Advanced Copper-Catalyzed Synthesis of Optically Active Muscone for Industrial Scale-Up
The global demand for high-quality, nature-identical fragrances has driven significant innovation in the synthesis of musk compounds, particularly optically active muscone. Patent CN101166708B presents a groundbreaking preparation method for optically active 3-methylcyclopentadecanone, a key component known for its superior olfactory profile compared to its racemic counterpart. This technology addresses critical limitations in existing synthetic routes by introducing a novel copper-catalyzed asymmetric methylation strategy. Unlike traditional methods that often suffer from low yields or require harsh reaction conditions, this invention utilizes a specific optically active phosphoramidite ligand in conjunction with an enolate anion scavenger. This combination not only enhances the stereo-selectivity of the 1,4-conjugate addition reaction but also significantly improves process efficiency by allowing for higher substrate concentrations. For industry stakeholders, this represents a pivotal shift towards more sustainable and economically viable production of premium fragrance ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active muscone has been fraught with challenges that hinder cost-effective manufacturing. Conventional approaches often rely on the optical resolution of racemic mixtures, a process inherently limited by a maximum theoretical yield of 50% for the desired enantiomer, leading to substantial material waste and increased processing costs. Alternatively, asymmetric hydrogenation methods have been explored, but these frequently necessitate the use of expensive noble metal catalysts and stringent reaction conditions, such as extremely low temperatures and high dilution, to prevent side reactions and maintain optical purity. These constraints drastically reduce reactor throughput and escalate energy consumption, making the final product prohibitively expensive for widespread commercial application. Furthermore, the generation of unwanted by-products due to the reactivity of intermediate enolate anions often complicates purification, further eroding overall process efficiency and yield.
The Novel Approach
The methodology disclosed in the patent offers a robust solution by leveraging a copper-catalyzed 1,4-conjugate addition of a methyl group to 2-cyclopentadecen-1-one. The core innovation lies in the simultaneous use of a tailored optically active phosphoramidite ligand and an enolate anion scavenger. This dual-component system effectively traps the reactive enolate intermediate as a stable derivative, thereby suppressing intermolecular side reactions that typically plague high-concentration reactions. As a result, the process can operate at significantly higher substrate concentrations and more moderate temperatures compared to prior art. This approach not only boosts the reaction yield to impressive levels, often exceeding 90% in optimized examples, but also simplifies the downstream purification process. By eliminating the need for extreme cryogenic conditions and reducing catalyst loading, this novel route provides a clear pathway for reducing the cost of goods sold (COGS) while ensuring consistent high quality.
Mechanistic Insights into Copper-Catalyzed Asymmetric Methylation
The success of this synthesis hinges on the precise design of the chiral ligand and the management of reaction intermediates. The optically active phosphoramidite ligand, represented by general formula (1) in the patent, features a binaphthyl backbone that induces axial chirality, working in concert with chiral substituents on the nitrogen atom to create a highly defined chiral environment around the copper center. This steric and electronic tuning is critical for directing the incoming methyl group from the organometallic reagent to the specific face of the enone substrate, ensuring high enantioselectivity. The structural versatility of this ligand class allows for fine-tuning by modifying the aryl or alkyl groups attached to the nitrogen, enabling chemists to optimize performance for specific substrates.
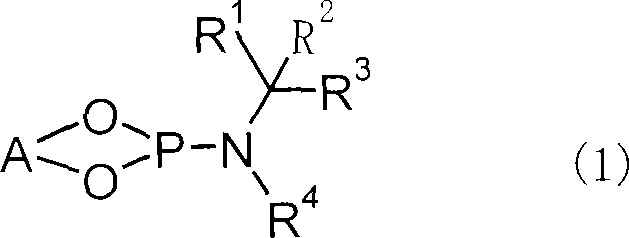
Furthermore, the mechanism incorporates a crucial step involving the capture of the enolate anion generated after the conjugate addition. Without intervention, this anionic species can act as a nucleophile in unwanted side reactions or undergo protonation that leads to racemization. By introducing an enolate anion scavenger, such as an acid anhydride or chlorosilane, the intermediate is converted into a stable enol derivative, such as an enol ester or silyl enol ether. This stabilization prevents degradation and allows the reaction mixture to be warmed to room temperature without loss of optical integrity. The subsequent solvolysis of this protected intermediate under mild acidic or basic conditions releases the final ketone product with retained stereochemistry. This mechanistic strategy decouples the stereoselective bond formation from the final product isolation, providing greater operational flexibility.
How to Synthesize Optically Active 3-Methylcyclopentadecanone Efficiently
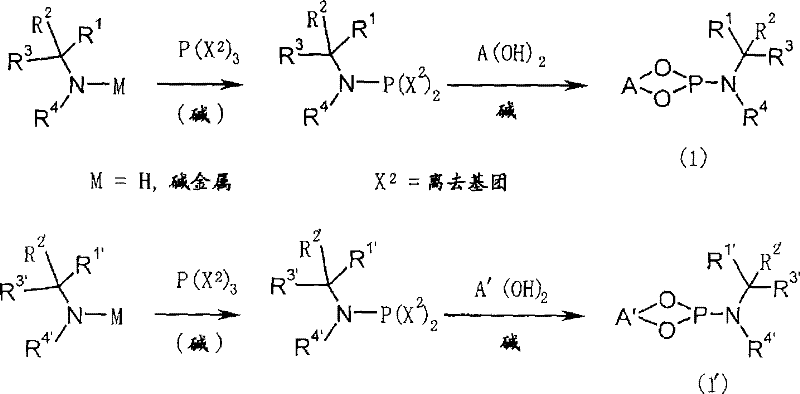
Implementing this technology requires a systematic approach divided into ligand preparation and the main catalytic cycle. The synthesis begins with the construction of the chiral phosphoramidite ligand, which involves reacting a chiral secondary amine with a phosphorus trihalide to form a P-N bond, followed by coupling with an optically active binaphthyl diol in the presence of a base. Once the ligand is secured, the main transformation involves mixing the ligand with a copper salt, such as copper(II) triflate, and a methylating agent like dimethylzinc. To this catalytic mixture, the substrate 2-cyclopentadecen-1-one and the enolate scavenger are added, typically at controlled temperatures ranging from -30°C to room temperature. The reaction progress is monitored to ensure complete conversion before quenching and proceeding to the solvolysis step to reveal the final muscone product.
- Synthesize the optically active phosphoramidite ligand by reacting a chiral secondary amine with a phosphorus halide, followed by coupling with a binaphthyl diol.
- Perform the asymmetric 1,4-conjugate addition of a methyl group to 2-cyclopentadecen-1-one using a copper catalyst and the prepared ligand in the presence of an enolate anion scavenger.
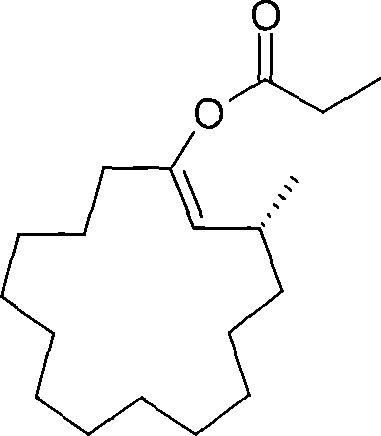
- Subject the resulting optically active enol derivative to solvolysis using a basic or acidic catalyst to yield the final optically active 3-methylcyclopentadecanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the adoption of this patented methodology offers tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the drastic simplification of the manufacturing process, which translates directly into operational cost reductions. By enabling reactions at higher concentrations and avoiding the need for prolonged cryogenic cooling, the process significantly lowers energy consumption and increases reactor capacity utilization. This efficiency gain means that existing infrastructure can produce larger volumes of high-value fragrance intermediates without requiring capital-intensive expansions. Additionally, the high yields reported in the patent examples suggest a substantial reduction in raw material waste, optimizing the usage of expensive chiral starting materials and organometallic reagents.
- Cost Reduction in Manufacturing: The elimination of extreme low-temperature requirements and the ability to use lower catalyst loadings significantly decrease utility and material costs. The high yield minimizes the need for extensive recycling or disposal of unreacted starting materials, leading to a leaner production budget. Furthermore, the use of readily available copper salts instead of precious metals like rhodium or palladium reduces the exposure to volatile commodity prices associated with noble metals.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed runs or off-spec material. The reliance on stable, storable intermediates and common reagents mitigates supply chain risks associated with specialized or hazardous chemicals that have long lead times. This reliability is crucial for maintaining continuous supply to downstream perfume manufacturers who demand strict consistency in odor profiles.
- Scalability and Environmental Compliance: The process is inherently scalable due to its tolerance for higher concentrations and simpler workup procedures. The reduction in solvent usage per unit of product aligns with green chemistry principles, lowering the environmental footprint and simplifying waste treatment compliance. This sustainability aspect is increasingly important for meeting corporate ESG goals and regulatory standards in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is essential for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of the supply source. The answers are derived directly from the experimental data and technical disclosures within the patent documentation.
Q: What is the primary advantage of using an enolate anion scavenger in this synthesis?
A: The use of an enolate anion scavenger suppresses intermolecular side reactions caused by the generated enolate anion, allowing the reaction to proceed at higher substrate concentrations and temperatures while maintaining high optical purity and yield.
Q: What level of optical purity can be achieved with this method?
A: According to the patent data, the method can achieve high optical purity, with specific examples demonstrating enantiomeric excess (ee) values ranging from approximately 86% to over 92%, depending on the specific ligand and reaction conditions employed.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is designed for industrial applicability by avoiding extremely low temperatures and low concentrations typically required in asymmetric synthesis, utilizing relatively small amounts of catalyst, and achieving high yields, which facilitates commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Muscone Supplier
While the patent CN101166708B outlines a superior chemical route, translating laboratory success into commercial reality requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands ready to assist in bridging this gap, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team specializes in optimizing complex asymmetric syntheses, ensuring that the high purity specifications and rigorous QC labs required for premium fragrance ingredients are met consistently. We understand the nuances of handling air-sensitive organometallics and chiral ligands, ensuring safety and efficiency at every stage of the scale-up process.
We invite industry partners to collaborate with us to unlock the full potential of this advanced muscone synthesis. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this technology can enhance your product portfolio and strengthen your supply chain resilience in the competitive flavors and fragrances market.
