Scalable Synthesis of Agomelatine Intermediates via Dual Catalytic Condensation
Scalable Synthesis of Agomelatine Intermediates via Dual Catalytic Condensation
The pharmaceutical industry constantly seeks robust pathways for synthesizing complex antidepressant agents, and the production of Agomelatine represents a significant area of focus due to its unique mechanism of action targeting melatonin receptors. Patent CN102060733A introduces a transformative methodology for preparing the critical intermediate (7-methoxy-3,4-dihydro-1-naphthyl) acetonitrile, achieving reaction yields exceeding 91% through a novel dual-catalyst system. This technical breakthrough addresses long-standing inefficiencies in previous synthetic routes by replacing expensive or hazardous reagents with a synergistic combination of ammonium acetate and piperidine. For R&D directors and procurement specialists, this innovation signals a shift towards more sustainable and cost-effective manufacturing protocols that do not compromise on chemical purity or structural integrity. The ability to execute this condensation in common industrial solvents like toluene further enhances its viability for commercial scale-up, positioning it as a superior alternative to legacy processes that struggled with reproducibility and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Agomelatine precursors relied heavily on the Reformatsky reaction, a classical organic transformation that, while effective in laboratory settings, presents severe drawbacks when transferred to industrial scales. As illustrated in the prior art, this conventional pathway involves reacting 7-methoxy-1-tetralone with ethyl bromoacetate in the presence of zinc, necessitating the use of benzene as a solvent, which poses significant health and environmental risks. Furthermore, the subsequent aromatization steps in these legacy routes are often incomplete, leading to difficult purification challenges and a cumulative average yield of less than 30%. The reliance on multiple sequential steps, including hydrolysis, acid chloride formation, and dehydration, introduces numerous points of failure and material loss. These inefficiencies not only inflate the cost of goods sold but also create supply chain bottlenecks due to the extended processing time and the handling of hazardous intermediates.
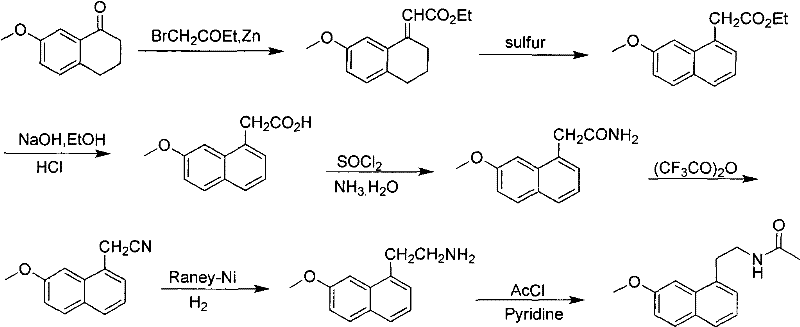
The Novel Approach
In stark contrast to the cumbersome multi-step sequences of the past, the patented method streamlines the synthesis into a direct condensation reaction that dramatically simplifies the operational workflow. By utilizing cyanoacetic acid and 7-methoxy-1-tetralone as starting materials, the process bypasses the need for halogenated intermediates and toxic benzene solvents entirely. The core innovation lies in the specific catalytic environment where ammonium acetate acts in concert with piperidine, creating a synergistic effect that drives the reaction to completion with exceptional efficiency. This approach eliminates the need for harsh dehydration agents and complex workup procedures, allowing for a straightforward isolation of the nitrile intermediate. The result is a process that is not only chemically elegant but also operationally robust, capable of delivering high-purity products suitable for downstream pharmaceutical applications without the burden of extensive purification protocols.
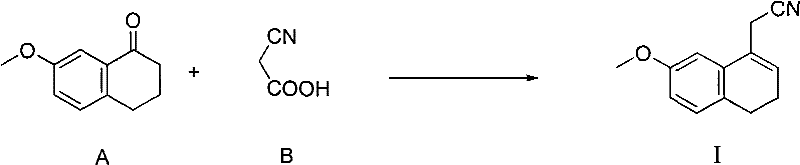
Mechanistic Insights into Dual-Catalyst Condensation
The success of this synthetic route is fundamentally rooted in the mechanistic interplay between the two catalysts, ammonium acetate and piperidine, which facilitate a Knoevenagel-type condensation followed by spontaneous decarboxylation. While literature such as Organic Syntheses Vol. 31 describes similar reactions using ammonium acetate alone, those methods typically suffer from low conversion rates and require extreme conditions. The addition of piperidine, a secondary amine, likely enhances the nucleophilicity of the active methylene group in cyanoacetic acid through the formation of an enamine intermediate, thereby lowering the activation energy for the attack on the carbonyl carbon of the tetralone. This cooperative catalysis ensures that the reaction proceeds rapidly under reflux conditions in toluene, driving the equilibrium forward through the continuous removal of water. Such mechanistic optimization is critical for maintaining high selectivity, minimizing the formation of polymeric byproducts, and ensuring that the final nitrile structure remains intact without unwanted side reactions.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. Traditional routes involving zinc or other transition metals often leave behind trace metal residues that are difficult to remove and can be detrimental to the stability of the final API. The organocatalytic nature of the ammonium acetate and piperidine system means that the reaction mixture is free from heavy metals, simplifying the downstream purification process to basic aqueous washes. The use of toluene as a solvent further aids in impurity management, as it allows for effective phase separation and the removal of polar byproducts during the washing stages. This results in a crude product of such high quality that it often requires minimal additional refinement, directly addressing the concerns of quality assurance teams regarding residual solvents and metallic contaminants in pharmaceutical intermediates.
How to Synthesize (7-methoxy-3,4-dihydro-1-naphthyl) acetonitrile Efficiently
Implementing this synthesis in a production environment requires precise control over stoichiometry and reaction conditions to maximize the synergistic effects of the catalysts. The standard protocol involves charging a reactor with 7-methoxy-1-tetralone, cyanoacetic acid, ammonium acetate, and piperidine in a toluene medium, followed by heating to reflux. The process relies on the azeotropic removal of water to drive the condensation to completion, which is typically monitored via TLC until the starting ketone is fully consumed. Detailed standardized operating procedures regarding specific molar ratios, temperature profiles, and safety precautions are essential for consistent batch-to-batch performance. For a comprehensive guide on the exact step-by-step execution of this protocol, please refer to the technical instructions below.
- Mix 7-methoxy-1-tetralone, cyanoacetic acid, ammonium acetate, and piperidine in toluene solvent.
- Reflux the mixture with water separation until the starting ketone is consumed.
- Cool, wash with sodium hydroxide and brine, dry over magnesium sulfate, and evaporate to obtain the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond simple chemical yield. The elimination of expensive and specialized catalysts, such as benzyl ammonium heptanoate used in other reported methods, drastically reduces the raw material costs associated with the synthesis. Furthermore, the replacement of hazardous solvents like benzene with widely available and cheaper toluene mitigates regulatory compliance costs and simplifies waste disposal logistics. This shift towards greener chemistry not only aligns with modern environmental standards but also insulates the supply chain from volatility in the pricing of specialty reagents. The simplified workup procedure, which avoids complex chromatographic separations, significantly shortens the production cycle time, allowing for faster turnaround and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substitution of high-cost reagents with commodity chemicals. By utilizing ammonium acetate and piperidine, which are inexpensive and readily available in bulk quantities, manufacturers can achieve substantial savings on the bill of materials. Additionally, the high reaction yield minimizes the waste of valuable starting materials, ensuring that a greater proportion of input mass is converted into saleable product. The avoidance of transition metals like zinc removes the need for costly scavenging resins or complex filtration steps required to meet strict heavy metal specifications, further lowering the overall processing expenses.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for uninterrupted API production, and this method strengthens that reliability through ingredient accessibility. The starting materials, 7-methoxy-1-tetralone and cyanoacetic acid, are established commodities with stable global supply chains, reducing the risk of shortages compared to proprietary or niche catalysts. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, leading to more predictable production schedules. This stability allows supply chain planners to maintain leaner inventory levels while confidently meeting delivery commitments to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen engineering challenges, but the simplicity of this condensation reaction facilitates a smooth transition from pilot to commercial scale. The use of toluene, a solvent with well-understood physical properties and handling protocols, simplifies reactor design and thermal management requirements. Moreover, the absence of halogenated solvents and heavy metals significantly reduces the environmental footprint of the manufacturing process, easing the burden on wastewater treatment facilities. This alignment with green chemistry principles not only ensures compliance with increasingly stringent environmental regulations but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into their existing manufacturing portfolios. The following questions address common inquiries regarding the catalyst system, solvent choices, and comparative advantages over legacy methods. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making. For more specific technical data or custom feasibility studies, our team is available to provide detailed consultations tailored to your production requirements.
Q: What is the primary advantage of the dual catalyst system in this synthesis?
A: The combination of ammonium acetate and piperidine significantly improves reaction yield to over 91%, overcoming the low yields associated with single-catalyst systems.
Q: Why is this method preferred over the traditional Reformatsky reaction?
A: Unlike the Reformatsky route which requires hazardous benzene solvents and complex purification, this method uses safer toluene and achieves much higher purity and yield.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common industrial solvents like toluene and inexpensive catalysts, making it highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (7-methoxy-3,4-dihydro-1-naphthyl) acetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands engineering excellence and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this dual-catalyst process are fully realized in practice. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (7-methoxy-3,4-dihydro-1-naphthyl) acetonitrile meets the exacting standards required for API synthesis. Our commitment to technical precision allows us to navigate the complexities of scale-up, delivering a product that is consistent, reliable, and ready for immediate integration into your drug substance manufacturing lines.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through the adoption of this superior synthetic route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits specific to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to verify how this technology can enhance your operational efficiency. Let us partner with you to secure a stable, high-quality supply of this critical intermediate, driving your projects forward with confidence and speed.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
