Advanced Synthesis of 2-Amino-3 5-Dibromopyrazine for Commercial Scale Pharmaceutical Manufacturing
The pharmaceutical industry continuously demands higher purity intermediates to ensure the safety and efficacy of final drug products. Patent CN110878056A introduces a transformative methodology for synthesizing 2-amino-3 5-dibromopyrazine a critical building block in medicinal chemistry. This innovation addresses longstanding challenges in heterocyclic halogenation by replacing hazardous elemental bromine with a safer sodium hypochlorite system coupled with precise temperature control. The technical breakthrough lies not only in the reaction pathway but also in the sophisticated post-treatment protocol that ensures exceptional impurity profiles. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier this patent represents a significant leap forward in process reliability and product quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for dibrominated pyrazines often suffer from severe drawbacks that hinder commercial viability and environmental compliance. Prior art methods frequently rely on direct bromination which generates substantial amounts of acidic wastewater and requires complex neutralization steps. These conventional processes typically result in lower yields due to over-bromination or side reactions that create difficult-to-remove impurities. Furthermore the use of volatile and toxic solvents in older methodologies poses significant occupational health risks and increases the cost of waste disposal. The inability to consistently achieve high purity without extensive chromatographic purification makes these legacy methods unsuitable for modern good manufacturing practice requirements.
The Novel Approach
The novel approach detailed in the patent data utilizes a two-stage strategy that fundamentally optimizes both the reaction kinetics and the purification workflow. By initiating the sequence with the conversion of 2-cyanopyrazine to 2-aminopyrazine under mild alkaline conditions the process establishes a high-purity starting material for the subsequent bromination step. The use of 1 3-dibromo-5 5-dimethylhydantoin as the brominating agent in N N-dimethylacetamide allows for a controlled release of bromine equivalents minimizing side reactions. This strategic choice of reagents combined with a rigorous recrystallization regimen using heptane and ethanol ensures that the final product meets stringent quality specifications without the need for costly chromatography.

Mechanistic Insights into Alkaline Hydrolysis and Controlled Bromination
The first stage of the synthesis involves the nucleophilic attack of hypochlorite on the cyano group of 2-cyanopyrazine in the presence of sodium hydroxide. This reaction is conducted at a strictly controlled temperature range of minus 5 to 0 degrees Celsius to prevent thermal degradation and ensure selective formation of the amine. The mechanistic pathway favors the formation of 2-aminopyrazine with minimal byproduct generation due to the specific molar ratio of reactants maintained between 1:1 and 1:1.5. Following the reaction the mixture is heated to 90 to 100 degrees Celsius to complete the hydrolysis before undergoing reduced pressure distillation to isolate the intermediate. This careful manipulation of thermal conditions is critical for maximizing the yield of the amine precursor which directly impacts the efficiency of the downstream bromination.
The subsequent bromination step leverages the electrophilic nature of the dibromohydantoin reagent to introduce bromine atoms at the 3 and 5 positions of the pyrazine ring. The reaction proceeds in N N-dimethylacetamide which solubilizes both the amine and the brominating agent effectively facilitating molecular collision. Temperature is gradually raised from below 0 degrees Celsius to 15 to 20 degrees Celsius over a period of 15 to 20 hours to ensure complete conversion while suppressing poly-brominated impurities. The resulting crude product is then subjected to a multi-step purification involving pH adjustment to 8 and activated carbon treatment to remove colored impurities. This meticulous attention to mechanistic detail ensures that the final 2-amino-3 5-dibromopyrazine achieves purity levels suitable for sensitive pharmaceutical applications.
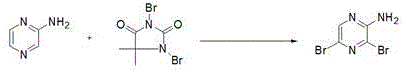
How to Synthesize 2-Amino-3 5-Dibromopyrazine Efficiently
Implementing this synthesis route requires strict adherence to the specified operational parameters to replicate the high yields reported in the patent examples. The process begins with the preparation of the alkaline hypochlorite solution followed by the controlled addition of the cyano precursor. Operators must monitor the exotherm closely during the dropwise addition to maintain the sub-zero temperature window. Once the intermediate is isolated the bromination reaction demands patience as the extended reaction time of up to 20 hours is necessary for full conversion. The final purification steps involving heptane and ethanol recrystallization are essential for removing trace solvents and achieving the desired needle-shaped crystal morphology. Detailed standardized synthesis steps see the guide below.
- Prepare 2-aminopyrazine by reacting 2-cyanopyrazine with sodium hypochlorite and sodium hydroxide at -5 to 0 degrees Celsius.
- Conduct bromination using 1 3-dibromo-5 5-dimethylhydantoin in N N-dimethylacetamide solvent at 15 to 20 degrees Celsius.
- Purify the crude product through extraction pH adjustment and sequential recrystallization using heptane and ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective this patented methodology offers substantial advantages that align with the strategic goals of cost reduction in pharmaceutical intermediate manufacturing. The elimination of hazardous elemental bromine reduces the regulatory burden and insurance costs associated with handling dangerous goods. Additionally the use of common solvents like ethanol and heptane simplifies the supply chain logistics and ensures consistent availability of raw materials. The robustness of the purification process means that batch-to-batch variability is minimized reducing the risk of production delays caused by out-of-specification results. These factors collectively contribute to a more resilient and predictable supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts and complex chromatographic purification steps which are major cost drivers in fine chemical synthesis. By utilizing readily available reagents like sodium hypochlorite and hydantoin derivatives the raw material costs are significantly optimized compared to traditional halogenation routes. The high yield and purity achieved reduce the volume of waste generated per kilogram of product lowering disposal fees. Furthermore the ability to recover and recycle solvents like ethyl acetate and heptane contributes to long-term operational savings.
- Enhanced Supply Chain Reliability: Sourcing 1 3-dibromo-5 5-dimethylhydantoin and 2-cyanopyrazine is straightforward as these are established commodities in the global chemical market. This reduces the risk of supply disruptions that often plague specialized reagent dependent processes. The simplified workflow with fewer unit operations translates to shorter production cycles allowing for faster turnaround times on customer orders. Consequently manufacturers can maintain lower inventory levels while still meeting just-in-time delivery requirements for their clients.
- Scalability and Environmental Compliance: The aqueous workup and crystallization steps are inherently scalable from laboratory benchtop to multi-ton reactors without requiring specialized equipment. The reduction in acidic wastewater generation simplifies effluent treatment and ensures compliance with increasingly strict environmental regulations. The use of activated carbon for decolorization is a proven industrial technique that scales linearly with batch size. This environmental compatibility enhances the corporate sustainability profile of the manufacturing site and facilitates easier permitting for capacity expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific beneficial effects and background technical problems outlined in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of adopting this route for their specific supply chain needs. The answers reflect the objective data provided in the invention disclosure without extrapolation.
Q: How does this novel method improve product purity compared to conventional bromination?
A: The patented process utilizes a specific post-treatment involving pH adjustment to 8 and sequential recrystallization with heptane and ethanol which effectively removes organic impurities and residual solvents achieving purity levels exceeding 99 percent.
Q: What are the environmental advantages of using sodium hypochlorite in this synthesis?
A: By employing sodium hypochlorite in an aqueous alkaline solution the process avoids the use of hazardous elemental bromine significantly reducing wastewater treatment complexity and environmental pollution risks associated with traditional halogenation methods.
Q: Is this synthesis route suitable for large scale industrial production?
A: Yes the method is designed for scalability utilizing common reagents like N N-dimethylacetamide and standard crystallization techniques that facilitate easy filtration and drying making it highly adaptable for commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-3 5-Dibromopyrazine Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced synthesis technology to support your drug development pipeline. As a dedicated CDMO partner we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensuring that your project transitions smoothly from lab to plant. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications including residual solvent analysis and heavy metal testing. We understand that consistency is key in pharmaceutical manufacturing and our quality management systems are designed to deliver batch-after-batch reliability.
We invite you to discuss how this optimized route can benefit your specific project requirements and cost structures. Our technical procurement team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us to request specific COA data and route feasibility assessments for your target molecules. Partnering with us ensures access to cutting-edge synthetic methodologies backed by a commitment to quality and supply security.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
