Optimizing Agomelatine Intermediate Synthesis: A Strategic Analysis of Patent CN101486665B for Commercial Scale-Up
Optimizing Agomelatine Intermediate Synthesis: A Strategic Analysis of Patent CN101486665B for Commercial Scale-Up
The pharmaceutical landscape for antidepressants has been significantly shaped by the development of agomelatine, a unique melatonin receptor agonist and 5-HT2C antagonist. As demand for this active pharmaceutical ingredient (API) continues to grow, the efficiency of its supply chain relies heavily on the robustness of its intermediate synthesis. Patent CN101486665B introduces a pivotal advancement in the preparation of 2-(7-methoxy-1-naphthyl)acetamide, a critical precursor in the agomelatine value chain. This technical insight report analyzes the strategic implications of this patent for R&D directors, procurement managers, and supply chain leaders, highlighting how a shift from multi-step traditional protocols to a streamlined direct ammonolysis process can redefine cost structures and operational reliability in fine chemical manufacturing.
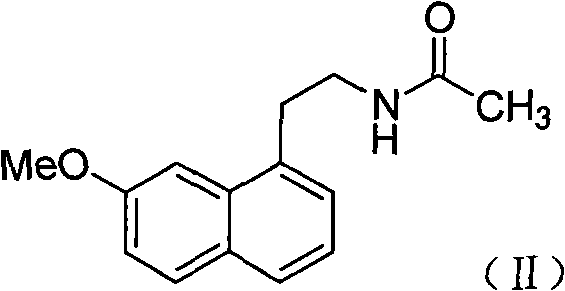
Agomelatine represents a breakthrough in treating major depressive disorder and sleep disorders, necessitating a supply chain capable of delivering high-purity intermediates consistently. The structural complexity of the naphthalene core requires precise synthetic control to avoid impurities that could compromise final drug safety. The methodology outlined in CN101486665B addresses these challenges by focusing on the conversion of ester precursors directly to the target amide, bypassing several hazardous and inefficient transformation stages found in legacy literature. For stakeholders evaluating potential partners, understanding the mechanistic superiority of this route is essential for ensuring long-term supply continuity and regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically European Patent EP0447285, established a foundational but operationally burdensome pathway for synthesizing the target intermediate. This conventional route necessitates a five-step sequence starting from 7-methoxytetralone, involving a Reformatsky reaction with ethyl bromoacetate followed by sulfur-mediated dehydrogenative aromatization. The reliance on sulfur for aromatization introduces significant purification challenges, often resulting in incomplete reactions and difficult-to-remove sulfur-containing byproducts. Furthermore, the initial Reformatsky step is notoriously sensitive to reproducibility issues when transitioning from laboratory to industrial scales, frequently leading to inconsistent yields. The use of benzene as a solvent in these early stages poses severe environmental and occupational health risks, conflicting with modern green chemistry mandates and increasing waste disposal costs for manufacturers.
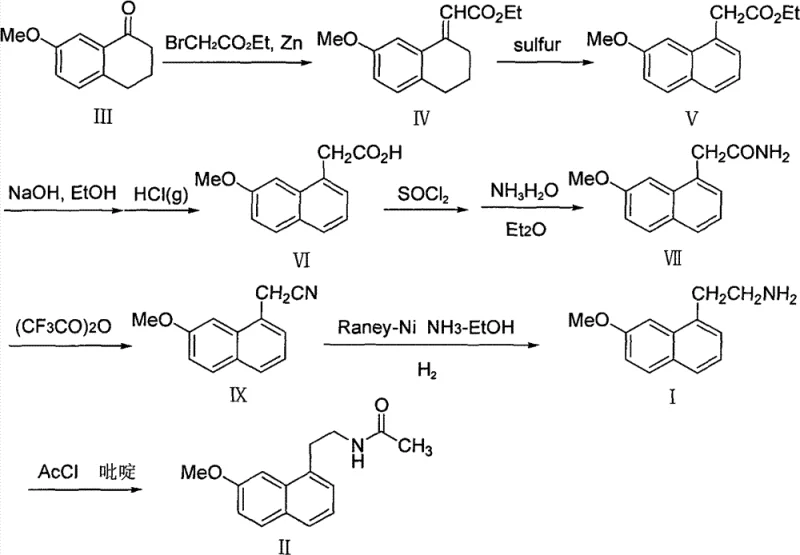
Beyond the solvent issues, the conventional pathway requires subsequent hydrolysis, acyl chloride formation, ammoniation, dehydration, and reduction steps before reaching the final amine precursor. Each additional unit operation introduces potential yield losses and increases the cumulative impurity profile. The saponification step following aromatization often produces mixtures that are arduous to purify, requiring extensive downstream processing that erodes profit margins. For a procurement manager, these inefficiencies translate into higher raw material consumption and longer lead times. For an R&D director, the presence of multiple reactive intermediates increases the risk of side reactions and complicates the control of the impurity spectrum, potentially jeopardizing regulatory filings for the final API.
The Novel Approach
In stark contrast, the methodology disclosed in CN101486665B offers a paradigm shift by condensing the synthesis into a highly efficient direct ammonolysis reaction. The core innovation lies in the direct conversion of (7-methoxy-1-naphthyl) alkyl acetates (Compound A) into 2-(7-methoxy-1-naphthyl)acetamide (Compound VII) using ammonia under anhydrous conditions. This single-step transformation eliminates the need for activating agents like thionyl chloride or hazardous dehydration reagents. By utilizing simple alkyl esters where the protecting group R can be methyl, ethyl, benzyl, or tert-butyl, the process gains remarkable flexibility in sourcing starting materials. The reaction proceeds smoothly in common alcoholic solvents such as methanol or ethanol, completely avoiding the use of carcinogenic benzene or chlorinated solvents often associated with older protocols.
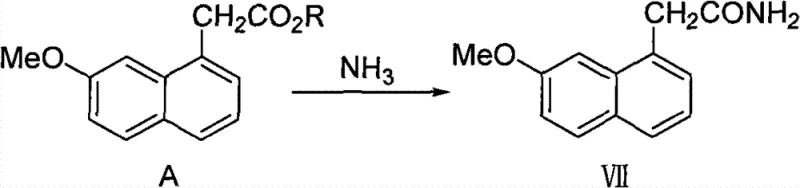
The operational simplicity of this novel approach cannot be overstated. The reaction conditions are mild, typically requiring temperatures that are easily manageable in standard stainless steel reactors, and the reaction time is predictable, ranging from 3 to 24 hours depending on the specific ester substrate. The reported yields are exceptionally high, consistently stabilizing above 95%, which drastically reduces the cost of goods sold (COGS) by minimizing raw material waste. For supply chain heads, this reliability means fewer batch failures and a more predictable production schedule. The elimination of toxic reagents also simplifies the environmental permitting process for manufacturing sites, allowing for faster scale-up and reduced regulatory friction in jurisdictions with strict environmental protections.
Mechanistic Insights into Direct Ammonolysis and DDQ Oxidation
To fully appreciate the technical robustness of this synthesis, one must examine the mechanistic underpinnings of both the precursor formation and the final amidation. The patent describes an optional but highly effective precursor synthesis involving the oxidation of 7-methoxy-tetralone derivatives (Compound B) using 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ). This oxidative aromatization is a critical enabling step that converts the saturated tetralone ring into the aromatic naphthalene system required for the final drug structure. DDQ acts as a potent hydride acceptor, facilitating the removal of hydrogen atoms to establish aromaticity without the need for elemental sulfur. This mechanism avoids the formation of polysulfides and other sulfur-based impurities that plague the conventional route, resulting in a cleaner reaction profile that is easier to monitor and control.
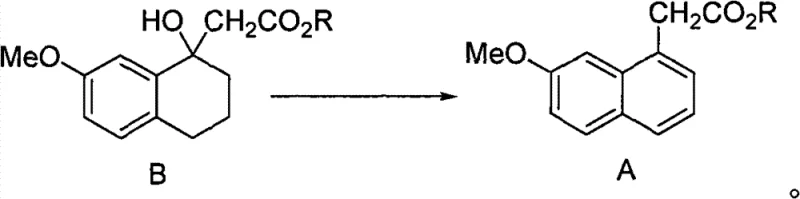
The subsequent ammonolysis mechanism involves a nucleophilic acyl substitution where ammonia attacks the carbonyl carbon of the ester group. Under anhydrous conditions, the equilibrium is driven towards the formation of the amide bond, releasing the corresponding alcohol as a byproduct. The choice of solvent plays a pivotal role here; alcoholic solvents not only dissolve the reactants effectively but also participate in the solvation of the transition state, lowering the activation energy. The absence of water is crucial to prevent the competitive hydrolysis of the ester to the carboxylic acid, which would reduce yield and complicate purification. This mechanistic precision ensures that the impurity profile remains minimal, primarily consisting of unreacted starting material which can be easily removed via recrystallization, a unit operation that is far more scalable and cost-effective than column chromatography.
From an impurity control perspective, the stability of the naphthalene ring system under these mild ammonolysis conditions is a significant advantage. Harsh acidic or basic conditions often used in alternative routes can lead to demethylation of the methoxy group or ring degradation, generating structurally related impurities that are difficult to separate. The neutral to slightly basic environment provided by ammonia in alcohol preserves the integrity of the methoxy substituent. This selectivity is vital for maintaining the pharmacological activity of the downstream API, as even minor structural deviations can alter receptor binding affinity. For quality assurance teams, this inherent selectivity reduces the burden on analytical testing and increases the confidence in batch-to-batch consistency.
How to Synthesize 2-(7-methoxy-1-naphthyl)acetamide Efficiently
Implementing this synthesis in a commercial setting requires strict adherence to the optimized parameters defined in the patent to maximize yield and purity. The process begins with the preparation of the ester precursor, preferably via the DDQ oxidation route for superior purity, followed by the critical ammonolysis step. Operators must ensure that the reaction environment remains strictly anhydrous to prevent hydrolysis side reactions. The selection of the ester group (methyl or ethyl) allows for tuning the reaction kinetics based on available infrastructure, with ethyl esters often providing a balance between reactivity and handling safety. Detailed standard operating procedures (SOPs) should reflect the specific temperature ramps and ammonia flow rates validated in the examples to ensure reproducibility across different batch sizes.
- Oxidize the 7-methoxy-tetralone derivative (Compound B) using DDQ in glacial acetic acid at 50-100°C to form the naphthyl ester (Compound A).
- Dissolve the resulting ester (Compound A) in an alcoholic solvent such as ethanol or methanol under anhydrous conditions.
- Introduce pure ammonia gas into the reaction system and maintain reaction until substrate consumption is complete, typically requiring 3 to 24 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of the technology described in CN101486665B offers tangible economic and logistical benefits that extend beyond simple yield improvements. The reduction in synthetic steps from five to essentially one (for the amidation) fundamentally alters the cost structure of the intermediate. Fewer unit operations mean lower energy consumption, reduced labor hours, and decreased equipment occupancy time, all of which contribute to a leaner manufacturing footprint. The elimination of expensive and hazardous reagents like thionyl chloride and elemental sulfur removes significant cost drivers associated with specialized handling, storage, and waste disposal. This streamlining allows for a more competitive pricing model, enabling suppliers to offer cost reduction in pharmaceutical intermediate manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The primary economic driver is the drastic simplification of the process workflow. By removing the need for acyl chloride formation and subsequent dehydration steps, the process eliminates the capital expenditure required for corrosion-resistant equipment often needed for handling chlorinating agents. Furthermore, the high yield stability above 95% ensures that raw material utilization is maximized, reducing the effective cost per kilogram of the final product. The use of commodity solvents like ethanol and methanol, instead of specialized or regulated solvents like benzene, further lowers the variable costs associated with solvent recovery and procurement. This efficiency translates directly into improved margins for the manufacturer and potential price stability for the buyer.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and reproducibility of the reaction conditions. The conventional Reformatsky reaction is sensitive to moisture and surface activation of zinc, often leading to batch-to-batch variability that can disrupt production schedules. In contrast, the direct ammonolysis described here is chemically robust and less prone to failure due to minor fluctuations in reaction parameters. The availability of diverse ester precursors (methyl, ethyl, benzyl) provides procurement teams with flexibility in sourcing; if one ester variant faces supply constraints, another can be substituted with minimal process re-validation. This redundancy mitigates the risk of supply interruptions and ensures continuous availability of this critical pharma intermediate.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden bottlenecks related to heat transfer and mixing, particularly in exothermic reactions or those involving solids like sulfur. The proposed method operates under mild thermal conditions and homogeneous liquid phases, making it inherently easier to scale from pilot plant to multi-ton production. From an environmental standpoint, the absence of toxic heavy metals, sulfur waste, and carcinogenic solvents aligns perfectly with increasingly stringent global environmental regulations. This compliance reduces the administrative burden of waste reporting and permits, facilitating faster approval for capacity expansions. It also enhances the corporate social responsibility (CSR) profile of the supply chain, a factor of growing importance to downstream pharmaceutical customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing supply networks.
Q: What are the primary limitations of the conventional EP0447285 synthesis route?
A: The conventional route described in EP0447285 involves five distinct steps including a Reformatsky reaction and sulfur dehydrogenation. It suffers from low average yields, reproducibility issues during scale-up, and the use of hazardous solvents like benzene, making it environmentally unfriendly and difficult to purify.
Q: How does the novel ammonolysis method improve process safety and efficiency?
A: The novel method reduces the synthesis of the target amide to a single step from the ester precursor. It eliminates the need for toxic reagents and harsh conditions, utilizes mild reaction parameters, and achieves high yields stable above 95%, significantly simplifying the purification process.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly suitable for industrial application. It avoids highly toxic solvents, uses commercially available reagents like DDQ and ammonia, operates under controllable mild conditions, and provides high purity products without complex chromatographic separations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(7-methoxy-1-naphthyl)acetamide Supplier
As the global demand for mental health therapeutics rises, securing a dependable source for high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands ready to leverage the advancements detailed in CN101486665B to deliver superior value to our partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial reality is seamless. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-(7-methoxy-1-naphthyl)acetamide meets the exacting standards required for API synthesis. Our commitment to green chemistry principles ensures that our manufacturing processes are not only efficient but also sustainable.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that prioritizes both economic efficiency and technical excellence. Contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our optimized synthesis capabilities can enhance your project's timeline and bottom line, establishing a long-term partnership built on trust and scientific rigor.
