intermediates. The use of commercially available catalysts and ligands eliminates the dependency on custom-synthesized reagents that often carry long lead times and exorbitant price tags. Furthermore, the ability to operate at moderate temperatures reduces energy consumption compared to cryogenic processes often required for other asymmetric syntheses, contributing to lower operational expenditures. The high selectivity of the reaction minimizes waste generation and simplifies the isolation of the final product, which directly correlates to reduced solvent usage and lower disposal costs. These factors combined create a more sustainable and economically viable manufacturing process that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The elimination of expensive chiral column chromatography steps represents a significant driver for cost reduction in pharmaceutical intermediate manufacturing. By achieving high optical purity directly through catalytic kinetic resolution, the need for resource-intensive separation techniques is drastically reduced, leading to substantial savings in both materials and labor. Additionally, the high atom economy of the reaction ensures that a greater proportion of the starting material is converted into the desired product, minimizing raw material waste. The use of standard solvents and readily available additives further lowers the input costs, making the overall process highly competitive against traditional synthetic routes that rely on stoichiometric chiral auxiliaries.
- Enhanced Supply Chain Reliability: Sourcing high-purity fluorinated building blocks is often a bottleneck due to the limited number of suppliers capable of handling complex asymmetric synthesis. This method utilizes robust catalysts and simple reaction conditions that are easily transferable to multiple manufacturing sites, thereby diversifying the supply base and mitigating risk. The scalability of the process allows for the production of large batches in a single run, ensuring consistent availability of key intermediates for downstream drug substance manufacturing. This reliability is crucial for maintaining continuous production schedules and avoiding costly delays in the development of new therapeutic agents.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing equipment and conditions that are standard in the fine chemical industry. The mild reaction temperatures and absence of hazardous reagents simplify the engineering controls required for safe operation, facilitating easier scale-up from gram to ton scale. From an environmental perspective, the reduction in solvent waste and the avoidance of heavy metal contaminants often associated with other catalytic systems support stricter environmental compliance standards. This alignment with regulatory requirements ensures smoother audit processes and long-term sustainability for the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the method. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing workflows. The information covers catalyst availability, reaction scope, and purification requirements to assist in decision-making.
Q: What is the primary advantage of this Rhodium-catalyzed method over traditional carbene addition?
A: Traditional carbene addition typically yields racemic mixtures requiring inefficient chiral column separation. This patented Rhodium-catalyzed kinetic resolution directly produces high-optical-purity products with ee values exceeding 98%, significantly streamlining the purification process.
Q: Are the catalysts and ligands used in this process commercially available?
A: Yes, the patent specifies the use of commercially available monovalent rhodium compounds such as dirhodium tetracarbonyl dichloride and standard chiral bisphosphine ligands like BINAP derivatives, ensuring supply chain stability and cost-effectiveness.
Q: Can this synthesis method be scaled for industrial production?
A: The process operates under mild conditions (70°C to 120°C) and uses common solvents like tetrahydrofuran or trifluorotoluene. The patent explicitly states that large quantities can be prepared in a single batch, indicating high scalability for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gem-Difluoro Cyclopropane Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality fluorinated intermediates in the development of innovative medicines and agrochemicals. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical instrumentation to verify every batch. By leveraging advanced catalytic technologies like the one described in CN115043704A, we can offer customized synthesis solutions that optimize both performance and cost for your specific application needs.
We invite you to collaborate with us to explore how this efficient synthetic route can enhance your supply chain and reduce your overall production costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments for your target molecules. Let us help you secure a reliable supply of high-purity intermediates that drive your innovation forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
